 Back
BackAlkanes: Structure, Nomenclature, Isomerism, and Conformations
Study Guide - Smart Notes

Alkanes: Structure and Properties
Basic Structure of Alkanes
Alkanes are saturated hydrocarbons consisting only of carbon and hydrogen atoms, connected by single bonds. Their general formula is CnH2n+2 for acyclic (open-chain) alkanes. The geometry around each carbon is tetrahedral, with bond angles close to 109.5°.
Methane (CH4) and Ethane (C2H6) are the simplest alkanes.
Alkanes do not contain π bonds; all bonds are σ bonds.
Alkanes are nonpolar and insoluble in water.
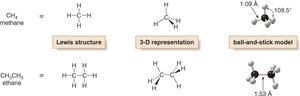
Structural Representations
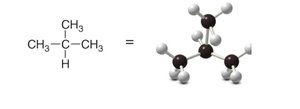
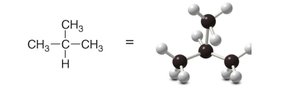
Alkanes can be represented in several ways: Lewis structures, 3-D representations, and ball-and-stick models. These models help visualize the spatial arrangement and bond angles.
Lewis structure: Shows connectivity and all atoms.
3-D representation: Shows geometry and bond angles.
Ball-and-stick model: Physical model emphasizing spatial relationships.
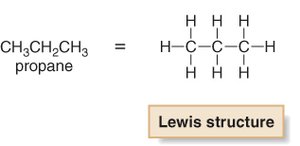
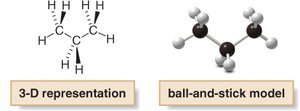
Alkane Series and Molecular Formulas
The alkane series follows a predictable trend in molecular formula and structure:
Methane: CH4
Ethane: C2H6
Propane: C3H8
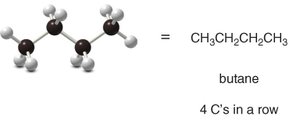
Butane: C4H10
General formula: CnH2n+2
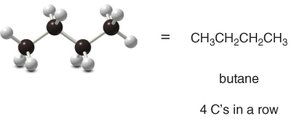
Isomerism in Alkanes
Constitutional Isomers
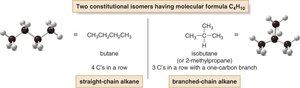
Constitutional isomers are compounds with the same molecular formula but different connectivity of atoms. For example, butane (C4H10) has two isomers: n-butane and isobutane.
n-Butane: Four carbons in a straight chain.
Isobutane: Three carbons in a chain with a one-carbon branch.
Nomenclature of Alkanes
Principles of IUPAC Nomenclature
The IUPAC system provides systematic rules for naming organic molecules. Each name consists of three parts:
Parent name: Number of carbons in the longest continuous chain.
Suffix: Indicates the functional group (for alkanes, the suffix is -ane).
Prefix: Identity, location, and number of substituents.

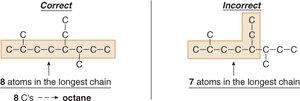
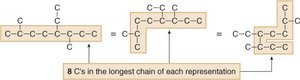
Finding the Longest Chain
To name an alkane, first identify the longest continuous carbon chain. This determines the parent name.
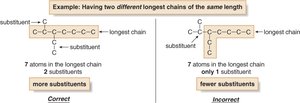
If multiple chains of equal length exist, choose the one with more substituents.
Numbering the Chain
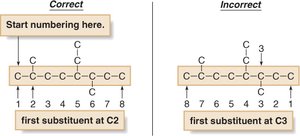
Number the chain to give the first substituent the lowest possible number. In case of ties, give the second substituent the lower number. If still tied, use alphabetization.
Each substituent gets its own number.
Use prefixes (di-, tri-, tetra-) for multiple identical substituents.
Alphabetize substituents, ignoring all prefixes except 'iso'.
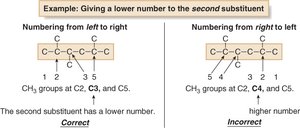
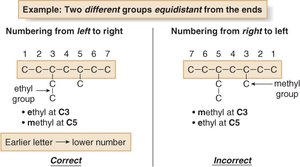
Examples of Nomenclature
2,3,4-trimethylheptane: Indicates three methyl groups at positions 2, 3, and 4 on a seven-carbon chain.
6-ethyl-4,4-dimethylnonane: Indicates an ethyl group at position 6 and two methyl groups at position 4 on a nine-carbon chain.
Cycloalkanes
Cycloalkanes are ring-shaped alkanes with the general formula CnH2n. Naming follows similar rules, but numbering starts at a substituent and proceeds to give the lowest numbers to other substituents.
For rings with one substituent, no number is needed.
For rings with multiple substituents, number to give the lowest set of numbers.
Alphabetize substituents when numbering.
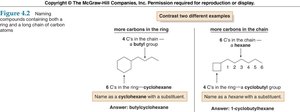
Conformations of Alkanes
Free Rotation and Conformations
Alkanes exhibit free rotation around C–C single bonds, resulting in different conformations. The two most important are the staggered and eclipsed conformations.
Staggered: Hydrogens are as far apart as possible; more stable.
Eclipsed: Hydrogens are aligned; less stable due to torsional strain.
The dihedral angle between hydrogens is 60° in staggered and 0° in eclipsed.

