 Back
BackMicrobial Nutrition, Cultivation, and Measurement of Growth
Study Guide - Smart Notes

Nutrition, Cultivation, and Measurement of Microbial Growth
Requirements for Life
All living cells, including microbes, require certain basic elements and conditions to sustain life and growth. Understanding these requirements is fundamental to microbiology and the cultivation of microorganisms in the laboratory.
Liquid Water: Essential for biochemical reactions and cellular processes.
Carbon: The backbone of organic molecules; required for cell structure and metabolism.
Nutrients: Elements and compounds necessary for cellular function and growth.
Free Energy: Needed to drive metabolic reactions (e.g., from light or chemical sources).
Microbial Nutrition
Microbes require a variety of nutrients, which can be classified as macronutrients, trace elements, and growth factors. The composition of an Escherichia coli cell by dry weight illustrates the relative abundance of these elements.
Macronutrients: Required in large amounts. All cells need C, O, N, H, P, S. Most also require K, Na, Ca, Mg, Cl.
Trace Elements: Inorganic micronutrients, often as enzyme cofactors (e.g., Fe, Se, B, Cu, Mn, Mo, Zn, F, Si, As, Cd, Sr, Ba, V, Co, W, Ni). Requirements vary by species.
Growth Factors: Organic micronutrients such as vitamins, amino acids, purines, and pyrimidines. Vitamins often act as coenzymes. Requirements also vary by species.
Example: Nucleic acids and lipids are approximately 10% and 5% phosphorus by mass, respectively. Evolutionary adaptations to low phosphorus environments may include reducing genome size or ribosome number.
Microbial Cultivation
Culture Media
Microbes are grown in nutrient solutions called media, which can be liquid or solid (solidified with agents like agar). Media are classified based on their chemical composition and purpose.
Defined Media: Precise chemical composition is known. Used for controlled studies.
Complex Media: Contains digests of undefined substances (e.g., yeast, meat, or plant extracts).
Composition of Defined Media:
Carbon: Organic (for heterotrophs) or CO2 (for autotrophs)
Nitrogen: NH4+, NO3-, N2, or organic N
Phosphorus: PO42- or organic P
Sulfur: SO42- or organic S
Other macronutrients: Inorganic salts
Trace elements and growth factors (e.g., vitamins)
pH buffer
Diagnostic Media
Specialized media are used to identify and isolate specific bacteria.
Selective Media: Inhibits growth of unwanted microbes, allowing target bacteria to grow (e.g., bile salts agar selects for bile-resistant bacteria).
Differential Media: Contains indicators that reveal differences between organisms (e.g., blood agar identifies hemolytic bacteria by color change).
Selective & Differential Media: Combines both properties (e.g., MacConkey agar selects for Gram-negative, lactose-fermenting bacteria).
Medium | Type | Purpose |
|---|---|---|
Bile Salts Agar | Selective | Isolates bile-resistant bacteria |
Blood Agar | Differential | Detects hemolysis patterns |
MacConkey Agar | Selective & Differential | Isolates Gram-negative, lactose fermenters |
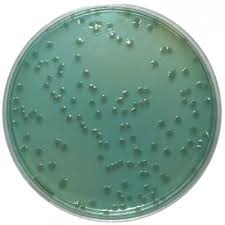
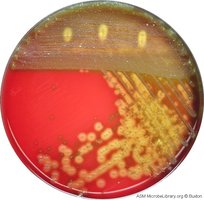
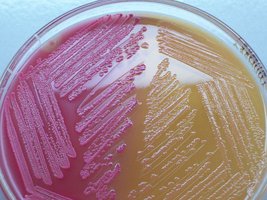
Approaches to Microbial Cultivation
Enrichment Culture: Uses media that provide a selective advantage to the target microbe, increasing its abundance relative to others. Often results in a mixed culture dominated by the target organism.
Direct Isolation: Uses selective/differential agar and streak or spread plate techniques to isolate single colonies, leading to pure cultures.
Example: Enrichment of aerobic N2-fixing bacteria using media lacking nitrogen sources other than atmospheric N2.
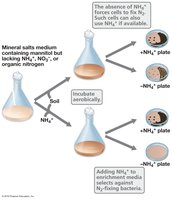
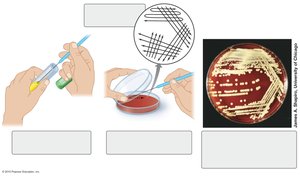
Streak Plate and Spread Plate Methods
These are standard techniques for isolating and counting bacteria.
Streak Plate: Involves spreading a loopful of culture over the surface of an agar plate in a pattern that thins out the sample and leads to isolated colonies.
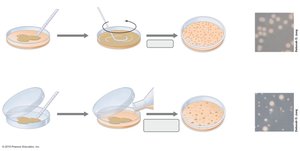
Spread Plate: A diluted sample is spread evenly over the surface of an agar plate using a sterile spreader.
Pour Plate: The sample is mixed with molten agar and poured into a plate, allowing colonies to grow both on the surface and within the medium.
Measuring Microbial Growth
Microscopic Counts
Direct microscopic counts involve counting cells under a microscope. This method is labor-intensive, subject to human error, and does not distinguish between live and dead cells.

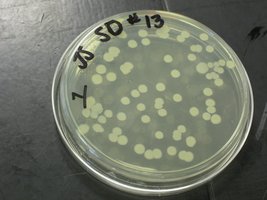
Viable Counts (Plate Counts)
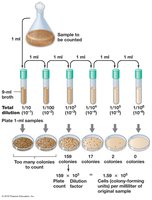
Viable counts estimate the number of living cells by counting colony-forming units (CFU) after plating diluted samples. The assumption is that each colony arises from a single cell, though this may not hold for filamentous or clumping organisms.
Serial Dilution: The sample is diluted in steps to obtain countable numbers of colonies (30-300 per plate is ideal).
Spread Plating: Diluted samples are spread on agar plates and incubated.
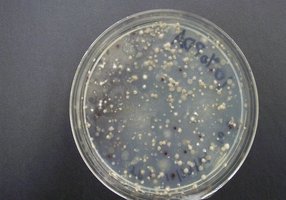
The Great Plate Anomaly: Direct microscopic counts of natural samples are often 10-1000 times higher than viable counts, due to the presence of non-culturable cells.
Spectrophotometry (Turbidity Measurement)
Spectrophotometry measures the turbidity (cloudiness) of a microbial culture, which correlates with cell density. Light scattering by cells is measured as optical density (OD) or absorbance. This method is rapid, non-destructive, and suitable for monitoring growth over time, but requires a standard curve for accurate cell number estimation.
Measurement Sensitivity: Varies with wavelength (e.g., 450 nm is more sensitive than 660 nm).
Cell Shape: Affects light scattering, so OD is a relative measure.
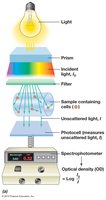
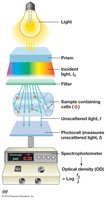
Equation for Optical Density (OD):
Where is the incident light intensity and is the transmitted light intensity.
Summary Table: Methods for Measuring Microbial Growth
Method | Measures | Advantages | Limitations |
|---|---|---|---|
Microscopic Count | Total cells (live & dead) | Direct, fast | Laborious, human error, can't distinguish live/dead |
Viable Count | Living cells (CFU) | Counts only live cells | Misses non-culturable cells, time-consuming |
Spectrophotometry | Cell density (OD) | Fast, non-destructive | Relative measure, affected by cell shape |
Additional info: For accurate quantification, always use appropriate controls and standard curves, and be aware of the limitations of each method.
