 Back
BackAcid-Base Chemistry: Structure, Mechanisms, and Reactivity
Study Guide - Smart Notes

Chapter 3: Acid-Base Chemistry
Brønsted-Lowry Acids and Bases
The Brønsted-Lowry theory defines acids as proton donors and bases as proton acceptors. Acid-base reactions involve the transfer of a proton (H+) from one molecule to another, and these reactions are typically reversible. The concept of conjugate acids and bases is central: when an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
Acid: Proton donor
Base: Proton acceptor (must have a lone pair)
Conjugate acid: Formed when a base gains a proton
Conjugate base: Formed when an acid loses a proton
Amphoteric compounds: Substances like water can act as either acids or bases depending on the reaction context
Example: In the reaction between water and ammonia, water acts as an acid and ammonia as a base. The reverse reaction demonstrates the conjugate acid-base pairs.
The Mechanism of Acid-Base Reactions
Acid-base reaction mechanisms describe how protons are transferred. Mechanisms can be explained verbally, with curved arrows, or with orbital overlap diagrams. Curved arrows are the most common notation, showing the movement of electron pairs during bond formation and breaking.
Curved arrows: Start from a lone pair or bond and point to where electrons appear in the product
Bond breaking: Arrow from a bond to an atom indicates bond breaking and formation of a lone pair
Bond formation: Arrow from a lone pair to an atom indicates bond formation
Multiple arrows: Used when several electron pairs change simultaneously
Example: In the reaction of ammonia and water, a lone pair on nitrogen forms a bond with a proton from water, while the O-H bond breaks, resulting in hydroxide.
Acidity of Organic Molecules: pKa
The acidity of a compound is quantified by its acid dissociation constant (Ka) or more commonly by its pKa value, which is the negative logarithm of Ka:
Strong acids: Completely dissociate in water, have large Ka and negative pKa
Weak acids: Partially dissociate, have smaller Ka and positive pKa
pKa values: Allow comparison of acid strengths; lower pKa means stronger acid
Functional group trends: Similar functional groups have similar pKa values
Example: Acetic acid (pKa = 4.73) is a stronger acid than water (pKa = 15.75).
Acid-Base Equilibria
pKa values are used to predict the favorability of acid-base reactions. The equilibrium lies toward the weaker acid and base. A difference of 10 pKa units generally means the reaction goes to completion.
Equilibrium direction: Favors formation of weaker acid/base
Completion: Large pKa difference indicates nearly complete reaction
Example: The reaction of propoxide and phenol can be predicted using their pKa values.
The Relationship of Conjugate Acids and Bases
The strength of an acid is inversely related to the strength of its conjugate base. Strong acids have weak conjugate bases, and weak acids have strong conjugate bases.
Strong acid: Weak conjugate base
Weak acid: Strong conjugate base
Example: Acetate (conjugate base of acetic acid) is a weak base; ethoxide (conjugate base of ethanol) is a strong base.
Structure Principle: Structure Determines Reactivity
Structural factors influence acid and base strength by affecting the stability of lone pairs. Four major factors are charge state, periodicity and hybridization, resonance, and inductive effects.
Charge state: Anions are less stable and stronger bases than their neutral counterparts
Periodicity: Electronegativity and size affect lone pair stability; more electronegative atoms stabilize lone pairs better
Size: Larger atoms stabilize negative charge better, making their conjugate bases weaker
Hybridization: Orbitals with more s character (sp > sp2 > sp3) stabilize lone pairs better
Example: CH3- is a stronger base than NH2-, HO-, or F- due to lower electronegativity.
Resonance Effects
Resonance stabilizes lone pairs by delocalizing charge, making bases weaker. The degree of stabilization depends on the number and quality of resonance contributors.
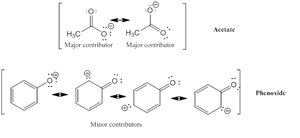
Acetate: Has two major resonance contributors, making it much more stable than ethoxide
Phenoxide: Has several minor resonance contributors, providing less stabilization than acetate
Example: Acetate is about 1012 times more stable than ethoxide due to resonance.
Inductive Effects
Inductive effects stabilize bases by withdrawing electron density through sigma bonds. The effect is additive and decreases with distance from the lone pair.
Electron-withdrawing groups: Increase acidity by stabilizing the conjugate base
Distance: Inductive effects weaken with increasing separation from the lone pair
Example: Trifluoroacetic acid is much more acidic than acetic acid due to the inductive effect of fluorine atoms.
Solvent Effects
Solvents can stabilize or destabilize bases, affecting acid-base reactivity. Solvation is more effective for smaller, less hindered ions.
Solvation: Stabilizes negative charge, making bases weaker
Steric hindrance: Bulky groups reduce solvation, making bases stronger
Example: Methoxide is more stabilized by solvation than t-butoxide, making t-butoxide a stronger base.
Lewis Acids and Bases
The Lewis definition describes acids as electron pair acceptors and bases as electron pair donors. This definition is broader and includes reactions where no proton is transferred.
Lewis acid: Electron pair acceptor
Lewis base: Electron pair donor
Example: In the reaction of hydroxide and BF3, hydroxide donates an electron pair to BF3.
Nucleophiles and Electrophiles
Organic chemists often use the terms nucleophile and electrophile to describe reactivity. Nucleophiles are electron-rich and donate electrons; electrophiles are electron-deficient and accept electrons.
Nucleophile: Electron-rich, donates electrons (Lewis base)
Electrophile: Electron-deficient, accepts electrons (Lewis acid)
Example: Hydroxide acts as a nucleophile in reactions with chloromethane or aldehydes.
Table: pKa Values of Common Functional Groups
pKa values are used to compare the acidity of different functional groups. The table below summarizes typical pKa values for common organic functional groups:
Functional Group | Typical pKa |
|---|---|
Carboxylic acid | ~5 |
Phenol | ~10 |
Alcohol | 16-18 |
Alkyne (sp C-H) | ~25 |
Amine | ~35 |
Alkane (sp3 C-H) | ~50 |
Additional info: Only the proton directly attached to the functional group is considered for pKa. |
