 Back
BackViral Molecular Biology: Structure, Replication, and Pathogenesis
Study Guide - Smart Notes

Viral Structure and Classification
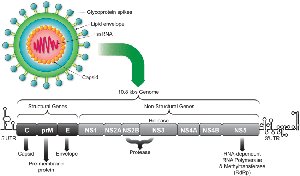
Capsid, Nucleocapsid, and Viral Envelopes
The capsid is a protein shell that protects the viral nucleic acid genome from degradation by nucleases. The nucleocapsid consists of the capsid and the genome. Viruses can be classified as naked viruses (lacking an envelope) or enveloped viruses (possessing a lipid membrane derived from the host cell).
Capsid protein: Encases and protects the viral genome.
Envelope: Lipid bilayer containing viral glycoproteins, present in enveloped viruses.
Matrix protein: Links the envelope and nucleocapsid in enveloped viruses.
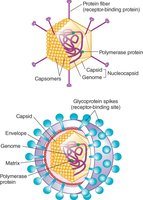
Additional info: The envelope often contains glycoprotein spikes that mediate attachment to host cells.
Viral Genes and Dependency on Host
Genes Required for Infectious Virus Assembly
Viruses encode genes for capsid proteins, receptor-binding proteins, and enzymes necessary for genome replication. Genome length and composition vary widely (e.g., ssRNA, dsRNA, large DNA viruses known as giruses). Viruses are obligate intracellular parasites, relying on host cell machinery for replication.
Capsid protein genes: Form the protective shell.
Receptor-binding protein genes: Mediate host cell attachment.
Polymerase genes: Encode enzymes for genome replication (e.g., RNA-dependent RNA polymerase).
Viral Replication Cycle
Overview of Viral Replication
Viruses hijack host cells by taking over their genetic machinery and resources in a process called viral replication. This process transforms the host cell into a factory for producing new viruses.
Attachment
Viral surface proteins bind to specific receptor molecules on the host cell membrane, determining host range and tissue specificity. For example, HIV binds to CD4 receptors on T-helper cells, while rhinoviruses bind to ICAM-1 on respiratory cells.
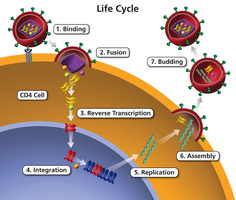
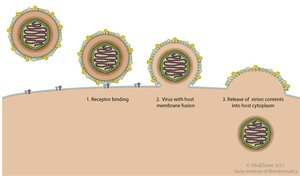
Entry (Penetration)
Membrane Fusion: Enveloped viruses fuse their membrane with the host cell membrane, releasing the viral core into the cytoplasm.
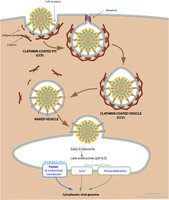
Endocytosis: Viruses are engulfed by the host cell, forming a vesicle that transports the virus inside.
Genetic Injection: Bacteriophages inject their genetic material into the host, leaving the capsid outside.
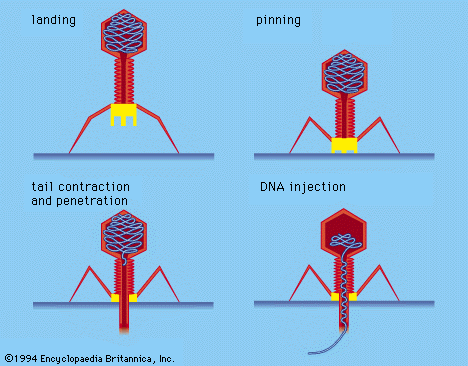
Uncoating
After entry, the viral capsid is removed, releasing the viral genome into the host cell. This process is often triggered by the acidic environment of endosomes.
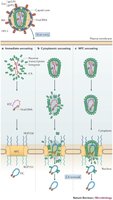
Replication and Biosynthesis
The viral genome directs the host cell to synthesize viral proteins and replicate the viral genome. DNA viruses typically replicate in the nucleus, while most RNA viruses replicate in the cytoplasm.
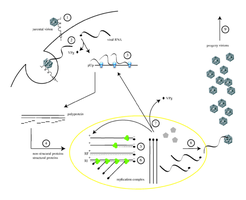
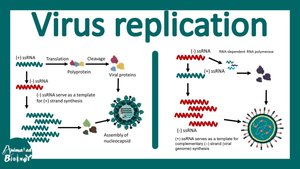
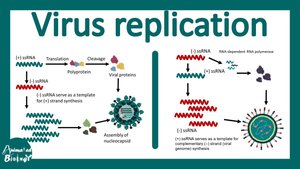
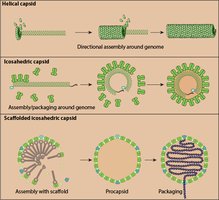
Assembly (Maturation)
New viral proteins and genomes are assembled into immature virus particles, which mature into infectious virions.
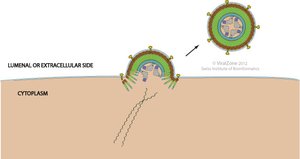
Release (Egress)
Lysis: The host cell bursts, releasing new viruses and killing the cell.
Budding: Enveloped viruses acquire their membrane by budding from the host cell, allowing continued virus production.