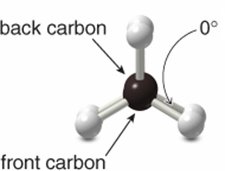
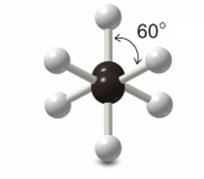
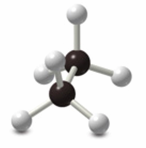
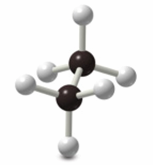
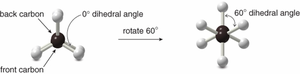
Newman Projections
Newman projections are used to visualize conformations by looking down the C–C bond axis. The front carbon is represented by a circle, and the back carbon by lines radiating from the center.
Staggered conformation: Hydrogens are offset.
Eclipsed conformation: Hydrogens overlap.
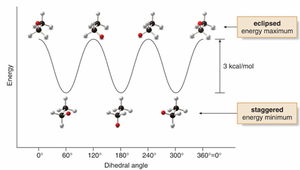
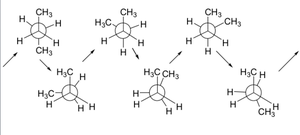
Stability and Energy Differences
The staggered conformation is more stable due to minimized electron repulsion. Each eclipsing interaction increases the energy by about 4 kJ/mol. The energy difference between staggered and eclipsed is about 12 kJ/mol for ethane.
Highest energy: Eclipsed conformation with largest groups eclipsed.
Lowest energy: Staggered, anti conformation.
The energy difference is called the barrier to rotation.
Conformations and Strain in Cycloalkanes
Cyclobutane and Cyclohexane Models
Cycloalkanes can experience strain due to their ring structure:
Torsional strain: Eclipsing of bonds.
Steric strain: Atoms are forced too close together.
Angle strain: Bond angles deviate from the ideal 109.5°.



Cyclohexane Conformations
Cyclohexane adopts a 'chair' conformation to minimize strain. The chair form is the most stable, while the boat form is less stable due to steric and torsional strain.
Chair conformation: Alternating up and down carbons, hydrogens in axial and equatorial positions.
Boat conformation: Less stable, more strain.
Ring-flip: Chair conformations interconvert, swapping axial and equatorial positions.






Physical Properties and Biological Relevance
Physical Properties of Alkanes
Alkanes are insoluble in water and have lower boiling points than polar compounds of similar molecular weight.
Melting and boiling points increase with the number of carbons.
Branching decreases boiling point due to reduced surface area.
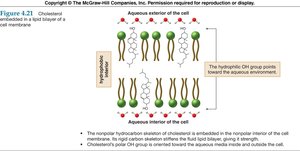
Lipids and Alkanes
Lipids are biological molecules soluble in organic solvents, consisting mainly of carbon and hydrogen. Cholesterol, a steroid, is an example of a polycyclic alkane derivative, important in cell membranes.
Summary Table: Alkane Nomenclature Rules
Rule | Explanation |
|---|---|
Longest Chain | Choose the chain with the most carbons; if tied, choose the one with more substituents. |
Numbering | Number to give the first substituent the lowest number; resolve ties by second substituent or alphabetization. |
Prefixes | Use di-, tri-, tetra- for multiple identical substituents. |
Alphabetization | Alphabetize substituents, ignoring all prefixes except 'iso'. |
Cycloalkanes | Number ring to give lowest set of numbers; start at substituent. |
Additional info: This guide expands on the original notes with academic context, definitions, and examples for clarity and completeness.
