 Back
Backchap 6 handouts Alkyl Halides and Nucleophilic Substitution: Structure, Properties, and Mechanisms
Study Guide - Smart Notes

Alkyl Halides: Structure, Classification, and Nomenclature Chapter 6
Introduction to Alkyl Halides
Alkyl halides (haloalkanes) are organic compounds in which a halogen atom (F, Cl, Br, or I) is bonded to an sp3 hybridized carbon atom. Their structure, classification, and nomenclature are foundational for understanding their reactivity and applications in organic chemistry.
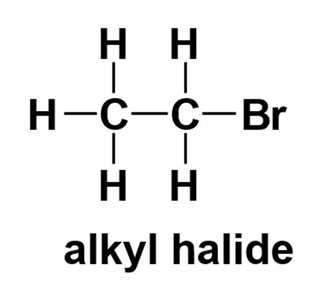
Alkyl halide: Halogen attached to an sp3 carbon.
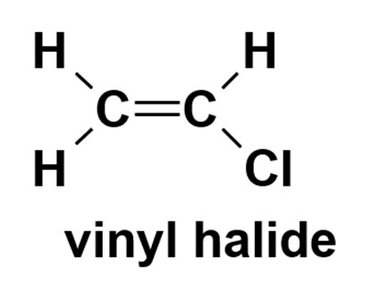
Vinyl halide: Halogen attached to an sp2 carbon of an alkene.
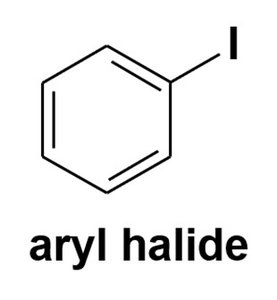
Aryl halide: Halogen attached to an sp2 carbon of a benzene ring.
The chemistry of haloalkanes differs significantly from vinyl and aryl halides due to differences in bond polarization and reactivity.
Classification of Alkyl Halides
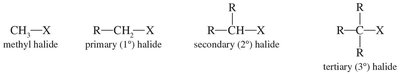
Alkyl halides are classified based on the carbon to which the halogen is attached:
Methyl halide: Halogen attached to a methyl group (CH3X).
Primary (1°) alkyl halide: Halogen attached to a carbon bonded to one other carbon (RCH2X).
Secondary (2°) alkyl halide: Halogen attached to a carbon bonded to two other carbons (R2CHX).
Tertiary (3°) alkyl halide: Halogen attached to a carbon bonded to three other carbons (R3CX).
Geminal dihalide: Two halogens on the same carbon.
Vicinal dihalide: Two halogens on adjacent carbons.
Nomenclature of Alkyl Halides
Alkyl halides are named using IUPAC rules as haloalkanes. The longest carbon chain is chosen as the parent, and the halogen is treated as a substituent. Numbering begins at the end nearest a substituent, and substituents are listed alphabetically.
Prefixes: fluoro-, chloro-, bromo-, iodo-
Common names are also used, especially for simple compounds (e.g., methyl chloride for chloromethane).

Physical Properties and Applications of Alkyl Halides
Physical Properties
The physical properties of alkyl halides, such as boiling points and densities, are influenced by molecular weight, polarity, and branching.
Boiling points increase with the size and polarizability of the halogen: RI > RBr > RCl > RF > RH.
Branching decreases boiling point due to less efficient van der Waals interactions.
Density: Fluoro- and chloroalkanes with one halogen are less dense than water; bromo- and iodoalkanes are denser than water.
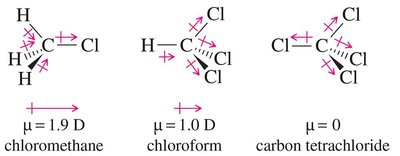
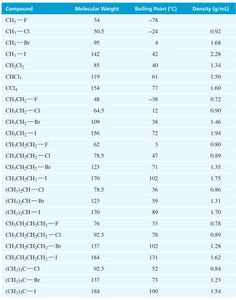
Applications of Halogenated Hydrocarbons
Halogenated hydrocarbons have diverse applications due to their chemical and physical properties:
Solvents: e.g., chloroform (CHCl3), dichloromethane (CH2Cl2).
Anesthetics: e.g., halothane (CF3CHClBr).
Refrigerants: e.g., Freons (CHClF2).
Polymers: e.g., tetrafluoroethylene (TFE) is the monomer for Teflon®.
Pesticides: e.g., DDT, a persistent organic pollutant.
Hormones: e.g., thyroxine, a thyroid hormone.
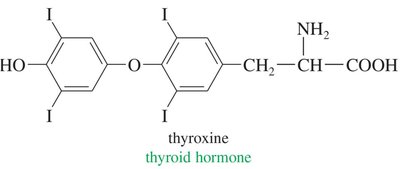



