 Back
BackThe Control of Microbial Growth: Principles and Methods
Study Guide - Smart Notes

Chapter 7: The Control of Microbial Growth
Introduction
The control of microbial growth is essential in healthcare, food safety, and laboratory settings. This chapter explores the terminology, mechanisms, and methods used to control or eliminate microorganisms, including both physical and chemical approaches.
How do we control their growth and grow when we want it to
Terminology of Microbial Control
Key Definitions
Sepsis: Refers to bacterial contamination by a significant amount of bacteria. Body is overwhelmed.
Asepsis: The absence of significant contamination; aseptic techniques prevent microbial contamination of wounds.
not sterile,
Sterilization: The removal or destruction of all microbial life, including endospores.
Commercial Sterilization: The process of killing Clostridium botulinum endospores in canned goods.
Disinfection: The destruction of harmful microorganisms on inanimate surfaces.
Antisepsis: The destruction of harmful microorganisms from living tissue.
Degerming: Mechanical removal of microbes from a limited area (e.g., skin before injection).
Sanitization: Lowering microbial counts on eating utensils to safe levels.
Biocide (Germicide): Treatments that kill microbes.
Bacteriostasis: Inhibiting, not killing, microbes.
The Rate of Microbial Death
Patterns and Factors Affecting Microbial Death
Microbial death occurs at a constant logarithmic rate when exposed to microbial control agents.
Effectiveness of treatment depends on:
Number of microbes present
Environmental factors (organic matter, temperature, biofilms)
Time of exposure
Microbial characteristics (e.g., endospores, cell wall structure)
Actions of Microbial Control Agents
Mechanisms of Action
Damage to plasma membrane: Causes leakage of cellular contents and interferes with cell growth.
Damage to proteins (enzymes): Denaturation or inactivation of essential enzymes.
Damage to nucleic acids: Prevents replication and normal cellular function.
Physical Methods of Microbial Control
Heat
Heat denatures enzymes, leading to microbial death.
Thermal death point (TDP): Lowest temperature at which all cells in a liquid culture are killed in 10 minutes.
Thermal death time (TDT): Minimal time for all bacteria in a liquid culture to be killed at a given temperature.
Decimal reduction time (DRT): Minutes to kill 90% of a specific population of bacteria at a given temperature.
Moist Heat Sterilization
Coagulates and denatures proteins.
Methods include boiling, free-flowing steam, and autoclaving.
Autoclave: Uses steam under pressure (15 psi, 15 min) to kill all organisms (except prions) and endospores. Steam must contact the item’s surface. Large containers require longer times. Test strips indicate sterility.
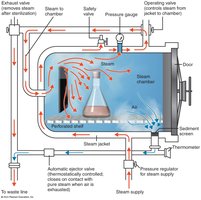
Pasteurization
Reduces spoilage organisms and pathogens in liquids (milk, juices).
High-temperature short-time (HTST): 72°C for 15 sec.
Ultra-high-temperature (UHT): 140°C for 4 sec, followed by rapid cooling; sterilizes milk and juices for non-refrigerated storage.
Dry Heat Sterilization
Kills by oxidation (flaming, incineration, hot-air oven at 170°C for 2 hours).
Filtration
Passage of a substance through a screenlike material; used for heat-sensitive materials.
HEPA filters: Remove microbes >0.3 μm.
Membrane filters: Remove microbes >0.22 μm; some can filter out viruses and large proteins.
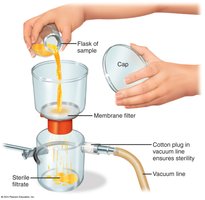
Other Physical Methods
Low temperature: Bacteriostatic effect (refrigeration, deep-freezing, lyophilization).
High pressure: Denatures proteins and alters carbohydrate structure.
Desiccation: Absence of water prevents metabolism.
Osmotic pressure: High concentrations of salts and sugars create a hypertonic environment, causing plasmolysis.
