 Back
BackChapter 6 Viruses and Prions: Structure, Replication, and Clinical Relevance
Study Guide - Smart Notes

Viruses, Viroids, & Prions
Introduction to Viruses
Viruses are submicroscopic, acellular infectious agents that require a host cell for replication. They are considered nonliving because they lack cellular structure and metabolism. Virology is the study of viruses, which can infect all forms of life, including bacteria (bacteriophages), animals, and plants.
Size: Typically 20–400 nm, much smaller than prokaryotic and eukaryotic cells.
Obligate intracellular pathogens: Cannot reproduce outside a host cell.
Host range: Viruses can infect every branch in the tree of life.

Comparison of Viruses, Prokaryotes, and Eukaryotes
Viruses differ fundamentally from prokaryotic and eukaryotic cells in structure, replication, and metabolism.
Characteristic | Viruses | Prokaryotes | Eukaryotes |
|---|---|---|---|
Cells? | No | Yes | Yes |
Considered alive? | No | Yes | Yes |
Relative size | Smaller than prokaryotes | Bigger than viruses, smaller than eukaryotes | Bigger than prokaryotes and viruses |
Filterable | Yes | Rarely | No |
Structure | Capsid and nucleic acid | Cells without nuclei | Cells with nuclei |
Replication | Hijack host machinery | Binary fission | Mitosis/Meiosis |
Metabolism | No | Yes | Yes |
Genome | DNA or RNA | DNA | DNA |
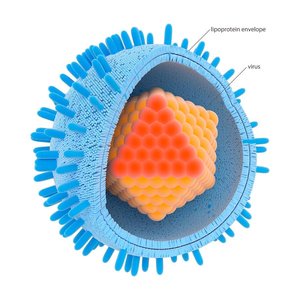
Virion Structure
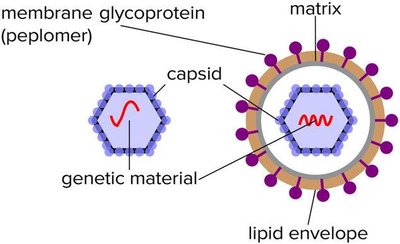
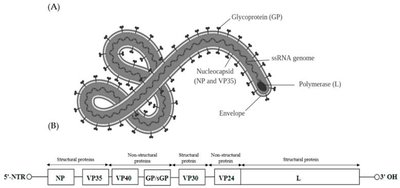
A virion is a single, infectious virus particle. It consists of a protective protein shell called a capsid, which encloses the viral genome (DNA or RNA). Some viruses also possess an outer lipid envelope derived from the host cell membrane.
Capsid: Made of protein subunits called capsomeres.
Envelope: Present in enveloped viruses; absent in naked viruses.
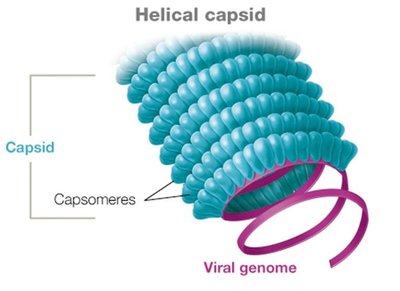
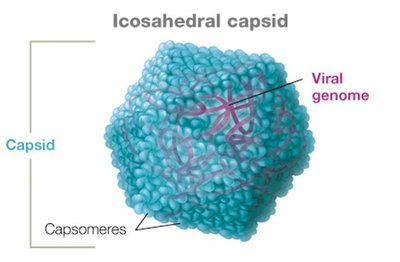
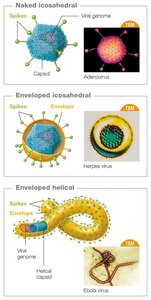
Capsid Symmetry and Types
Animal viruses typically have either helical or icosahedral capsids, while some (especially bacteriophages) have complex structures.
Helical capsid: Hollow tube-like structure.
Icosahedral capsid: Three-dimensional polygonal shape.
Complex capsid: Found in some viruses like bacteriophages and poxviruses.
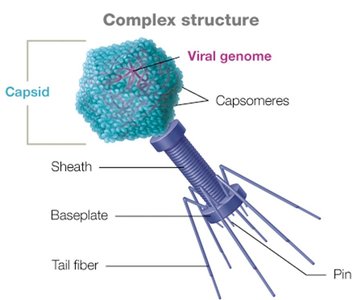
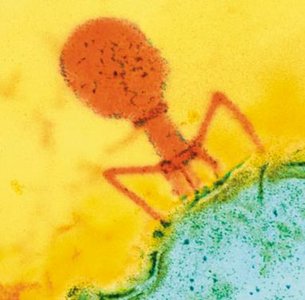
Bacteriophage Structure
Bacteriophages have complex capsids with icosahedral heads and additional structures (tail fibers, baseplate, sheath) for injecting their genome into bacterial cells.
