 Back
BackAlcohols, Amines, Ethers, Epoxides, and Thiols: Reactions and Properties
Study Guide - Smart Notes

Alcohols, Amines, Ethers, Epoxides, and Thiols
A. Alcohols
Alcohols are organic compounds containing a hydroxyl group (-OH) attached to a saturated carbon atom. Their reactivity is largely determined by the properties of the hydroxyl group, especially in substitution and oxidation reactions.
Substitution Reactions of Alcohols
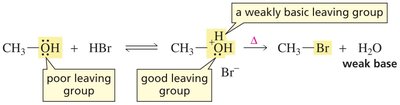
Neutral alcohols do not undergo substitution reactions because the hydroxide ion (OH−) is a poor leaving group due to its strong basicity.
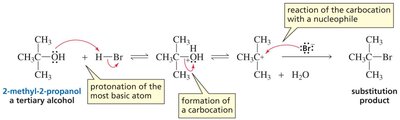
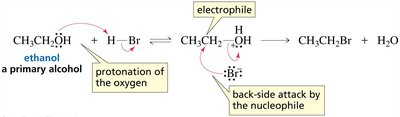
Protonation of the alcohol in acidic conditions converts the OH group into a better leaving group (water), enabling substitution reactions to occur.
The mechanism (SN1 or SN2) depends on the structure of the alcohol:
Tertiary alcohols react via the SN1 mechanism due to the stability of the tertiary carbocation.
Primary alcohols react via the SN2 mechanism because primary carbocations are unstable.
Common halides (Cl−, Br−, I−) are good nucleophiles and facilitate these reactions.
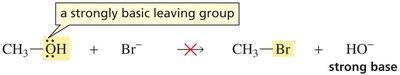
Oxidation of Alcohols
Oxidation involves increasing the number of C–O bonds or decreasing the number of C–H bonds at the alcohol carbon.
Primary alcohols are oxidized to aldehydes, and further to carboxylic acids.
Secondary alcohols are oxidized to ketones, which cannot be further oxidized under normal conditions.
Tertiary alcohols do not undergo oxidation because they lack a hydrogen atom on the alcohol carbon.
Common oxidizing agents include chromic acid (H2CrO4), but milder agents like HOCl in acetic acid can stop oxidation at the aldehyde or ketone stage.
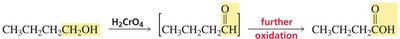
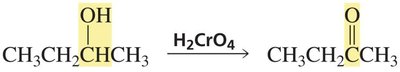
B. Ethers
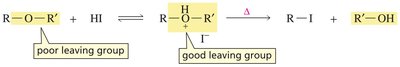
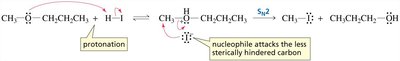
Ethers are compounds with the general formula R–O–R'. Their chemistry is similar to alcohols, but the leaving group (RO−) is also poor, so substitution reactions require acidic conditions.
Substitution Reactions of Ethers
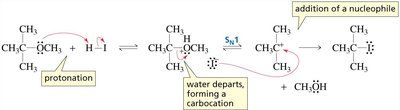
Under acidic conditions, the ether oxygen is protonated, making it a better leaving group.
The mechanism (SN1 or SN2) depends on the ability to form a stable carbocation:
If a stable carbocation can form, the reaction proceeds via SN1.
If not, the reaction proceeds via SN2, with nucleophilic attack at the less hindered carbon.
C. Epoxides
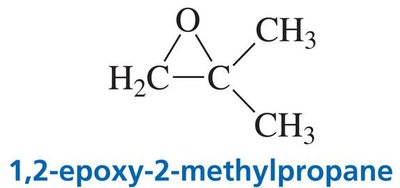
Epoxides are three-membered cyclic ethers, notable for their ring strain and high reactivity. They are typically synthesized by the oxidation of alkenes.
Nomenclature of Epoxides
Named as derivatives of alkenes by adding the suffix "-oxide" (e.g., ethylene oxide).
Alternatively, named as alkanes with the prefix "epoxy-" to indicate the presence of the epoxide ring.

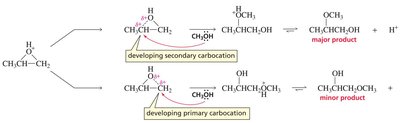
Substitution Reactions of Epoxides
Epoxides are more reactive than ethers due to ring strain.
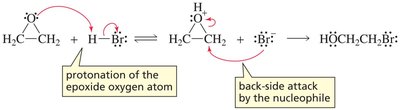
Strong nucleophiles can open the ring directly (SN2-like), while weak nucleophiles require acid-catalyzed ring opening.
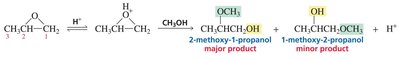
Under acidic conditions, the oxygen is protonated, and nucleophilic attack occurs at the more substituted carbon (carbocation-like transition state).
Under basic or neutral conditions, nucleophilic attack occurs at the less hindered carbon (pure SN2 mechanism).
If the epoxide is asymmetric, attack at different carbons yields different products.
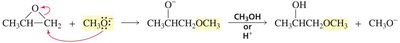
Toxicity and Biological Relevance of Epoxides
Epoxides are highly reactive and can alkylate biological nucleophiles, such as DNA bases (especially guanine at N7), leading to mutations and cancer.
Benzene is metabolized in the body to a carcinogenic epoxide intermediate.
Polyaromatic hydrocarbons (PAHs), formed during high-temperature cooking, are also carcinogenic due to their ability to intercalate and react with DNA.
Some carcinogens, such as aflatoxin, are natural products produced by fungi and can cause severe health effects.
Epoxide-like structures are used in chemotherapy to deliberately damage DNA in cancer cells (e.g., cyclophosphamide forms an aziridine intermediate).

D. Amines
Amines are organic derivatives of ammonia (NH3) and are classified as primary, secondary, or tertiary based on the number of organic groups attached to the nitrogen atom.
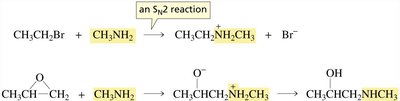
Amines do not undergo substitution or elimination reactions readily, even when protonated, because the leaving group (NH2−) is very poor.
However, amines are moderate nucleophiles and can participate in SN2 reactions, especially with alkyl halides and epoxides.
Additional info: This guide covers the core reactions and properties of alcohols, ethers, epoxides, and amines, focusing on substitution and oxidation mechanisms, as well as the biological implications of epoxide reactivity. Thiols are mentioned in the chapter title but not discussed in detail in the provided material.
