 Back
BackThe Chemistry of Microbiology: Foundations for Life and Cellular Function
Study Guide - Smart Notes

Chapter 2: The Chemistry of Microbiology
Atoms and Atomic Structure
The study of microbiology requires an understanding of the chemical foundations of life, beginning with atoms and their structure. Atoms are the smallest units of matter, and their arrangement determines the properties of elements and molecules.
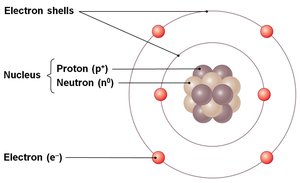
Atom: The basic unit of matter, composed of a nucleus (containing protons and neutrons) and electrons orbiting in shells.
Element: A substance consisting of only one type of atom.
Atomic Number: The number of protons in the nucleus, defining the element.
Atomic Mass: The sum of protons and neutrons (electrons contribute negligibly).
Isotopes: Atoms of the same element with different numbers of neutrons; some are radioactive and undergo decay.
Electron Configuration: Electrons occupy shells around the nucleus; the outermost shell (valence shell) determines chemical reactivity.
Reactivity: Atoms with incomplete valence shells tend to gain, lose, or share electrons to achieve stability.
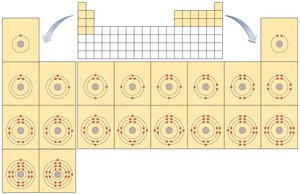
Common Elements of Life: Carbon, hydrogen, oxygen, nitrogen, phosphorus, sulfur, and others are essential for biological molecules.
Chemical Bonds
Chemical bonds are formed when atoms interact to achieve stable electron configurations. The type of bond influences the properties of molecules and their biological functions.
Molecule: Two or more atoms held together by chemical bonds.
Compound: A molecule containing atoms of more than one element.
Types of Chemical Bonds:
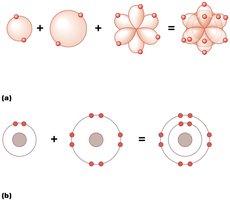
Nonpolar Covalent Bonds: Electrons are shared equally between atoms (e.g., O2).
Polar Covalent Bonds: Electrons are shared unequally, resulting in partial charges (e.g., H2O).
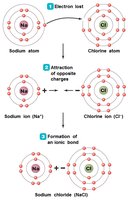
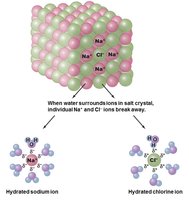
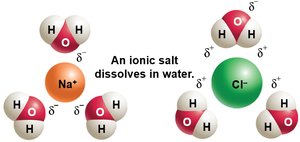
Ionic Bonds: Electrons are transferred from one atom to another, forming charged ions (e.g., NaCl).
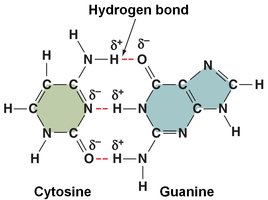
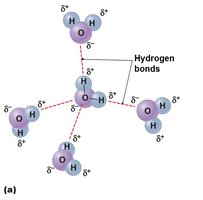
Hydrogen Bonds: Weak attractions between a partially positive hydrogen and a partially negative atom (e.g., between water molecules, DNA base pairs).
Electronegativity: The tendency of an atom to attract electrons; differences in electronegativity drive bond formation.
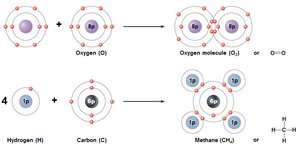
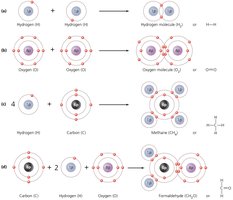

