 Back
BackAlcohols, Ethers, Redox, Grignard, and Conjugated Systems: Orgo II Exam 1 Study Guide
Study Guide - Smart Notes

Alcohols: Structure, Properties, and Synthesis
Classification and Physical Properties
Alcohols are organic compounds containing a hydroxyl group (-OH) attached to a carbon atom. Their classification depends on the carbon to which the -OH is attached:
Primary (1º) alcohol: -OH attached to a carbon bonded to one other carbon.
Secondary (2º) alcohol: -OH attached to a carbon bonded to two other carbons.
Tertiary (3º) alcohol: -OH attached to a carbon bonded to three other carbons.
Allylic and benzylic alcohols are special cases where the -OH is attached to a carbon adjacent to a double bond (allylic) or aromatic ring (benzylic), affecting hybridization and reactivity.
Hydrogen bonding: Alcohols form strong hydrogen bonds, resulting in high boiling points and solubility in water.
Phenols: Aromatic alcohols with unique acidity and reactivity.
Synthesis of Alcohols
Alcohols can be synthesized from alkenes via three main methods:
Acid-Catalyzed Hydration: Addition of water across a double bond in the presence of acid, following Markovnikov's rule. Equation:
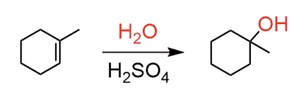
Oxymercuration-Demercuration: Markovnikov addition of water without carbocation rearrangement. Equation:
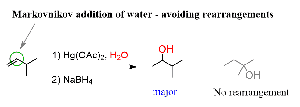
Hydroboration-Oxidation: Anti-Markovnikov addition of water. Equation:
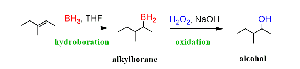
Uses of Alcohols
Alcohols are precursors to alkyl halides via substitution reactions.
Lewis Acid-Catalyzed Chlorination: ZnCl2 converts alcohols to alkyl chlorides.
Thionyl Chloride (SOCl2): Converts alcohols to alkyl chlorides.
Ethers: Structure, Synthesis, and Reactions
Naming and Physical Properties
Ethers are compounds with an oxygen atom connected to two alkyl or aryl groups (R-O-R).
Alkoxy groups: The smaller group is named as an alkoxy substituent.
Epoxides: Cyclic ethers with a three-membered ring.
Crown ethers: Large cyclic ethers capable of binding cations.
Physical properties: Ethers have lower boiling points and solubility than alcohols due to lack of hydrogen bonding.
Synthesis of Ethers
Dehydration of Alcohols: Requires high temperature (140ºC) and acid (H2SO4).
Williamson Ether Synthesis: Reaction of an alkoxide ion with an alkyl halide. Equation:
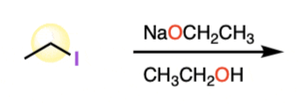
Alkoxymercuration-Demercuration: Similar to alcohol synthesis, but forms ethers.
Epoxide Synthesis: mCPBA (meta-chloroperoxybenzoic acid) oxidizes alkenes to epoxides (syn addition).
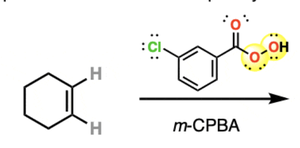
Reactions of Ethers and Epoxides
Ether Cleavage: Ethers can be cleaved by strong acids (e.g., HBr).
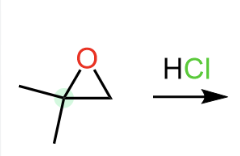
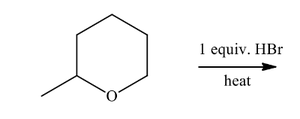
Epoxide Cleavage: Epoxides are highly reactive and can be opened by acids or bases, leading to different regioselectivity.
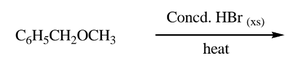
Redox Chemistry: Alcohols and Carbonyls
Oxidation States and Transformations
Organic molecules undergo oxidation and reduction, changing their functional groups and oxidation states.
Oxidation: Loss of electrons, addition of oxygen, or removal of hydrogen.
Reduction: Gain of electrons, addition of hydrogen, or removal of oxygen.
Oxidation state progression: Alkane → Alcohol → Aldehyde → Carboxylic acid.
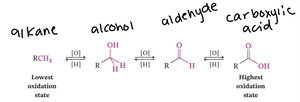
Reduction of Carbonyls
LAH (Lithium Aluminum Hydride): Strong reducing agent, reduces aldehydes, ketones, esters, and carboxylic acids.
NaBH4 (Sodium Borohydride): Milder, reduces aldehydes and ketones but not esters or acids.
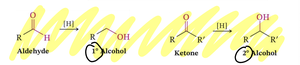
Oxidation of Alcohols
Swern Oxidation: Uses DMSO for mild oxidation.
Chromic Acid/Jones Reagent: Strong oxidizer, visible color change (orange to green).
PCC: Stops at aldehyde or ketone, avoids overoxidation.
KMnO4: Strong oxidizer.
Grignard Reagents: Formation and Applications
Formation of Grignard Reagents
Grignard reagents are organomagnesium compounds formed by reacting alkyl or aryl halides with magnesium in ether.
Equation:
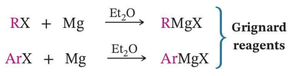
Mechanism and Synthetic Utility
Grignard reagents are strong nucleophiles and bases, used to form carbon-carbon bonds.
Mechanism: Nucleophilic addition to carbonyls, forming alcohols after hydrolysis.
Applications: Synthesis of alcohols, carboxylic acids, and other functional groups.
Protecting Groups in Alcohol Chemistry
Protecting groups are used to temporarily mask alcohols to prevent unwanted reactions.
Types: NaNH2, sulfonates, TBS, silyl ethers, PMB, THP.
Purpose: Selective reactivity, especially in multi-functional molecules.
Conjugated Systems and Diels-Alder Reaction
Resonance and Conjugation
Conjugated systems have alternating double and single bonds, allowing electron delocalization.
Resonance: Movement of electrons (not atoms) increases stability.
Conjugation: More conjugation lowers heat of hydrogenation, indicating greater stability.
Kinetic vs. Thermodynamic Control: Determines product distribution based on reaction conditions.
Diels-Alder Cycloaddition
The Diels-Alder reaction is a [4+2] cycloaddition between a diene and a dienophile, forming a six-membered ring.
Endo vs. Exo: Endo product is preferred in bicyclic systems due to secondary orbital interactions.
Mechanism: Concerted movement of electrons forms new sigma bonds.
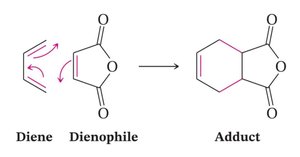
Examples and Applications
The Diels-Alder reaction is widely used in synthetic organic chemistry for constructing complex ring systems. Example: Formation of cyclohexene derivatives from butadiene and ethene derivatives. Additional info: The notes cover key reactions and mechanisms relevant to Ch.11 (Alcohols and Ethers), Ch.12 (Redox), Ch.20 (Amines/Grignard), and Ch.13 (Conjugated Molecules/Diels-Alder) from the organic chemistry curriculum.