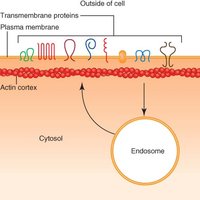
Molecular Hurdles of the Host Cell
Barriers to Viral Infection
Eukaryotic cells present several barriers to viral infection:
Receptors and Polymerases: Host RNA polymerases cannot replicate viral RNA genomes; viruses encode their own RNA-dependent RNA polymerases.
Actin Remodeling: Host cell actin dynamics affect all stages of viral replication.
Ribosome and mRNA Compatibility: Eukaryotic ribosomes translate monocistronic mRNAs; viruses use alternative splicing, segmented genomes, or polyproteins to overcome this.
mRNA Competition: Viruses evolve mechanisms to outcompete host mRNA for translation.
Genetic Diversity in RNA Viruses
RNA viruses mutate rapidly due to the lack of proofreading by RNA-dependent RNA polymerases, leading to high genetic diversity and adaptability.
Targets for Antiviral Therapies
Effective antiviral drugs target essential viral processes without harming the host. Examples include inhibitors of viral entry, genome replication, or assembly.
Mechanisms of Viral Entry and Spread in the Body
Preferred Routes of Entry
Viruses enter the body through:
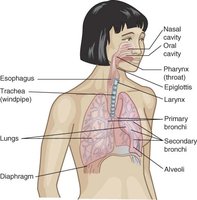
Respiratory tract (most common route)
Gastrointestinal tract (via fecal-oral transmission)
Genital tract (sexually transmitted infections)
Conjunctiva (eye surface)
Placenta (vertical transmission)
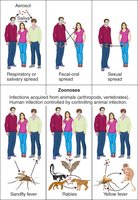
Respiratory Tract Entry
Viruses such as rhinoviruses, coronaviruses, and influenza viruses enter via inhalation of aerosolized droplets.
Gastrointestinal Tract Entry
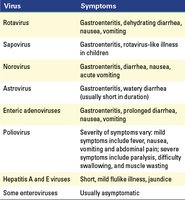
Viruses must resist acidic stomach conditions to infect via ingestion (e.g., norovirus, rotavirus).
Genital Tract Entry
Viruses enter through abrasions or infect reproductive tract cells (e.g., HIV, herpes simplex virus).
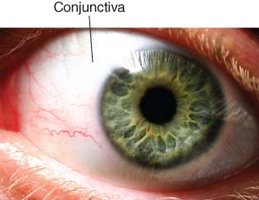
Conjunctiva Entry
The conjunctiva is a thin tissue covering the eye; viruses can cause conjunctivitis or more severe ocular diseases.
Other Routes of Entry
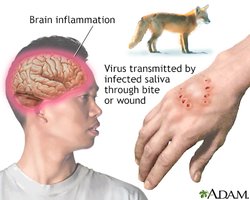
Skin: Requires breaches for viral entry (e.g., cuts, bites).
Transplants and transfusions: Viruses can be introduced via medical procedures.
Mechanisms of Viral Spread and Pathogenesis
Transmission Routes
Respiratory droplets (coughing, sneezing)
Fecal-oral route (contaminated food/water)
Skin breaches (cuts, bites, needles)
Sexual contact (genital tract)
Vertical transmission (mother to child)
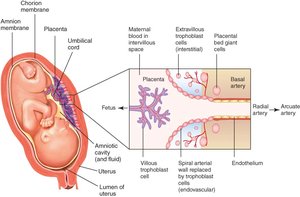
Viral Infections and Pregnancy
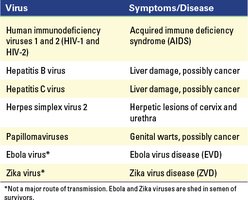
Viruses can cross the placenta, causing spontaneous abortion, stillbirth, neonatal death, or congenital malformations (teratogenic effects). Zika virus is a notable example.
Spread Within the Host
Local spread: Virus spreads to neighboring cells at the site of entry.
Systemic spread: Virus disseminates via the bloodstream (hematogenous) or nerves (neurotropic).
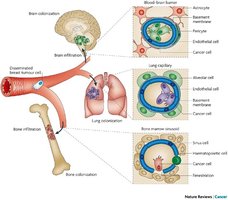
Virus Exit: Shedding
Viruses are shed from their primary sites of replication, often through the same routes as entry. Zoonotic viruses can be shed from animals to humans, with segmented genome viruses (e.g., hantavirus, influenza) being notable emerging pathogens.
Survival of Viruses in the Environment
Virus survival is influenced by composition, presence in waste, temperature, humidity, and pH. Enteric viruses are stable in water and can contaminate food and water supplies.
Human Viruses in Water Environments
Exposure occurs via undercooked shellfish, crops irrigated with wastewater, or contaminated recreational/drinking water. Viruses are more stable in water than bacteria.
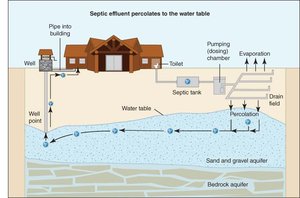
Groundwater Safety
Contaminated groundwater can transmit viruses, as seen in outbreaks linked to well water.
Patterns of Viral Disease
Viral infections can follow four patterns:
Acute infection
Acute infection followed by persistent latent infection
Chronic infection with continuous shedding
Slow infection
