 Back
BackViruses, Prions, and Fundamentals of Microbial Growth: Structured Study Notes
Study Guide - Smart Notes

Chapter 6: Viruses and Prions
General Virus Characteristics
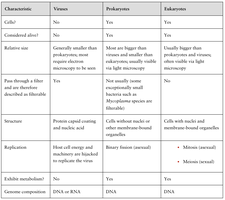
Viruses are nonliving, noncellular entities that infect animal, plant, or bacterial cells. They are especially small and are obligate intracellular pathogens, meaning they must invade living cells to replicate. The study of viruses is called virology. Viruses are classified based on their structural and genomic features, which dictate their host range and infection progression.
Bacteriophages (phages): Viruses that infect bacteria.
Animal Viruses: Viruses that infect humans and other animals.
Virion: A single, infectious virus particle with a protective protein capsid and genetic material (DNA or RNA).
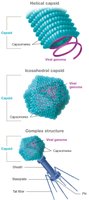
Viral Capsids
The capsid is the protein shell that protects the viral genome and gives shape to the virus. Capsids are made of protein subunits called capsomeres and can have helical, icosahedral, or complex symmetry.
Helical: Hollow tube shape, common in plant viruses.
Icosahedral: Polygonal shape, strong and space-efficient, common in animal viruses.
Complex Structure: Elaborate structures with multiple components, typical of bacteriophages.
Capsid assembly is a potential target for antiviral drugs.
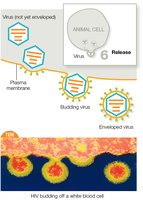
Viral Envelopes
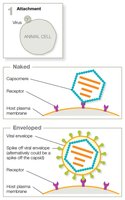
Some animal viruses have a lipid-based envelope surrounding the capsid. Enveloped viruses acquire their envelope by budding from the host cell, while naked viruses lyse out of the host cell without an envelope.
Enveloped viruses: e.g., coronaviruses, influenza viruses, herpes viruses.
Naked viruses: e.g., human papilloma viruses, poliovirus, rotavirus.
Viral Spikes (Peplomers)
Many viruses have glycoprotein extensions called spikes that help them attach and gain entry to host cells. Spikes determine host and tissue specificity and are targets for immune responses and vaccines.
Influenza viruses have two types of spikes: Hemagglutinin (HA) for attachment and entry, and Neuraminidase (NA) for release and spread.
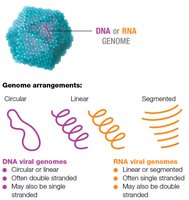
Viral Genomes
Viral genomes can be DNA or RNA, single or double stranded, and arranged as segmented, circular, or linear molecules. Viruses require a host cell to produce proteins and replicate.
dsDNA viruses: Transcribe DNA to mRNA, then translate to protein.
ssDNA viruses: Convert to dsDNA before transcription.
ssRNA+ viruses: RNA genome acts as mRNA, translated directly.
ssRNA- viruses: RNA genome must be transcribed to mRNA by viral RNA-dependent RNA polymerase (RdRP).
Retroviruses: Reverse transcription of RNA to DNA, which integrates into host genome.
dsRNA viruses: Require viral RdRP to transcribe RNA to mRNA.

Viral Genome Evolution
Viruses evolve rapidly due to fast replication and frequent mutations, especially in RNA viruses. Reassortment events occur when two viral strains coinfect a host cell, mixing genomes and generating new strains.
Antigenic drift: Small, gradual genetic changes in viral genes, leading to seasonal flu outbreaks.
Antigenic shift: Major genetic reassortment, leading to new viral strains and pandemics.
Classifying and Naming Viruses
Viruses are classified by nucleic acid type, capsid symmetry, envelope presence, and genome architecture. The highest taxon is phylum, followed by order, family, genus, and species. 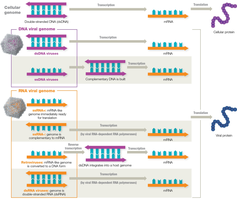
Host Range and Tropism
Viruses exhibit specificity for host species and tissues, determined by surface factors and spikes.