Radiation
Ionizing radiation (X-rays, gamma rays, electron beams): Ionizes water to create reactive hydroxyl radicals, damaging DNA and causing lethal mutations. Used for sterilizing pharmaceuticals, medical supplies, and food.
Nonionizing radiation (UV, 260 nm): Damages DNA by creating thymine dimers; effective for surface sterilization.
Visible blue light (470 nm): Kills bacteria via singlet oxygen formation.
Microwaves: Kill by heat, not especially antimicrobial.
Chemical Methods of Microbial Control
Principles of Effective Disinfection
Effectiveness depends on concentration, presence of organic matter, pH, temperature, and time of exposure.
The Disk-Diffusion Method
Evaluates efficacy of chemical agents using filter paper disks on microbial cultures; zones of inhibition indicate effectiveness.
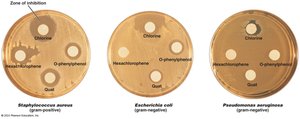
Major Types of Chemical Agents
Phenol and Phenolics: Disrupt plasma membranes; remain active in organic matter. Example: O-phenylphenol (Lysol®).

Bisphenols: Two phenol groups connected by a bridge; disrupt plasma membranes. Examples: Hexachlorophene, triclosan.
Biguanides: Disrupt plasma membranes; effective against gram-positive, many gram-negative bacteria, and enveloped viruses. Example: Chlorhexidine.
Essential Oils: Plant extracts with antimicrobial activity, mainly against gram-positive bacteria and fungi.
Halogens: Iodine (impairs protein synthesis, alters membranes; used as tincture or iodophor) and chlorine (oxidizing agent; used in bleach and water disinfection).
Alcohols: Denature proteins and dissolve lipids; ineffective against endospores and nonenveloped viruses. Examples: Ethanol, isopropanol.
Heavy Metals: Oligodynamic action; denature proteins. Examples: Silver nitrate, mercuric chloride, copper sulfate, zinc chloride.
Surface-Active Agents: Soaps and detergents; aid in mechanical removal of microbes.
Chemical Food Preservatives: Sulfur dioxide, organic acids, nitrites/nitrates; inhibit microbial growth in foods.
Antibiotics (for food preservation): Bacteriocins such as nisin and natamycin prevent spoilage in cheese.
Aldehydes: Inactivate proteins by cross-linking; glutaraldehyde is a liquid sterilant.
Gaseous Chemosterilants: Sterilize by alkylation; examples include ethylene oxide and chlorine dioxide.
Plasma: Electrically excited gas with free radicals that destroy microbes; used for sterilizing tubular instruments.
Supercritical Fluids: Used for sterilizing food and medical implants.
Peroxygens: Oxidizing agents such as hydrogen peroxide and peracetic acid; used for disinfecting surfaces and equipment.
Microbial Characteristics and Microbial Control
Microbial Resistance
Gram-negative bacteria: More resistant to biocides due to their outer membrane.
Pseudomonas and Burkholderia: Highly resistant.
Mycobacteria: Considerable resistance; require special testing for tuberculocidal activity.
Bacterial endospores: Very resistant to many biocides.
Nonenveloped viruses: More resistant than enveloped viruses.
Prions: Extremely resistant; require immersion in NaOH and autoclaving for 1 hour.
Comparative Effectiveness of Antiseptics
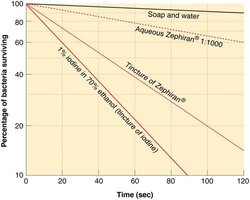
This graph demonstrates the percentage of bacteria surviving over time when exposed to different antiseptics, highlighting the relative effectiveness of each agent.
Additional info: Where necessary, academic context and definitions have been expanded for clarity and completeness. Tables referenced in the original material (e.g., Table 7-6, Table 7-7) are not included due to lack of specific data in the provided text.
