 Back
BackAromatic Chemistry: Structure, Properties, and Reactions of Benzene
Study Guide - Smart Notes

Aromatic Chemistry
Introduction to Aromatic Compounds
Aromatic compounds are a fundamental class of organic molecules characterized by their unique stability and reactivity, primarily due to the delocalization of π-electrons in a cyclic structure. Benzene is the prototypical aromatic compound, and understanding its chemistry is essential for grasping the behavior of many biologically and industrially important molecules.
Aromatic compounds contain a ring with delocalized π-electrons.
The study of benzene chemistry provides insight into the structure-reactivity relationship of aromatic systems, including those found in nucleic acids and pharmaceuticals.
Kekulé Structure and Limitations
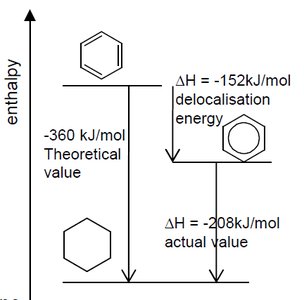
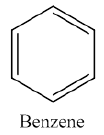
The Kekulé structure of benzene was an early model proposing alternating single and double bonds in a six-membered ring (1,3,5-cyclohexatriene). However, experimental evidence shows that benzene does not behave like a typical alkene and is much more stable than predicted by this model.
Benzene does not react with bromine water as expected for alkenes.
The enthalpy of hydrogenation for benzene is much less exothermic than for three isolated double bonds, indicating extra stability (delocalization energy).
Structure and Resonance of Benzene
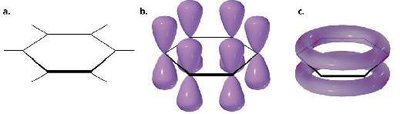
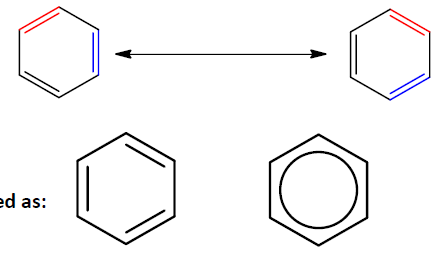
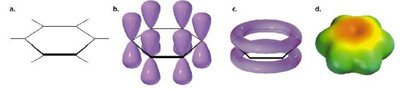
Benzene is less reactive than isolated alkenes due to the continuous overlap of six p-orbitals, forming a conjugated π-system. This delocalization leads to a resonance hybrid structure, where all carbon-carbon bonds are equivalent.
Each p-orbital overlaps with neighbors, creating a planar, cyclic, fully conjugated system.
Benzene is best represented as a resonance hybrid of two equivalent forms, often depicted with a circle inside the hexagon.
Definition and Examples of Aromatic Compounds
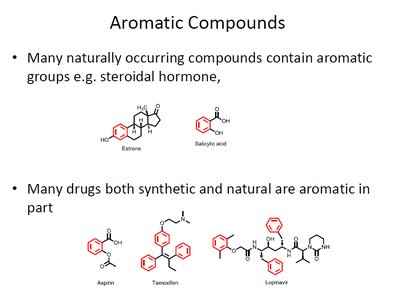
The term "aromatic" originally referred to fragrant substances but now denotes compounds with a ring of delocalized π-electrons. Many naturally occurring and synthetic compounds, including drugs and hormones, contain aromatic rings.
Examples: benzene, benzaldehyde, vanillin, estrone, salicylic acid, aspirin, tamoxifen, lopinavir.
Naming Aromatic Compounds
Monosubstituted and Alkylbenzenes
Monosubstituted benzenes are named with benzene as the parent. When the alkyl group is smaller than the ring, the compound is named as an alkylbenzene; if larger, as a phenyl-substituted alkane.
Examples: bromobenzene, nitrobenzene, propylbenzene.
The benzene ring as a substituent is called a phenyl group.
Alkyl-substituted benzenes are called arenes.

