 Back
BackAldehydes and Ketones: Structure, Preparation, and Reactions
Study Guide - Smart Notes

Aldehydes and Ketones
Introduction
Aldehydes and ketones are fundamental classes of organic compounds characterized by the presence of a carbonyl group (C=O). Their chemistry is central to organic synthesis, as they serve as key intermediates in numerous reactions.
Nomenclature of Aldehydes and Ketones
Parent Chain Selection and Naming
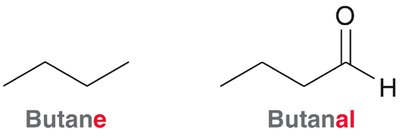
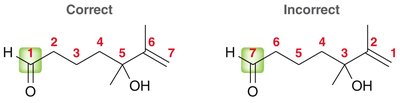
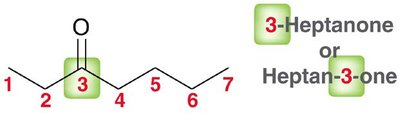
The nomenclature of aldehydes and ketones follows IUPAC rules, with the parent chain always including the carbonyl carbon. Aldehydes are named by replacing the '-e' ending of the parent alkane with '-al', while ketones use '-one'. - Aldehydes: The carbonyl group is always at the end of the chain, and the parent chain must include this carbon. - Ketones: The carbonyl group is within the chain, and its position is indicated by a number. 
Preparation of Aldehydes and Ketones
Methods for Aldehyde Preparation
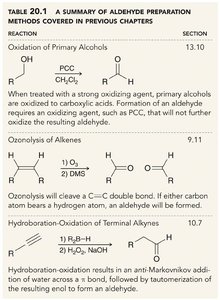
Aldehydes can be synthesized through several methods, including oxidation of primary alcohols, ozonolysis of alkenes, and hydroboration-oxidation of terminal alkynes.
Reaction | Summary |
|---|---|
Oxidation of Primary Alcohols | Primary alcohols are oxidized to aldehydes using mild oxidizing agents like PCC. |
Ozonolysis of Alkenes | Cleavage of C=C bonds forms aldehydes if a hydrogen is present on the carbon. |
Hydroboration-Oxidation of Terminal Alkynes | Anti-Markovnikov addition followed by tautomerization yields aldehydes. |
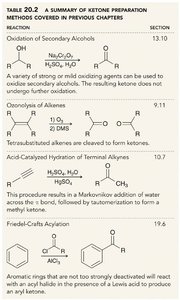
Methods for Ketone Preparation
Ketones are commonly prepared by oxidation of secondary alcohols, ozonolysis of alkenes, acid-catalyzed hydration of terminal alkynes, and Friedel-Crafts acylation.
Reaction | Summary |
|---|---|
Oxidation of Secondary Alcohols | Secondary alcohols are oxidized to ketones using strong or mild oxidizing agents. |
Ozonolysis of Alkenes | Tetrasubstituted alkenes are cleaved to form ketones. |
Acid-Catalyzed Hydration of Terminal Alkynes | Markovnikov addition followed by tautomerization yields methyl ketones. |
Friedel-Crafts Acylation | Aromatic rings react with acyl halides in the presence of Lewis acids to produce aryl ketones. |
Structure and Reactivity of Carbonyl Compounds
Electrophilicity and Inductive Effects
