 Back
BackPractical Applications of Immunology: Vaccines and Monoclonal Antibodies
Study Guide - Smart Notes

Practical Applications of Immunology
Vaccines: History and Principles
Vaccines are one of the most effective tools in preventing infectious diseases. Their development is rooted in historical practices and scientific advancements.
Variolation: An early method for smallpox prevention, involving the inoculation of material from dried smallpox scabs into the respiratory tract or skin (practiced in China, 1400s–1700s).
Jenner's Contribution: In 1798, Edward Jenner used cowpox to immunize against smallpox, leading to the term vaccination (from Latin vacca, meaning cow).
Vaccine Definition: A suspension of organisms or their fractions that induce immunity.
Principles and Effects of Vaccination
Vaccination stimulates the immune system to recognize and combat pathogens.
Primary Immune Response: Initial exposure leads to antibody and memory cell formation.
Secondary Response: Subsequent exposures provoke a rapid, intense immune reaction.
Herd Immunity: When most of a population is immune, outbreaks are rare due to a lack of susceptible hosts.
CDC-Recommended Vaccines
The CDC recommends various vaccines to prevent bacterial and viral diseases. These vaccines differ in their composition and administration schedules.
Bacterial Diseases
Disease(s) | Vaccine | Recommendation | Booster |
|---|---|---|---|
H. influenzae type b meningitis | Polysaccharide from H. influenzae type b | Children 2–18 months | None |
Meningococcal A C W Y meningitis | Purified polysaccharide from Neisseria meningitidis | Age 11–12 years | At 16 years; outbreak settings |
Pneumococcal pneumonia | Polysaccharide from Streptococcus pneumoniae | Adults with chronic diseases, over 65; children 2–18 months | None if first dose ≥24 months |
Tetanus, diphtheria, pertussis | DTaP, Tdap, Td | Children 2–18 months, 4–6 years; Tdap for 11–12 years and adults | Tdap or Td every 10 years |
Viral Diseases
Disease(s) | Vaccine | Recommendation | Booster |
|---|---|---|---|
Chickenpox (varicella) | Attenuated virus | Infants 12 months | Unknown |
COVID-19 | mRNA | Two-dose series, ages vary | Recommended |
Hepatitis B | Virus antigen in yeast cells | Infants, children, high-risk adults | At least 7 years; boosters uncertain |
Influenza | Inactivated or attenuated virus | Everyone over 6 months | Annual |
Measles, Mumps, Rubella | Attenuated virus | Infants 15 months; adults if exposed | During outbreaks |
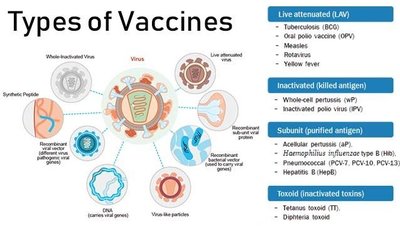
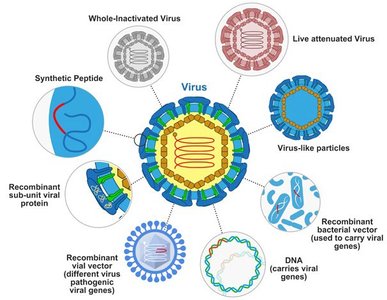
Types of Vaccines and Their Characteristics
Vaccines can be classified based on their composition and method of action.
Attenuated Vaccines: Contain weakened pathogens; mimic natural infection, confer lifelong immunity, but not for immunocompromised individuals due to risk of reversion to virulence.
Inactivated Vaccines: Contain killed microbes; safer but require boosters and induce mainly humoral immunity.
Subunit Vaccines: Use antigenic fragments; include recombinant vaccines (produced by genetic modification), toxoids (inactivated toxins), and virus-like particle (VLP) vaccines.
Polysaccharide Vaccines: Made from pathogen capsules; not very immunogenic, often used with protein carriers (conjugated vaccines) for young children.
Nucleic Acid Vaccines: DNA or mRNA vaccines direct host cells to produce antigenic proteins, stimulating both humoral and cellular immunity.
Recombinant Vector Vaccines: Use genetically modified avirulent viruses or bacteria to deliver DNA coding for antigens.
Nucleic Acid Vaccines
Nucleic acid vaccines represent a modern approach to immunization, utilizing genetic material to induce an immune response.
DNA Vaccines: Injected DNA (naked or encapsulated) is taken up by muscle cells, which then produce the encoded antigen, stimulating immunity.