Preparation of Alkyl Halides
Free Radical Halogenation
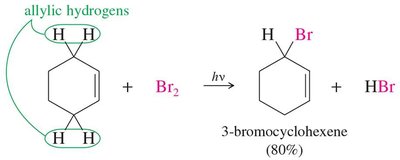
Alkyl halides can be synthesized by the free radical halogenation of alkanes. Chlorination produces mixtures, while bromination is more selective (3° >> 2° >> 1°). Fluorination is too violent, and iodination is too slow for practical use.
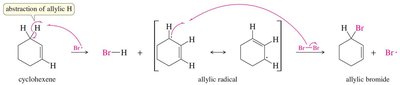
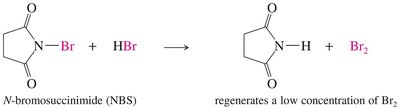
Allylic halogenation introduces a halogen at the allylic position (next to a double bond), often using N-bromosuccinimide (NBS) to maintain low Br2 concentration.
Other Methods of Preparation
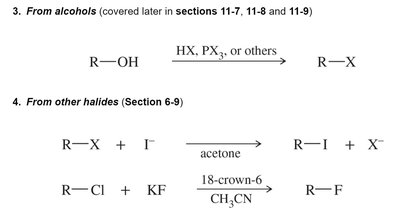
From alcohols: Reaction with HX, PX3, or other reagents.
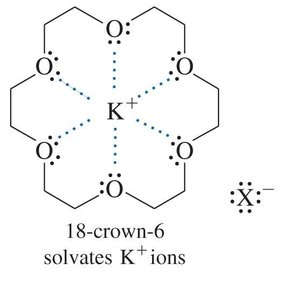
From other halides: Halide exchange reactions, often using acetone or crown ethers to facilitate the process.
Nucleophilic Substitution Reactions
General Mechanism
Nucleophilic substitution involves the replacement of a halogen atom (leaving group) by a nucleophile. The carbon-halogen bond is polarized, making the carbon electrophilic and susceptible to nucleophilic attack.
Two main mechanisms: SN2 (bimolecular) and SN1 (unimolecular).
Elimination reactions (not covered in detail here) compete with substitution and produce alkenes.
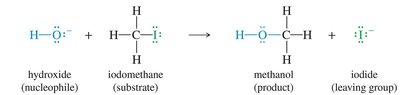
SN2 Mechanism (Bimolecular Nucleophilic Substitution)
The SN2 reaction is a one-step, concerted process where the nucleophile attacks the electrophilic carbon from the side opposite the leaving group, resulting in inversion of configuration (Walden inversion).
Rate law:
Transition state is trigonal bipyramidal.
Stereospecific: inversion at the chiral center.
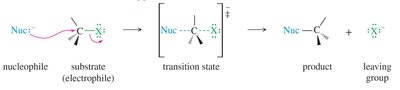
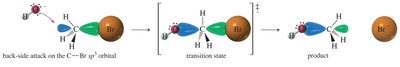
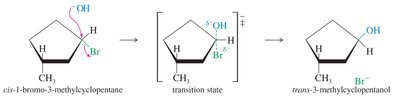
Factors Affecting SN2 Reactions
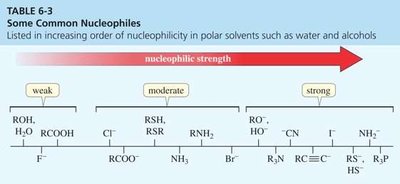
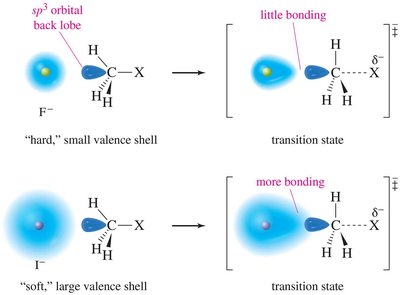
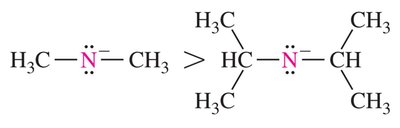
Nucleophile strength: Stronger nucleophiles increase the reaction rate. Negatively charged nucleophiles are generally stronger than their neutral counterparts.
Steric hindrance: Bulky substrates or nucleophiles slow the reaction.
Solvent effects: Polar aprotic solvents (e.g., acetone, DMSO) enhance nucleophilicity and favor SN2 reactions.
Leaving group: Good leaving groups are weak bases and stabilize the negative charge (I- > Br- > Cl- > F-).
Substrate structure: Reactivity order: methyl > 1° > 2° >> 3° (steric hindrance prevents SN2 on tertiary centers).
SN1 Mechanism (Unimolecular Nucleophilic Substitution)
The SN1 reaction proceeds via a two-step mechanism: first, the leaving group departs, forming a carbocation intermediate; second, the nucleophile attacks the carbocation. The reaction is not stereospecific and often leads to racemization.
Rate law:
Carbocation stability is crucial: 3° > 2° > 1° >> methyl.
Good leaving groups and polar protic solvents favor SN1.
Rearrangements (hydride or methyl shifts) can occur to form more stable carbocations.
Comparison of SN1 and SN2 Mechanisms
Feature | SN2 | SN1 |
|---|---|---|
Nucleophile | Strong | Weak |
Substrate Reactivity | CH3X > 1° > 2° | 3° > 2° |
Solvent | Polar aprotic | Polar protic |
Rate Law | Second order | First order |
Stereochemistry | Inversion | Racemization |
Rearrangements | No | Possible |
Summary Table: Reactivity of Alkyl Halides in SN1 and SN2
Substrate | SN1 Conditions (Weak Nucleophile) | SN2 Conditions (Strong Nucleophile) |
|---|---|---|
Methyl halides (CH3X) | No reaction (methyl cation too unstable) | Favored (unhindered) |
Primary halides (RCH2X) | Rare, unless resonance-stabilized cation | Favored unless R group is bulky |
Secondary halides (R2CHX) | SN1 possible, often with rearrangement | SN2 possible unless bulky |
Tertiary halides (R3CX) | SN1 occurs readily | SN2 cannot occur (steric hindrance) |
Additional info: The above notes integrate textbook-style explanations, key definitions, and relevant images to reinforce the concepts of alkyl halide structure, nomenclature, physical properties, preparation, and nucleophilic substitution mechanisms (SN1 and SN2). This content is suitable for college-level organic chemistry students preparing for exams or seeking a comprehensive overview of Chapter 6 topics.
