 Back
BackAdvanced Organic Synthesis: Semibullvalene and Estrone Pathways
Study Guide - Smart Notes

Semibullvalene Synthesis and Reactivity
Electrocyclic and Pericyclic Reactions in Semibullvalene Formation
Semibullvalene is a unique organic compound notable for its fluxional behavior and its synthesis via pericyclic reactions. The pathway illustrated involves several key organic transformations, including electrocyclic reactions, Diels-Alder cycloadditions, and photochemical processes.
Electrocyclic Reaction: The initial step involves an electrocyclic ring closure, converting a linear precursor into a cyclic intermediate.
Diels-Alder Reaction: This cycloaddition forms a bicyclic structure, a common method for constructing complex ring systems in organic chemistry.
Photochemical [2+2] Reaction: Exposure to light induces a [2+2] cycloaddition, further increasing molecular complexity.
Fragmentation and Rearrangement: The pathway includes fragmentation (Aza fragmentation) and rearrangement steps, leading to the formation of semibullvalene.
Oxidation and Hydrolysis: Subsequent steps involve oxidation (with O2) and hydrolysis, which modify the functional groups and ring structure.
Cyclopropanation: The final transformation involves cyclopropanation, a reaction that introduces a three-membered ring, often catalyzed by silver ions (Ag+).
Example: The synthesis of semibullvalene demonstrates the application of pericyclic reactions and rearrangements in constructing complex organic molecules.
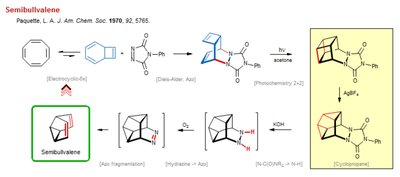
Additional info: Semibullvalene is a classic example in organic chemistry for studying fluxional molecules and pericyclic reaction mechanisms.
Estrone Synthesis (Vollhardt Method)
Complex Steroid Synthesis via Diels-Alder and Silyl Chemistry
Estrone is a steroid hormone, and its synthesis involves advanced organic transformations, including Diels-Alder reactions, silyl group manipulations, and regioselective functionalization. The pathway shown is based on the Vollhardt method, which is notable for its efficiency and selectivity.
Steroid Synthesis: The initial steps involve constructing the steroid framework, often using Diels-Alder cycloadditions to form the fused ring system.
Silyl Group Chemistry: Silyl protecting groups (e.g., Me3Si-) are used to control regioselectivity and protect sensitive functional groups during the synthesis.
Regioselective Functionalization: The pathway achieves high regioselectivity (90% 9:1 regioisomers) in the introduction of functional groups, such as hydroxyl and carbonyl groups.
Deprotection and Oxidation: Final steps involve deprotection (removal of silyl groups) and oxidation to yield estrone.
Example: The synthesis of estrone illustrates the use of Diels-Alder reactions and silyl chemistry in constructing complex natural products.
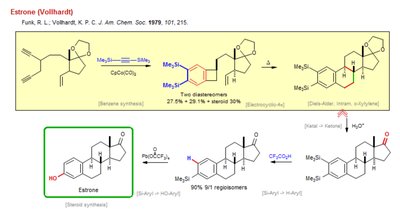
Additional info: Estrone synthesis is a key topic in advanced organic chemistry, demonstrating the principles of regioselectivity, stereochemistry, and protecting group strategies.
