Back
BackCarbohydrates: Structure, Function, and Health Implications
Study Guide - Smart Notes

Carbohydrate Overview
Definition and Chemical Structure
Carbohydrates are organic compounds composed of carbon, hydrogen, and oxygen, typically with the empirical formula Cn(H2O)n. The term 'carbohydrate' reflects their composition: 'carbo' for carbon and 'hydrate' for water. They are a primary energy source for the body, especially for the brain and red blood cells.
Key Elements: Carbon, hydrogen, and oxygen
General Formula: C6H12O6 for glucose
Essential Nutrients: Carbohydrates are not classified as essential nutrients because the body can synthesize glucose from protein and fat.

Are Carbohydrates Essential?
Although carbohydrates are not strictly essential (since glucose can be synthesized from other macronutrients), they are the preferred energy source for the brain and red blood cells. Diets low in carbohydrates, such as the traditional Inuit diet or popular diets like Atkins and Keto, demonstrate that humans can survive on minimal carbohydrate intake, but adequate carbohydrate consumption supports optimal health and fiber intake.
AMDR for Adults: 225–325 g/day for a 2000 kcal diet
Brain Requirement: ~100 g/day of glucose
Red Blood Cells: Require glucose for energy
Fiber: Adequate intake is linked to numerous health benefits
Classification of Carbohydrates
Simple vs. Complex Carbohydrates
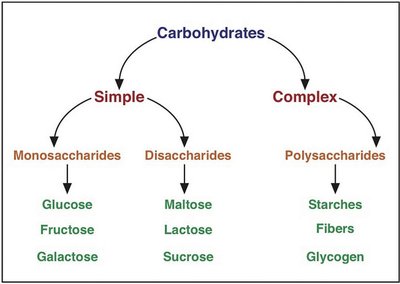
Carbohydrates are classified based on their chemical structure and complexity. Simple carbohydrates include monosaccharides and disaccharides, while complex carbohydrates are polysaccharides.
Simple Carbohydrates: Monosaccharides (glucose, fructose, galactose) and disaccharides (maltose, sucrose, lactose)
Complex Carbohydrates: Polysaccharides (starches, fibers, glycogen)
Monosaccharides
Structure and Types
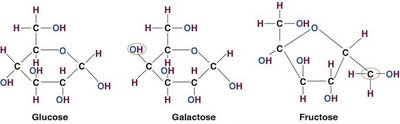
Monosaccharides are the simplest form of carbohydrates, consisting of a single sugar unit. All dietary carbohydrates must be broken down into monosaccharides for absorption in the gastrointestinal tract.
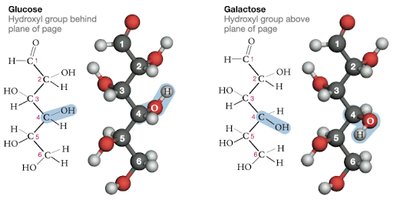
Glucose: The most abundant monosaccharide, primary energy source for the body, especially the brain and red blood cells.
Fructose: The sweetest monosaccharide, found in fruits, honey, and high-fructose corn syrup.
Galactose: Rarely found free in nature; primarily found as part of lactose in dairy products.
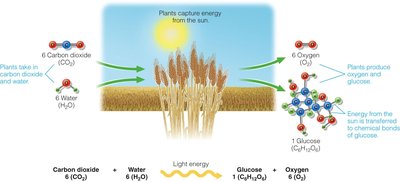
Photosynthesis and Glucose Production
Plants synthesize glucose through photosynthesis, using sunlight to convert carbon dioxide and water into glucose and oxygen.
Photosynthesis Equation:
Potential Energy in Carbohydrates
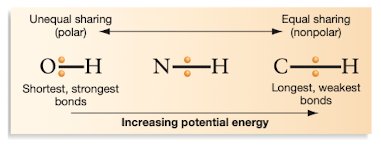
Chemical Bonds and Energy Storage
The energy stored in carbohydrates is found in the chemical bonds between carbon, hydrogen, and oxygen atoms. The potential energy is higher in bonds where electrons are shared equally (nonpolar), such as C-H bonds, compared to bonds where electrons are held tightly by electronegative atoms (polar), such as O-H bonds.
Nonpolar Bonds: Higher potential energy (e.g., C-H)
Polar Bonds: Lower potential energy (e.g., O-H)
Disaccharides
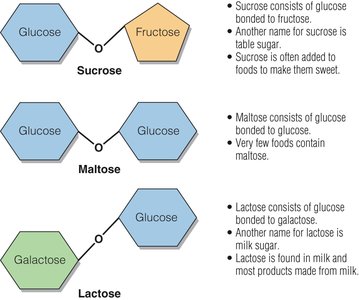
Structure and Types
Disaccharides are composed of two monosaccharide units linked by a glycosidic bond. They must be broken down into monosaccharides for absorption.