The carbonyl carbon is highly electrophilic due to the polarization of the C=O bond, making it susceptible to nucleophilic attack. - Inductive effect: The oxygen atom withdraws electron density, creating a partial positive charge on the carbonyl carbon. 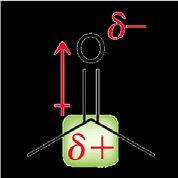
Geometry of Carbonyl Group
The carbonyl carbon is sp2 hybridized, resulting in a trigonal planar geometry. Upon nucleophilic attack, the geometry changes to sp3 (tetrahedral). 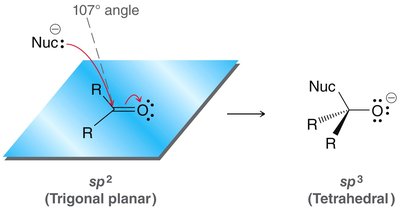
Nucleophilic Addition to Carbonyls
Mechanism with Strong Nucleophiles
Strong nucleophiles attack the carbonyl carbon directly, forming an anionic intermediate, which is then protonated. 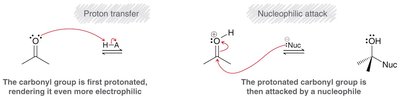
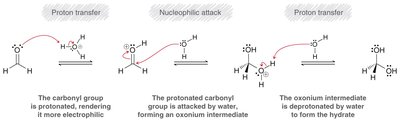
Mechanism with Weak Nucleophiles
Weak nucleophiles require acid catalysis to increase the electrophilicity of the carbonyl group. Protonation precedes nucleophilic attack. 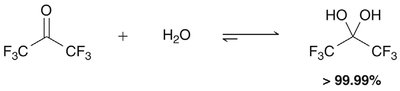
Hydration of Aldehydes and Ketones
Equilibrium and Reactivity
The equilibrium between carbonyl compounds and their hydrates depends on the structure. Electron-withdrawing groups favor hydrate formation. 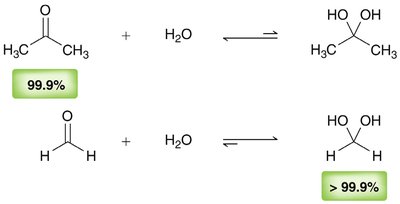
Acid and Base Catalysis
Hydration can be catalyzed by acids or bases, each following a distinct mechanism. - Acid-catalyzed: Protonation of the carbonyl increases electrophilicity. - Base-catalyzed: Hydroxide attacks the carbonyl directly. 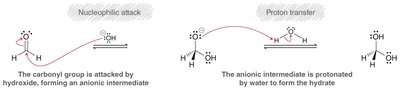
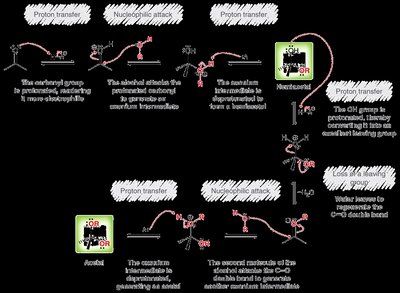
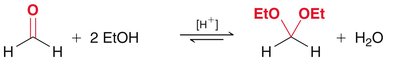
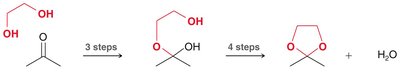
Acetal Formation
Mechanism and Equilibrium
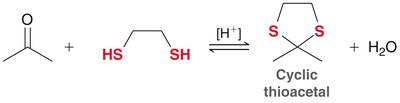
Acetals are formed from aldehydes or ketones and alcohols under acidic conditions. The reaction is reversible and both formation and hydrolysis are acid-catalyzed. - Acetals: Useful as protecting groups for carbonyls in synthesis. - Cyclic acetals: Five- and six-membered rings are favored. 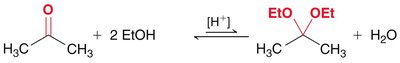
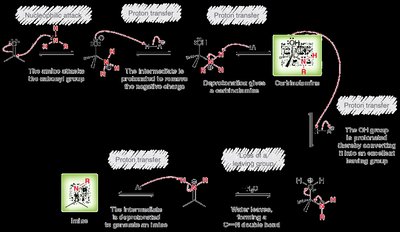
Reactions with Amines: Imines and Enamines
Primary and Secondary Amines
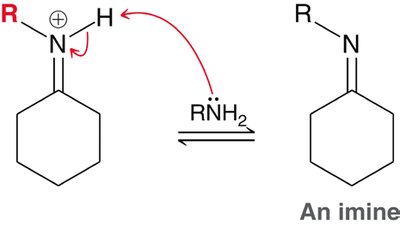
Aldehydes and ketones react with primary amines to form imines, and with secondary amines to form enamines. Acid catalysis is required, with optimal pH around 4-5. - Imines: Formed by direct loss of a proton from nitrogen. - Enamines: Formed by loss of a proton from a neighboring carbon. 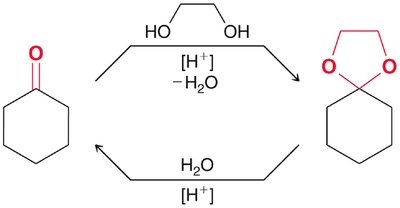
Reduction of Carbonyls: Wolff-Kishner and Alternatives