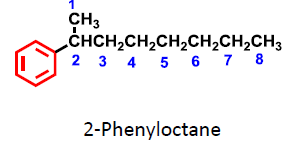
Disubstituted Benzenes: Ortho, Meta, Para
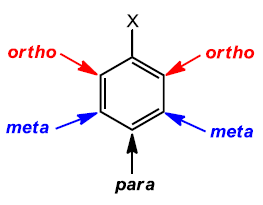
When two substituents are present, their relative positions are indicated by the prefixes ortho- (o-, 1,2-), meta- (m-, 1,3-), and para- (p-, 1,4-).
Ortho-: 1,2-substitution
Meta-: 1,3-substitution
Para-: 1,4-substitution


Aromaticity and Huckel’s Rule
Criteria for Aromaticity
Not all cyclic, conjugated molecules are aromatic. Aromaticity is defined by Huckel’s 4n+2 rule, which states that a molecule is aromatic if it is cyclic, planar, fully conjugated, and contains 4n+2 π-electrons (where n = 0, 1, 2, ...).
Only molecules with 2, 6, 10, 14, 18, ... π-electrons are aromatic.
Examples: Benzene (6 π-electrons, aromatic), cyclobutadiene (4 π-electrons, non-aromatic).


Application to Heterocycles and Biological Molecules
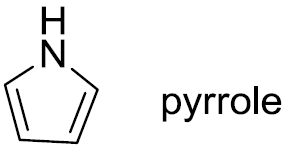
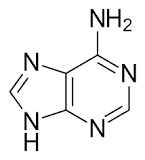
Huckel’s rule can be applied to heterocyclic compounds and nucleic acid bases. For example, pyrrole (5-membered ring with 6 π-electrons) is aromatic, while cyclopentadiene is not. Purine bases such as adenine and guanine are also aromatic due to their conjugated ring systems.
Pyrrole: aromatic (6 π-electrons)
Cyclopentadiene: non-aromatic (4 π-electrons)
Adenine and guanine: aromatic (conjugated systems with 10 π-electrons)
Reactions of Benzene: Electrophilic Aromatic Substitution (EAS)
General Mechanism of EAS
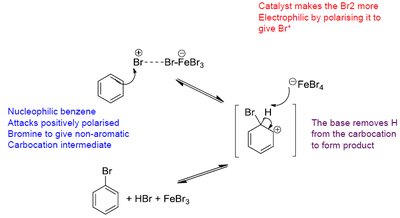
The defining reaction of benzene and other aromatic compounds is electrophilic aromatic substitution (EAS). In this reaction, an electrophile replaces a hydrogen atom on the aromatic ring. The mechanism involves the formation of a resonance-stabilized carbocation intermediate (σ-complex or arenium ion).
Step 1: Electrophile attacks the aromatic ring, forming a non-aromatic intermediate.
Step 2: Loss of a proton restores aromaticity.
Examples of EAS Reactions
Common types of EAS include halogenation, nitration, sulfonation, Friedel-Crafts alkylation, and acylation. Catalysts are often required to generate a sufficiently reactive electrophile.
Halogenation: Benzene reacts with Br2 in the presence of FeBr3 to form bromobenzene.
Nitration: Benzene reacts with a mixture of concentrated HNO3 and H2SO4 to form nitrobenzene (NO2+ is the electrophile).
Friedel-Crafts Alkylation/Acylation: Introduction of alkyl or acyl groups using AlCl3 as a catalyst.
Reaction Type | Electrophile | Catalyst | Product Example |
|---|---|---|---|
Halogenation | Br+, Cl+ | FeBr3, AlCl3 | Bromobenzene, Chlorobenzene |
Nitration | NO2+ | H2SO4 | Nitrobenzene |
Sulfonation | SO3H+ | H2SO4 | Benzenesulfonic acid |
Friedel-Crafts Alkylation | R+ | AlCl3 | Alkylbenzene |
Friedel-Crafts Acylation | RCO+ | AlCl3 | Aryl ketone |
Summary
Aromatic compounds are defined by cyclic, planar, conjugated systems with 4n+2 π-electrons (Huckel’s rule).
Benzene is the archetype, exhibiting unique stability and undergoing electrophilic aromatic substitution rather than addition reactions typical of alkenes.
Naming conventions and substitution patterns (ortho, meta, para) are essential for describing aromatic compounds.
Aromaticity extends to heterocycles and biologically important molecules such as nucleic acid bases.
