 Back
BackThe Innate Immune System: Structure, Function, and Cellular Components
Study Guide - Smart Notes

Introduction to the Immune System
Definition and Overview
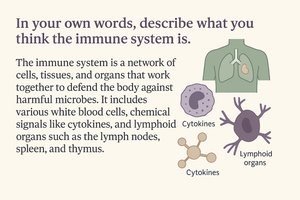
The immune system is a complex network of cells, tissues, and organs that work together to defend the body against infection and disease. It recognizes and destroys bacteria, viruses, fungi, and abnormal cells such as cancer cells, and also removes infected or damaged host cells. The immune system is divided into two main branches: innate immunity and adaptive immunity.
Microbe-Host Interactions
Host-Pathogen Dynamics
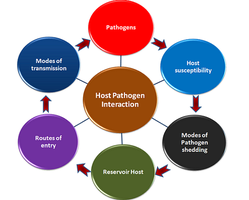
Microbe-host interactions involve complex relationships between beneficial and harmful microbes and the human body. Many bacterial structures act as danger signals, alerting the immune system to potential threats.
Three Lines of Host Defense Against Microbes
Overview of Defense Mechanisms
First Line: Physical and chemical barriers that prevent microbial entry.
Second Line: Innate immune defenses that are rapid and nonspecific.
Third Line: Adaptive immunity, which is targeted and has memory.

First Line of Defense: Physical & Chemical Barriers
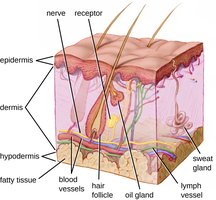
Physical Barriers: Skin & Mucous Membranes
The skin and mucous membranes form the body's outer protective layer, preventing pathogen entry. These barriers include:
Skin: Multilayered structure with constant shedding of dead cells, removing attached microbes.
Mucous membranes: Line body cavities and secrete mucus to trap pathogens.
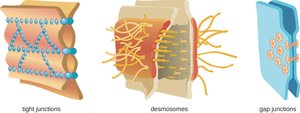
Cell junctions: Tight junctions, desmosomes, and gap junctions seal tissues and prevent pathogen entry.
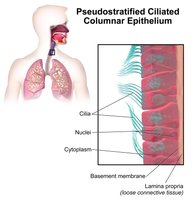
Mucociliary Escalator
The mucociliary escalator is a system in the respiratory tract where ciliated cells move mucus upward, trapping microbes and debris and moving them to the esophagus for removal.

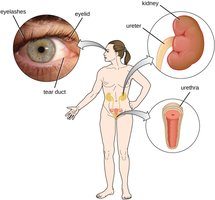
Mechanical Barriers: Flushing Actions
Secretions such as tears and urine physically wash pathogens away before they can attach. Tears and saliva also contain lysozyme, an enzyme that breaks down bacterial cell walls.
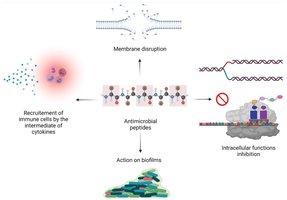
Chemical Barriers: Antimicrobial Peptides (AMPs)
AMPs, Bacteriocins, and Defensins
Antimicrobial peptides (AMPs) are fast-acting, broad-spectrum chemical defenses produced by epithelial and immune cells. They disrupt microbial membranes, inhibit intracellular processes, recruit immune cells, and prevent biofilm formation. Bacteriocins are AMPs produced by bacteria to kill closely related species, while defensins are host-made peptides that disrupt microbial membranes and recruit immune cells.
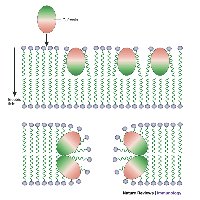
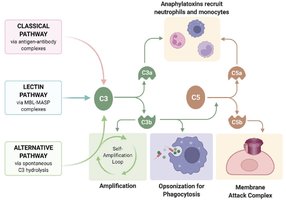
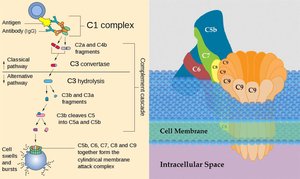
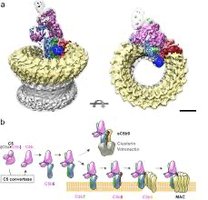
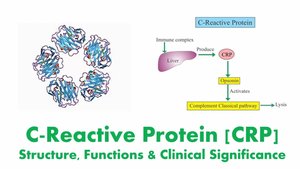
The Complement System
Plasma Protein Cascade
The complement system is a group of about 30 plasma proteins activated in a cascade. It enhances inflammation, opsonizes microbes, and forms the membrane attack complex (MAC) to lyse cells. The classical pathway is triggered by antibody binding, leading to C3 activation and formation of the MAC.