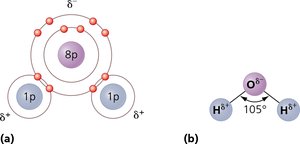

Type of Bond | Description | Relative Strength |
|---|---|---|
Nonpolar covalent | Pair of electrons is nearly equally shared between two atoms | Strong |
Polar covalent | Electrons spend more time around the more electronegative of two atoms | Strong |
Ionic | Electrons are stripped from a cation by an anion | Weaker than covalent in aqueous environments |
Hydrogen | Partial positive charges on hydrogen atoms are attracted to full and partial negative charges on other molecules or other regions of the same molecule | Weaker than ionic |
Chemical Reactions
Chemical reactions involve the making or breaking of bonds, transforming reactants into products. These reactions are fundamental to metabolism and cellular processes.
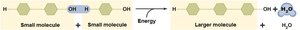
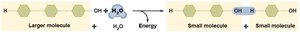
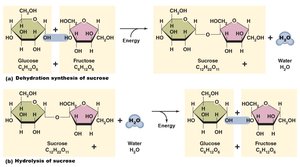
Synthesis Reactions: Combine smaller molecules into larger, more complex ones. Dehydration synthesis releases water and stores energy. Also called anabolism.
Decomposition Reactions: Break down larger molecules into smaller components. Hydrolysis adds water and releases energy. Also called catabolism.
Exchange Reactions: Atoms or groups are transferred between molecules, breaking and forming new bonds.
Metabolism: The sum of all chemical reactions in an organism.
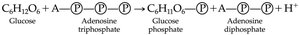
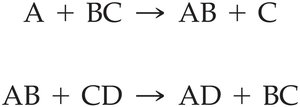
Water, Acids, Bases, and Salts
Inorganic molecules such as water, acids, bases, and salts are essential for life. Their properties influence cellular function, chemical reactions, and environmental adaptation.
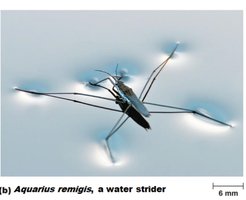
Water: Makes up 50–99% of living organisms' mass. Cohesive due to hydrogen bonds, creating surface tension. Excellent solvent and participates in many reactions.
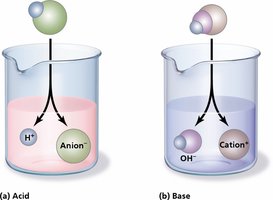
Acids: Dissociate to release hydrogen ions (H+).
Bases: Dissociate to release hydroxyl ions (OH–) and bind H+.
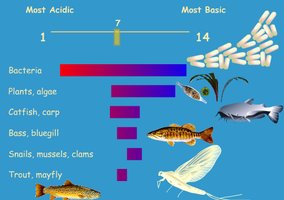
pH Scale: Measures the concentration of H+; organisms tolerate only a narrow pH range.
Buffers: Substances that prevent drastic changes in pH by absorbing excess H+ or OH–.
Salts: Compounds that dissociate into cations and anions in water; important for electrical gradients and electron transfer.

Organic Macromolecules
Organic macromolecules are large, complex molecules essential for metabolism and cellular structure. They are composed of carbon frameworks with functional groups and are classified into four main types.
Functional Groups: Specific arrangements of atoms that confer characteristic properties (e.g., hydroxyl, carboxyl, amino).
Monomers and Polymers: Macromolecules are built from monomers linked by covalent bonds to form polymers.
Types: Lipids, carbohydrates, proteins, nucleic acids.