Viral Envelopes and Spikes
Enveloped viruses acquire a lipid membrane from the host cell during budding, while naked viruses lack this envelope and are released by cell lysis. Many viruses have surface glycoproteins called spikes (peplomers) that mediate attachment to host cells.
Enveloped viruses: e.g., coronaviruses, herpesviruses, influenza.
Naked viruses: e.g., poliovirus, human papillomavirus.
Bacteriophages: Always naked.
Viral Genomes
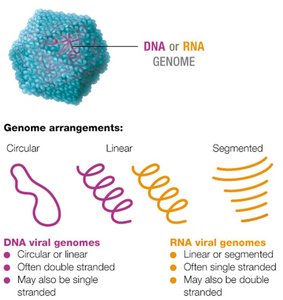
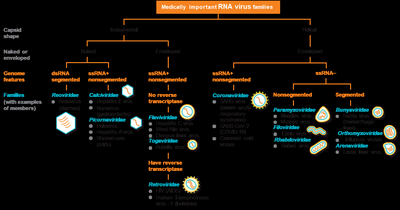
Viral genomes are highly variable and can be composed of DNA or RNA, which may be single- or double-stranded, linear, circular, or segmented. Most viruses have fewer than 300 genes, encoding structural proteins, enzymes for replication, and other factors.
Genome arrangements: DNA (circular/linear), RNA (linear/segmented).
Viral Genome Replication and Expression
The main goal of all viruses is to hijack the host cell machinery to produce viral proteins and assemble new virions. The process of making mRNA from viral genomes varies depending on the type of nucleic acid present.
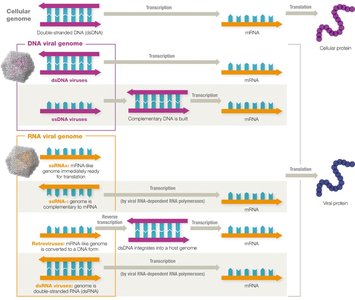
Viral Genome Evolution
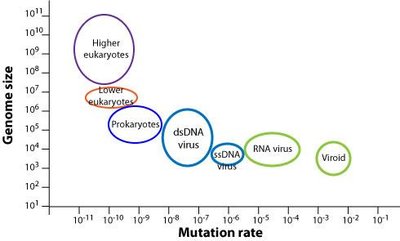
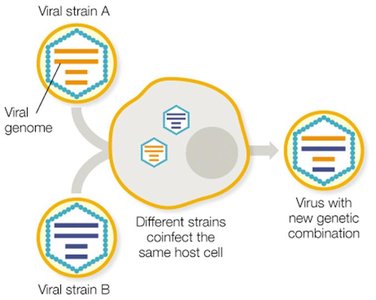
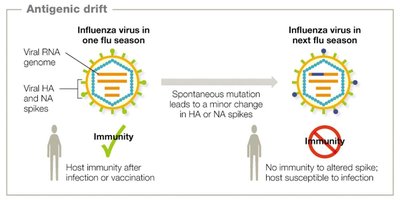
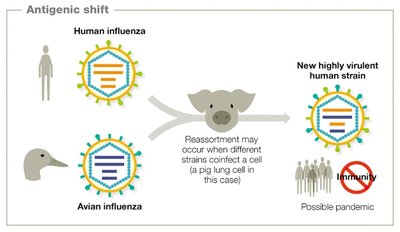
Viruses mutate rapidly due to their short replication cycles and lack of proofreading in RNA polymerases. This leads to high genetic variability, which can result in antigenic drift and shift, especially in RNA viruses like influenza.
Antigenic drift: Minor changes in viral antigens due to point mutations.
Antigenic shift: Major genetic reassortment, often leading to pandemics.
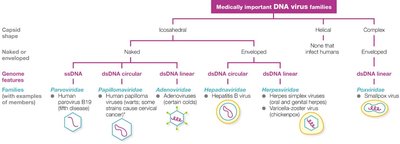
Classification and Taxonomy of Viruses
Viruses are classified based on their nucleic acid type, capsid symmetry, presence or absence of an envelope, and genome architecture. The highest taxonomic rank is phylum, followed by order, family, subfamily, genus, and species.
Taxon | Example | Naming Convention |
|---|---|---|
Order | Herpesvirales | Italicized, ends in 'virales' |
Family | Herpesviridae | Italicized, ends in 'viridae' |
Subfamily | Alphaherpesvirinae | Italicized, ends in 'virinae' |
Genus | Simplexvirus | Italicized, ends in 'virus' |
Species | Human herpesvirus-1 | Italicized, proper nouns capitalized |
Host Range and Tropism
Host range refers to the spectrum of species a virus can infect, while tropism describes the specific tissues or cell types targeted by a virus. Some viruses have a broad host range (e.g., Ebola), while others are highly specific (e.g., measles virus infects only humans).