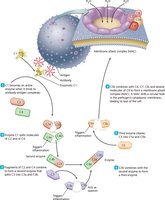
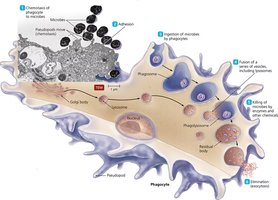
Phagocytosis
Mechanism and Steps
Phagocytosis is the process by which neutrophils, macrophages, and dendritic cells engulf and destroy microbes. Steps include chemotaxis, adherence, ingestion, digestion (lysosome fusion), and exocytosis of debris. Recognition is enhanced by pattern recognition receptors (PRRs) or complement opsonins (e.g., C3b).
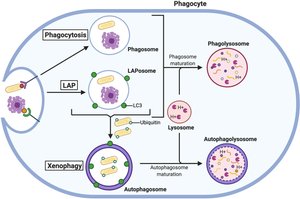
The Phagolysosome
The phagolysosome forms when a phagosome fuses with a lysosome, enabling enzymatic and acidic killing of pathogens. Acidification to pH 5–5.5 activates lysosomal enzymes.
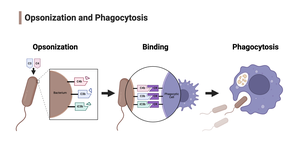
Opsonization
Opsonization is the process by which complement protein C3b coats microbes, making them easier for phagocytes to ingest. This is especially important for clearing encapsulated bacteria, as capsules can hide surface antigens.
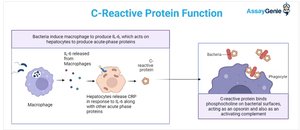
Acute Phase Proteins: C-reactive Protein (CRP)
Role in Innate Immunity
C-reactive protein (CRP) is an acute-phase protein produced by the liver during inflammation. It binds microbial surfaces, enhances complement activation, and acts as an opsonin to help phagocytes clear pathogens.
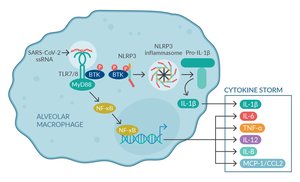
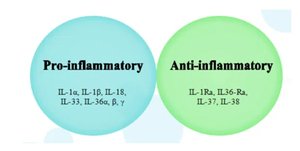
Cytokines: Immune Cell Communication
Types and Functions
Cytokines are small signaling proteins released by immune cells to coordinate responses. They regulate inflammation, cell activation, and communication between immune cells. Major groups include interleukins, chemokines, and interferons.
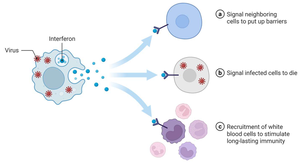
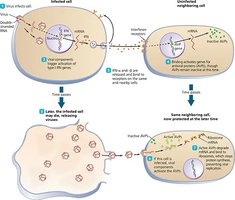
Interferons: Key Antiviral Cytokines
Type I interferons (IFN-α, IFN-β) are released by virus-infected cells and induce antiviral proteins in nearby cells, inhibiting viral replication and activating NK cells. Type II interferon (IFN-γ) is produced by T cells and NK cells, activating macrophages and regulating immune responses.
Inflammation-Eliciting Mediators
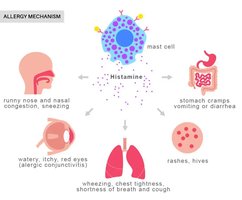
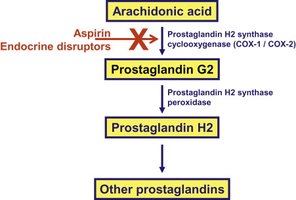
Histamines and Prostaglandins
Histamines are released by mast cells and basophils during inflammation and allergic reactions, causing vasodilation and increased vascular permeability. Prostaglandins are lipid mediators produced from arachidonic acid, increasing vasodilation and contributing to pain, fever, and swelling.
Leukocytes vs. Lymphocytes
Classification of Immune Cells
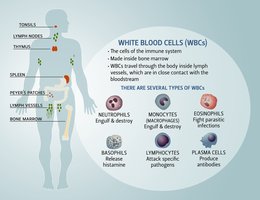
Leukocytes are all white blood cells, while lymphocytes are a specific subgroup (B cells, T cells, NK cells). All lymphocytes are leukocytes, but not all leukocytes are lymphocytes.