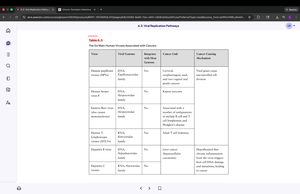
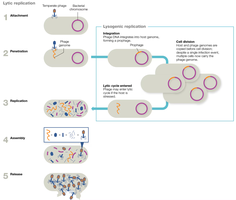
Viral Replication Pathways
Viruses hijack host cell machinery to build new virions. Bacteriophages replicate via lytic or lysogenic pathways.
Lytic Replication: Attachment, penetration, replication, assembly, release.
Lysogenic Replication: Integration of viral DNA as a prophage, silent replication, excision under stress, switch to lytic cycle.
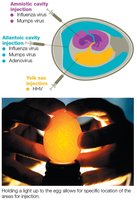
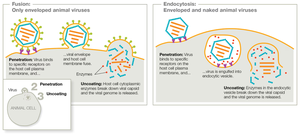
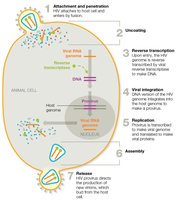
Animal Virus Replication
Animal viruses follow six main steps: attachment, entry, uncoating, replication, assembly, and release. Enveloped viruses bud off, while naked viruses lyse the cell. 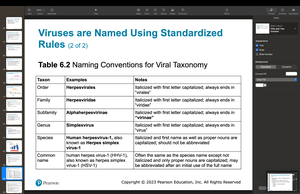
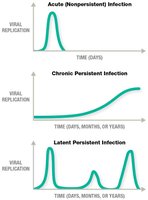
Persistent Infections
Some animal viruses cause persistent infections, which can be chronic, latent, or slow. Chronic infections involve continuous or intermittent replication, while latent infections involve periods of dormancy and flare-ups.
Mechanisms: Immune evasion, slow replication, infection of immune-privileged sites, integration into host genome.
Provirus: Viral genome integrated into host DNA.
Oncogenic Viruses
Viruses that cause persistent infections may be oncogenic, stimulating uncontrolled cell division and decreasing responsiveness to death signals. Examples include human papilloma viruses (HPVs) and human T-lymphotropic viruses (HTLV). 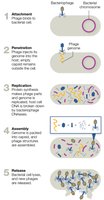
Clinical Aspects of Viruses
Bacteriophages are grown using bacterial hosts and media; lytic phages create plaques.
Animal viruses are cultured using tissue culture, live animal hosts, or embryonated eggs.
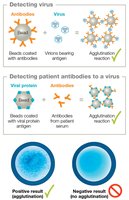
Diagnostic Tests for Viruses
Detection methods include searching for viral proteins (antigens), patient antibodies, or viral genetic material. Agglutination tests and ELISAs are common.
Agglutination: Rapid, inexpensive, but less sensitive and specific.
ELISA: Highly sensitive and specific, quantitative, but requires lab equipment.
PCR and RT-PCR: Detect viral nucleic acids, highly sensitive.
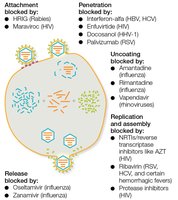
Antiviral Drugs
Antiviral drugs target viral attachment, penetration, uncoating, replication, assembly, and release. Most are nucleoside analogs or enzyme inhibitors.
Attachment/penetration blockers: e.g., maraviroc, enfuvirtide.
Replication blockers: e.g., acyclovir, ribavirin, remdesivir, AZT.
Release blockers: e.g., oseltamivir, zanamivir.
Prions
Prions are infectious proteins that cause transmissible spongiform encephalopathies, such as Creutzfeldt-Jakob disease. Prions do not contain genetic material and cause disease by inducing misfolding of normal proteins, leading to brain tissue degeneration.
Inherited or acquired forms exist.
No cures or vaccines are available.
Some neurodegenerative diseases (e.g., Alzheimer's, Parkinson's) exhibit prion-like features.
Chapter 7: Fundamentals of Microbial Growth and Decontamination
Microbial Growth and Biofilms
Microbial growth is cell division that increases population size. Biofilms are communities of microbes that adhere to surfaces and are highly resistant to treatment.
Budding and spore formation are methods of reproduction.
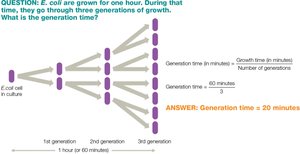
Generation time varies by species and environmental conditions.