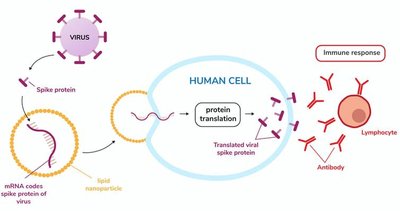
mRNA Vaccines: mRNA is delivered in lipid nanoparticles, directing cells to synthesize the antigen (e.g., COVID-19 vaccines produce spike protein).
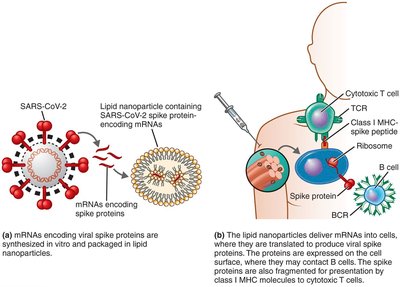
Recombinant Vector Vaccines
These vaccines use genetically engineered viruses or bacteria to deliver genes encoding antigens, stimulating an immune response without causing disease.
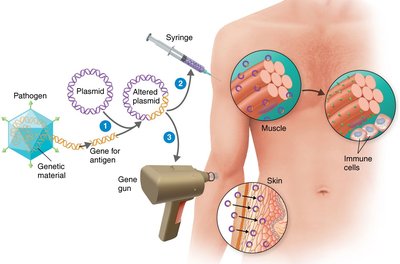
Vaccine Production, Administration, and Safety
Production
Modern vaccines (DNA, mRNA, recombinant vector) do not require animal hosts for pathogen growth.
Plants are being explored as production systems.
Adjuvants
Substances added to vaccines to enhance immune response (e.g., alum, monophosphoryl lipid A).
Activate innate immunity via Toll-like receptors.
Administration
Oral vaccines are effective for GI pathogens (e.g., rotavirus, cholera).
Nasal vaccines (e.g., influenza) and skin patch vaccines (Nanopatch™) are alternatives.
Multiple-combination vaccines simplify immunization schedules.
Safety
Vaccines are among the safest and most effective public health tools.
Rarely, vaccines can cause disease (e.g., oral polio vaccine).
No scientific evidence links MMR vaccines to autism.
mRNA vaccines have an outstanding safety record for COVID-19 prevention.
Impact of Vaccines on Disease
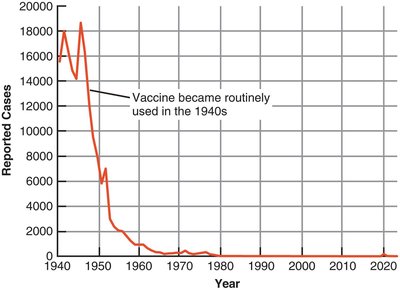
Vaccination campaigns have dramatically reduced the incidence of many infectious diseases.
Diphtheria: Over 15,000 deaths in 1921 (USA); only 14 cases since 1996 due to vaccination.
Challenges: Some diseases persist due to low vaccination rates, need for boosters, or vaccine hesitancy.
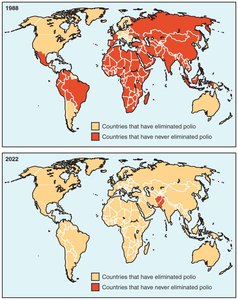
Global Initiatives: Measles and Rubella Initiative, Global Polio Eradication Initiative (polio remains only in Afghanistan and Pakistan).
Use of Monoclonal Antibodies
Production and Applications
Monoclonal antibodies (Mabs) are laboratory-produced molecules engineered to serve as substitute antibodies that can restore, enhance, or mimic the immune system's attack on cells.
Hybridoma Technology: Fusion of an "immortal" myeloma cell with an antibody-producing B cell creates a hybridoma, which produces large quantities of identical antibodies.
Specificity: Mabs are highly specific, targeting a single antigen epitope.
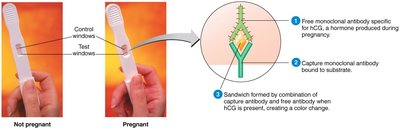
Applications: Used in diagnostics (e.g., pregnancy tests), and therapies for diseases such as multiple sclerosis, Crohn’s disease, psoriasis, cancer, asthma, arthritis, and COVID-19.
Limitations: Mabs derived from mouse cells can cause side effects in humans.
Additional info: Monoclonal antibodies are also used in research and as targeted therapies in oncology and autoimmune diseases.