Maltose: Glucose + Glucose; found in sprouted grains and produced during fermentation.
Sucrose: Glucose + Fructose; common table sugar, found in fruits, vegetables, and honey.
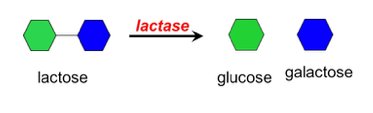
Lactose: Glucose + Galactose; found in milk and dairy products.
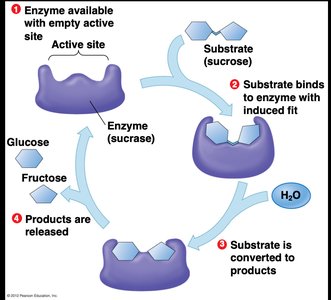
Enzymatic Digestion of Disaccharides
Specific enzymes are required to hydrolyze disaccharides into their monosaccharide components. For example, lactase breaks down lactose into glucose and galactose.
Lactase: Enzyme required for lactose digestion; deficiency leads to lactose intolerance.
Sugar Alcohols and Non-Nutritive Sweeteners
Sugar Alcohols
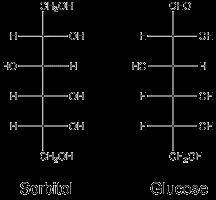
Sugar alcohols are carbohydrates that are only partially absorbed and metabolized, providing fewer calories than sugars. They are used as sweeteners in sugar-free and reduced-calorie foods.
Examples: Sorbitol, Xylitol, Erythritol
Properties: Water-soluble, sweet, low-calorie, can cause a laxative effect, may help prevent tooth decay
Non-Nutritive Sweeteners
Non-nutritive sweeteners provide little or no calories and are much sweeter than sucrose. They are used to reduce caloric intake and manage blood glucose levels.
Examples: Saccharin, Aspartame, Sucralose, Stevia
Safety: FDA-approved sweeteners are considered safe for the general population under specified conditions, though emerging research suggests possible effects on gut microbiome and glucose tolerance.
Scientific Name | Trade Name | kcal/g | Relative Sweetness | Heat-Stable |
|---|---|---|---|---|
Saccharin | Sweet & Low, Sweet Twin, Sweet 'N Low Brown, Necta Sweet | 0 | 200-700 | |
Aspartame | Nutrasweet, Equal, Sugar Twin | 4 | 160-220 | |
Neotame | 0 | 8000 | X | |
Acesulfame-K | Sunett, Sweet & Safe, Sweet One | 0 | 200 | X |
Sucralose | Splenda | 0 | 600 | X |
Tagatose | Nutralose | 1.5 | 0.75-0.92 | |
Stevia | Truvia, PureVia, SweetLeaf | 0 | 150-300 | X |
Added Sugars in the Diet
Sources and Recommendations
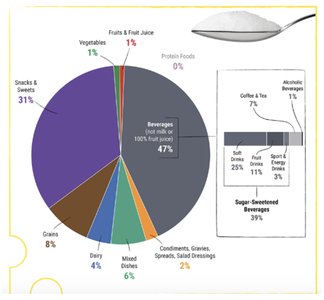
Added sugars are found in a wide variety of processed foods and contribute to excess caloric intake without providing essential nutrients. The American Heart Association recommends limiting added sugars to 25 g/day for women and 38 g/day for men.
Common Sources: Sugar-sweetened beverages, snacks, sweets, processed foods
Health Risks: High intake is associated with increased risk of obesity, type 2 diabetes, cardiovascular disease, and dental cavities
Recognizing Added Sugars
Added sugars appear under many names on ingredient lists, often ending in '-ose' or including terms like syrup, nectar, or juice. Awareness of these terms helps consumers reduce added sugar intake.
Examples: Dextrose, glucose, corn syrup, honey, agave nectar, malt syrup, molasses
Appendix: Carbohydrate Chemistry
Aldose and Ketose Sugars
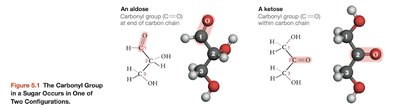
Monosaccharides are classified as aldoses or ketoses based on the position of their carbonyl group. Glucose and galactose are aldoses, while fructose is a ketose.
Aldose: Carbonyl group at the end of the carbon chain
Ketose: Carbonyl group within the carbon chain
Linear and Ring Forms of Sugars
Monosaccharides can exist in both linear and ring forms. In aqueous solutions, the ring form predominates due to its stability.
Glucose: Can cyclize to form α- or β-glucose