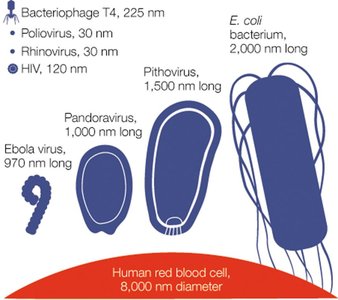
Virus Sizes
Viruses vary greatly in size, but are generally smaller than bacteria. For example, rhinoviruses are about 30 nm, while pandoraviruses can reach up to 1,500 nm.
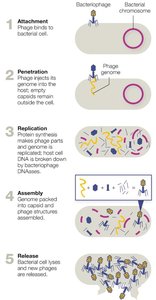
Viral Replication Cycles
Viruses replicate by hijacking host cell machinery. Bacteriophages typically follow either a lytic or lysogenic cycle, while animal viruses have more varied replication strategies.
Lytic cycle: Virus replicates and lyses the host cell.
Lysogenic cycle: Viral genome integrates into host DNA as a prophage.
Animal Virus Replication
Animal viruses replicate through a series of steps: attachment, penetration, uncoating, replication, assembly, and release. Enveloped viruses are released by budding, while naked viruses typically cause cell lysis.
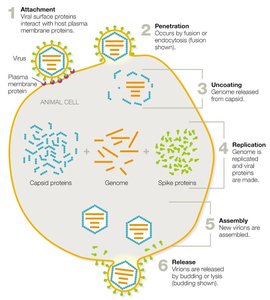

Persistent Viral Infections
Some animal viruses establish persistent infections, which can be chronic (continuous release of virions, e.g., HIV) or latent (periods of dormancy with occasional flare-ups, e.g., herpesviruses).
Chronic infection: Slow disease progression, continuous virion release.
Latent infection: Intermittent symptoms, viral genome persists in host cells.
Oncogenic Viruses
Oncogenic viruses can cause cancer by promoting uncontrolled cell division or inhibiting cell death. Examples include human papillomavirus (HPV), Epstein–Barr virus, and hepatitis B and C viruses.
Virus | Genome | Integrates? | Cancer Link | Mechanism |
|---|---|---|---|---|
HPV | DNA | Yes | Cervical, oropharyngeal, anal cancers | Uncontrolled cell division |
HTLV | RNA | Yes | Adult T-cell leukemia | Uncontrolled cell division |
Hepatitis B/C | DNA/RNA | No | Liver cancer | Chronic inflammation, DNA damage |
Virus Cultivation and Detection
Viruses are cultivated using bacterial cultures (for bacteriophages), embryonated eggs, tissue culture, or live animals. Detection methods include plaque assays, latex agglutination, ELISA, and nucleic acid amplification (PCR).
Plaque assay: Measures viral titer by counting clear zones (plaques) on a bacterial lawn.
ELISA: Detects viral antigens or antibodies via color change.
PCR: Amplifies viral genetic material for sensitive detection.
Antiviral Drugs and Vaccines
Antiviral drugs target various stages of the viral life cycle, including attachment, penetration, replication, and release. Vaccines are crucial for preventing viral diseases by training the immune system to recognize viral antigens.
Nucleoside analogs: Inhibit viral genome replication (e.g., acyclovir, ribavirin).
Reverse transcriptase inhibitors: Block retroviral replication (e.g., AZT).
Interferons: Enhance host antiviral defenses.
Entry inhibitors: Block viral attachment or fusion (e.g., docosanol, palivizumab).
Prions
Prions are infectious proteins that lack nucleic acids. They cause transmissible spongiform encephalopathies (TSEs), such as Creutzfeldt-Jakob disease, by inducing misfolding of normal prion proteins in the brain, leading to neurodegeneration.
Prion diseases: Characterized by sponge-like holes in brain tissue.
Diagnosis: Based on detection of spongiform changes in brain tissue.
Prion-like Mechanisms in Neurodegenerative Diseases
Some neurodegenerative diseases, such as Alzheimer's, Parkinson's, and ALS, exhibit prion-like mechanisms, where misfolded proteins propagate by inducing misfolding in normal proteins, leading to disease progression.
Clinical Case Study: Virus Detection and Classification
A clinical scenario involving blood transfusion and subsequent liver cancer highlights the importance of virus detection, classification, and understanding of viral oncogenesis. Key points include the use of molecular diagnostics, the role of viral genome analysis, and the interpretation of immune responses in vaccinated versus unvaccinated subjects.
Screening for new viruses: Use nucleic acid amplification and serological tests.
Classification: Based on genome type, structure, and replication strategy.
Oncogenic potential: Not ruled out by absence of cancer in most cases; requires further study.