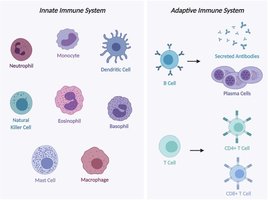
Innate and Adaptive Immune Cells
Myeloid and Lymphoid Lineages
Innate immune cells (neutrophils, macrophages, dendritic cells, eosinophils, basophils, mast cells) derive from the myeloid lineage and provide rapid, nonspecific responses. Adaptive immune cells (B cells, T cells, plasma cells) derive from the lymphoid lineage and provide antigen-specific, slower but stronger immunity.
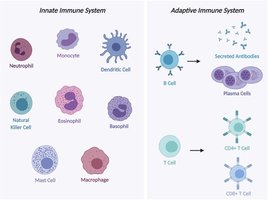
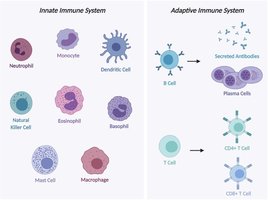
Granulocytes vs. Agranulocytes
Granulocytes (neutrophils, eosinophils, basophils) have visible cytoplasmic granules, while agranulocytes (lymphocytes, monocytes) do not. Granules store toxic chemicals used to fight pathogens.
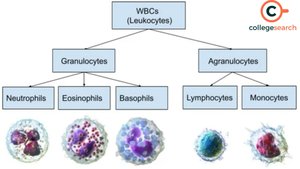
Hematopoiesis
Hematopoiesis is the process of making all blood cells from hematopoietic stem cells (HSCs) in the bone marrow. It produces myeloid (innate cells, RBCs, platelets) and lymphoid (B, T, NK cells) lineages.
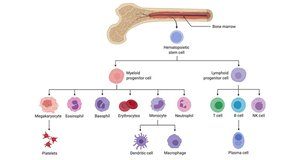
Major Players of the Innate Immune System
Neutrophils
Neutrophils are the most common WBCs and first responders to infection. They perform phagocytosis, release antimicrobial enzymes, and form pus when they die after battling microbes.
Eosinophils
Eosinophils specialize in fighting parasites and contribute to allergic responses by releasing toxic granules that damage parasites and contribute to allergy-related tissue damage.
Basophils and Mast Cells
Basophils and mast cells drive allergic reactions and inflammation. Mast cells release histamine and other mediators that recruit eosinophils.
Monocytes and Macrophages
Monocytes circulate in the blood as immature phagocytes and differentiate into macrophages or dendritic cells in tissues. Macrophages are long-lived phagocytes that engulf microbes, release cytokines, and present antigens to activate adaptive immunity.
Dendritic Cells
Dendritic cells are professional antigen-presenting cells that sample tissues for pathogens and activate naïve T cells, linking innate and adaptive immunity.
Natural Killer (NK) Cells
NK cells provide rapid defense against infected or abnormal cells by detecting low MHC I expression and releasing perforin and granzymes to induce apoptosis. They are important for killing virally infected and cancerous cells.
Inflammation and Innate Immunity
Definition and Process
Inflammation is the body's rapid response to damage or infection, characterized by increased vascular permeability, recruitment of immune cells, and induction of fever. It is triggered by PAMPs or DAMPs, sensed by PRRs, leading to cytokine/chemokine production and immune cell activation. Controlled inflammation is protective, but uncontrolled inflammation can cause tissue damage.
Pattern Recognition and Microbial Detection
Pattern Recognition Receptors (PRRs)
PRRs detect danger signals such as pathogen-associated molecular patterns (PAMPs) from microbes and damage-associated molecular patterns (DAMPs) from damaged host cells. Five main classes of PRRs include:
Toll-like receptors (TLRs): Detect LPS, flagellin, peptidoglycan, viral RNA, bacterial DNA.
C-type lectin receptors (CLRs): Recognize carbohydrates on fungi and some bacteria.
RIG-I-like receptors (RLRs): Detect viral RNA in the cytosol.
NOD-like receptors (NLRs): Sense bacterial cell wall fragments and cell stress signals.
AIM2-like receptors (ALRs): Recognize cytosolic double-stranded DNA.
PRR-PAMP Interaction Example: LPS-TLR4
LPS, a major PAMP on Gram-negative bacteria, is recognized by TLR4. LBP transfers LPS to CD14, which then presents it to the TLR4–MD2 complex, activating NF-κB signaling and cytokine production.
Summary
Most microbes fail to cause infection due to physical and chemical barriers. When microbes enter, the innate immune system recognizes conserved microbial structures via PRRs, rapidly triggering inflammation and immune cell recruitment. Innate immune cells use strategies such as phagocytosis, degranulation, and cytotoxic killing. Inflammation and fever help contain infection but must be regulated to prevent tissue damage. The innate immune response provides critical early defense and shapes the adaptive immune response.