Wolff-Kishner Reduction
The Wolff-Kishner reduction converts aldehydes and ketones to alkanes via hydrazine and base under heat. 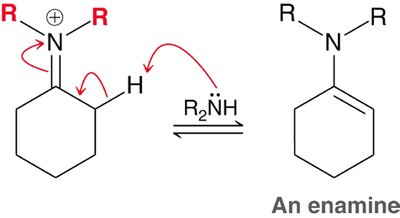
Thioacetal Reduction (Raney Nickel)
Ketones can be converted to alkanes by forming thioacetals and then reducing with Raney nickel. 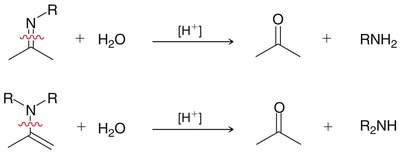
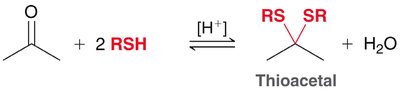
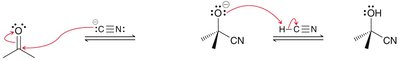
Cyanohydrin Formation
Mechanism and Applications
Cyanide ion acts as a nucleophile, adding to the carbonyl to form cyanohydrins, which are useful intermediates in organic synthesis. 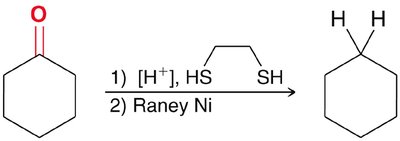
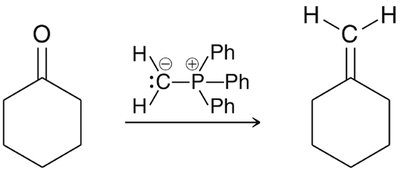
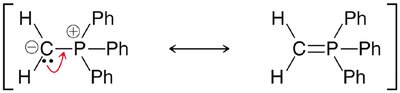
Wittig Reaction
Formation of Alkenes from Carbonyls
The Wittig reaction is a powerful method for converting aldehydes and ketones to alkenes using phosphonium ylides. - Ylide formation: Alkyl halide reacts with triphenylphosphine. - Mechanism: [2+2] cycloaddition forms oxaphosphetane, which fragments to yield alkene and triphenylphosphine oxide. 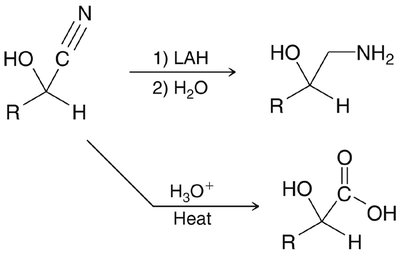
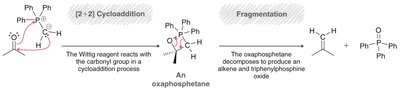
Baeyer-Villiger Oxidation
Insertion of Oxygen into Carbonyls
The Baeyer-Villiger oxidation inserts an oxygen atom between the carbonyl carbon and a neighboring group, converting ketones to esters. - Mechanism: Involves nucleophilic attack by peroxyacid, proton transfer, and rearrangement. 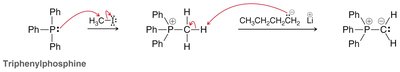
Summary Table: Key Reactions of Aldehydes and Ketones
Reaction | Product | Conditions |
|---|---|---|
Oxidation of Alcohols | Aldehyde/Ketone | PCC, Na2Cr2O7, etc. |
Ozonolysis | Aldehyde/Ketone | O3, DMS |
Hydration | Hydrate | Acid/Base catalysis |
Acetal Formation | Acetal | Alcohol, acid |
Imine/Enamine Formation | Imine/Enamine | Amine, acid |
Wolff-Kishner Reduction | Alkane | Hydrazine, base, heat |
Thioacetal Reduction | Alkane | Thiol, Raney Ni |
Cyanohydrin Formation | Cyanohydrin | HCN |
Wittig Reaction | Alkene | Phosphonium ylide |
Baeyer-Villiger Oxidation | Ester | Peroxyacid |
Conclusion
Aldehydes and ketones are versatile compounds in organic chemistry, with diverse methods of preparation and a wide array of reactions. Their reactivity is largely governed by the electrophilic nature of the carbonyl group, enabling nucleophilic addition, condensation, and oxidation-reduction transformations. Understanding these mechanisms is essential for mastering organic synthesis.