Lipids
Lipids are hydrophobic molecules composed mainly of carbon and hydrogen. They serve as energy storage, structural components, and signaling molecules.
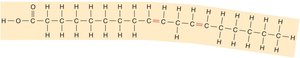
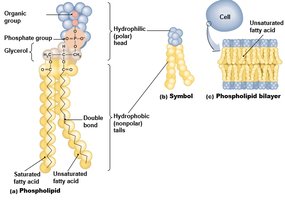
Fatty Acids: Saturated (single bonds, solid, animal origin) and unsaturated (double bonds, liquid, plant origin).
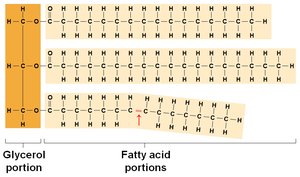
Triglycerides: Glycerol + 3 fatty acids; most abundant lipid, high energy content.
Phospholipids: Glycerol + 2 fatty acids + phosphate; amphipathic, major component of cell membranes.
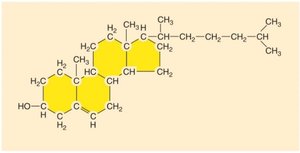
Steroids: Four-ring structure; includes cholesterol, hormones, and membrane components.
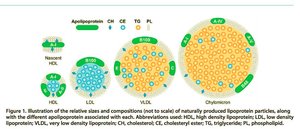
Cholesterol: LDL (bad, forms plaques) and HDL (good, removes cholesterol).
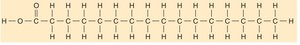
Carbohydrates
Carbohydrates are composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. They provide energy, structural support, and are involved in cell recognition.
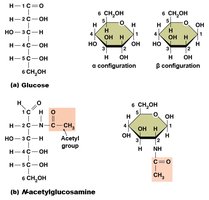
Monosaccharides: Simple sugars (glucose, fructose), usually cyclic, classified by number of carbons.
Disaccharides: Two monosaccharides linked by dehydration synthesis (e.g., sucrose, lactose).
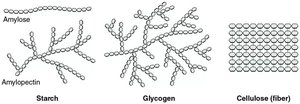
Polysaccharides: Long chains of monosaccharides; storage (glycogen, starch) and structural (cellulose).
Proteins
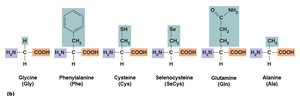
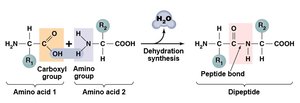
Proteins are polymers of amino acids and are the most complex macromolecules. They serve structural, enzymatic, transport, and defensive roles.
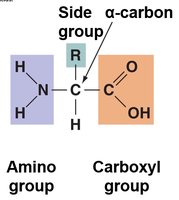
Amino Acids: Central carbon, amino group, carboxyl group, hydrogen, and variable side group (R).
Peptide Bonds: Link amino acids via dehydration synthesis.
Protein Structure: Primary (sequence), secondary (folding), tertiary (3D shape), quaternary (multiple polypeptides).
Denaturation: Loss of structure leads to loss of function.
Conjugated Proteins: Glycoproteins, lipoproteins, nucleoproteins.
Nucleotides and Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides and carry genetic information. Nucleotides consist of a phosphate, pentose sugar, and nitrogenous base.
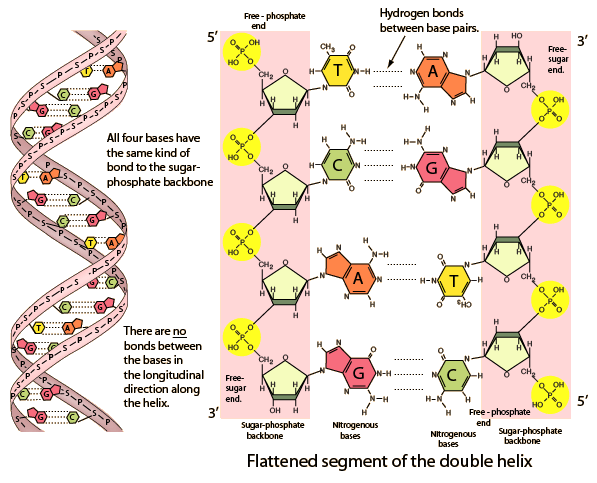
DNA: Double-stranded, antiparallel, held by hydrogen bonds (A-T, G-C).
RNA: Single-stranded, diverse functions in protein synthesis.
Function: Store and transmit genetic information, guide protein synthesis.
ATP (Adenosine Triphosphate)
ATP is the primary short-term energy storage molecule in cells, composed of adenine, ribose, and three phosphate groups. Hydrolysis of ATP releases energy for cellular processes.
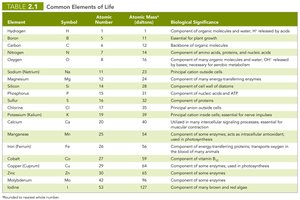
Summary Table: Common Elements of Life
Element | Symbol | Atomic Number | Atomic Mass | Biological Significance |
|---|---|---|---|---|
Hydrogen | H | 1 | 1 | Component of organic molecules and water; H+ released by acids |
Carbon | C | 6 | 12 | Backbone of organic molecules |
Nitrogen | N | 7 | 14 | Component of amino acids, proteins, and nucleic acids |
Oxygen | O | 8 | 16 | Component of water and organic molecules; O2 required for aerobic respiration |
Phosphorus | P | 15 | 31 | Component of nucleic acids and ATP |
Sulfur | S | 16 | 32 | Component of some amino acids and vitamins |
Others | - | - | - | Trace elements required for enzyme function |
Additional info: These notes expand on brief points with academic context, definitions, and examples to ensure completeness and clarity for microbiology students.