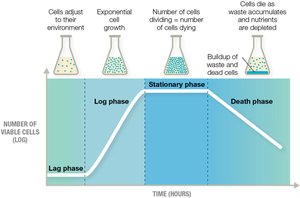
Bacterial Growth Phases
Bacteria in a closed batch system exhibit four growth phases: lag, log, stationary, and death.
Lag phase: Adjustment to environment.
Log phase: Rapid exponential growth.
Stationary phase: Growth rate slows, metabolic products produced.
Death phase: Exponential cell death.

Chemostat
A chemostat maintains constant bacterial growth by continuously adding fresh medium and removing waste. 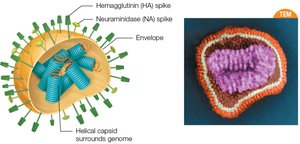
Prokaryotic Growth Requirements
Microbes adapt to temperature, pH, salinity, and oxygen levels.
Temperature: Psychrophiles (-20–10°C), psychrotrophs (0–30°C), mesophiles (10–50°C), thermophiles (40–75°C), extreme thermophiles (65–120°C).
pH: Acidophiles (pH 1–5), neutrophils (pH 5–8), alkaliphiles (pH 9–11).
Salinity: Halophiles tolerate up to 35% salt.
Oxygen: Obligate aerobes, microaerophiles, anaerobes, aerotolerant anaerobes, obligate anaerobes, facultative anaerobes.

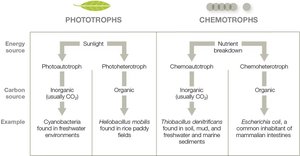
Microbial Nutrients and Energy Sources
Microbes require essential nutrients, growth factors, and energy sources.
Heterotrophs: Require external organic carbon.
Autotrophs: Use carbon fixation to convert inorganic carbon.
Phototrophs: Use light energy.
Chemotrophs: Break down chemical compounds for energy.
Growing, Isolating, and Counting Microbes
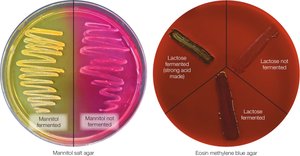
Media are classified by physical state (liquid, solid, semisolid), chemical composition (defined, complex), and function (differential, selective).
Blood agar: Differential for hemolytic bacteria.
Mannitol salt agar (MSA): Selective for Staphylococcus, differentiates mannitol fermenters.
Eosin methylene blue agar (EMB): Selective for gram-negative, differentiates lactose fermenters.
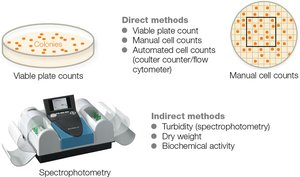
Counting Microbes
Direct methods: Plate counts, manual cell counts, automated cell counts.
Indirect methods: Turbidity, dry weight, biochemical activity.
Microbial Growth Reduction and Decontamination
Decontamination measures include sterilization (eliminates all microbes) and disinfection (reduces microbial numbers). Physical methods include temperature changes, radiation, and filtration.
Autoclaving: Steam heat and pressure for sterilization.
Boiling: Effective for decontamination, not sterilization.
Pasteurization: Moderate heat to eliminate pathogens in liquids.
Dry heat: Incineration or hot-air oven.
Radiation: Ionizing (gamma rays, x-rays), non-ionizing (UV).
Filtration: HEPA filters for air and liquids.
Chemical Controls: Germicides
Germicides are classified as disinfectants (inanimate objects) or antiseptics (living tissue), and ranked as high, intermediate, or low-level agents. Types include alcohols, aldehydes, phenols, halogens, peroxygens, ethylene oxide, and detergents.
Critical equipment requires sterilization.
Semicritical equipment requires high-level disinfection.
Noncritical equipment requires low-level disinfection.
Microbial Control Considerations
Mycobacterium: Requires strong disinfectants due to waxy cell wall.
Endospores: Resistant to many treatments; autoclaving is most effective.
Viruses: Enveloped viruses are easier to destroy than naked viruses.
Protozoa: Some stages resist chlorine; require filtration and UV.
Prions: Require aggressive chemical and heat treatments.
