 Back
BackAromatic Compounds: Structure, Nomenclature, Stability, and Reactions
Study Guide - Smart Notes

Chapter 17: Aromatic Compounds
17.1 Introduction to Aromatic Compounds
Aromatic compounds, also known as arenes, are a fundamental class of organic molecules characterized by the presence of one or more benzene rings. These rings are highly prevalent in pharmaceuticals and other chemical products due to their unique stability and reactivity.
Aromatic rings are found in many drugs and natural products.
Benzene is the prototypical aromatic compound, with the formula C6H6.
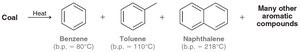
Coal is a source of aromatic compounds such as benzene, toluene, and naphthalene.
17.2 Nomenclature of Aromatic Compounds
The nomenclature of aromatic compounds is governed by IUPAC rules, but several common names are widely accepted. Benzene is typically the parent name for monosubstituted derivatives, and the phenyl group (Ph) is used to represent a benzene ring as a substituent.
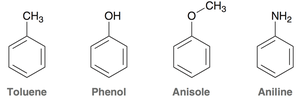
Common aromatic derivatives: Toluene, phenol, anisole, and aniline.
If the substituent is larger than the ring, it becomes the parent chain.
Dimethyl benzene derivatives are called xylene (ortho-, meta-, para-).
Substituent positions are indicated by ortho (1,2-), meta (1,3-), and para (1,4-).
Systematic naming involves identifying the parent, naming substituents, numbering the ring, and listing substituents alphabetically (ignoring prefixes except 'iso').
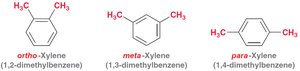
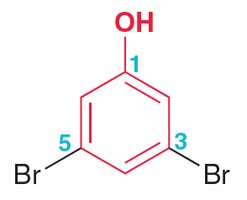
Example: Naming Aromatic Compounds
Assign locant 1 to the substituent that is part of the parent name.
List substituents in alphabetical order before the parent name.
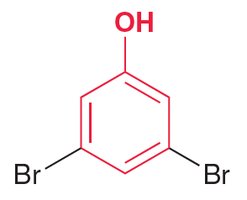
Example: 3,5-dibromophenol.

17.3 Structure of Benzene
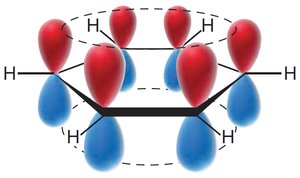
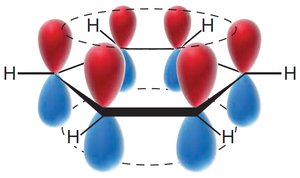
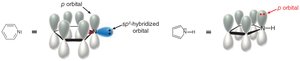
Benzene's structure was first proposed by Kekulé, who suggested alternating single and double bonds. However, modern understanding recognizes benzene as a resonance hybrid, with delocalized π electrons.
Benzene's molecular formula is C6H6.
Resonance structures show electron delocalization.
The circle in the hexagon represents delocalized π electrons.
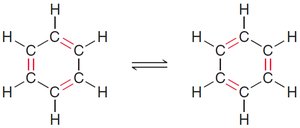
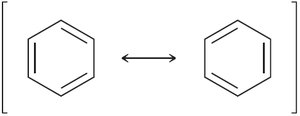
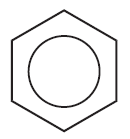
17.4 Stability of Benzene
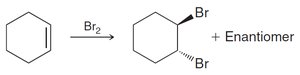
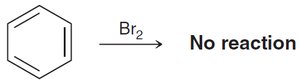
Benzene and other aromatic rings are remarkably stable compared to alkenes, which readily undergo addition reactions. This stability is explained by molecular orbital (MO) theory and Hückel's rule.
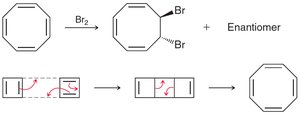
Benzene does not react with Br2 under normal conditions, unlike alkenes.
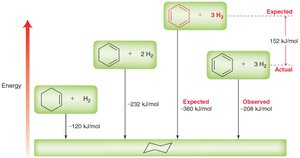
Stabilization energy is much higher than expected for a conjugated system.
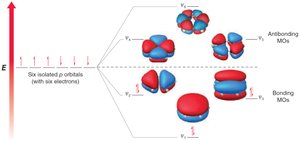
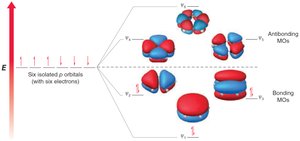
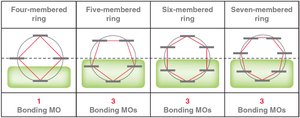
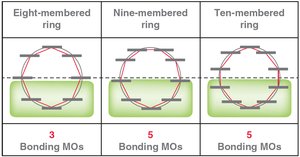
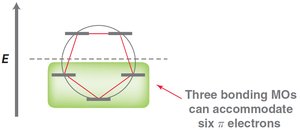
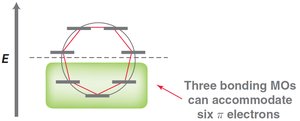
MO theory: Six p-orbitals overlap to form six MOs, with three bonding MOs accommodating six π electrons.
Hückel's rule: Aromatic compounds must have 4n+2 π electrons (n = 0, 1, 2...).
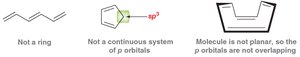
Antiaromatic compounds have 4n π electrons and are destabilized.
Nonaromatic compounds lack a fully conjugated ring or are not planar.


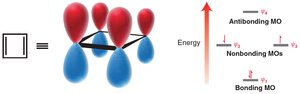
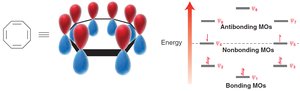
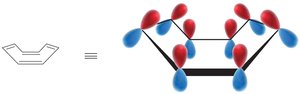
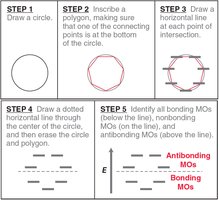
Frost Circles
Frost circles are a graphical method to predict the relative energies of molecular orbitals in cyclic conjugated systems, helping to explain the 4n+2 rule.
17.5 Other Aromatic Compounds
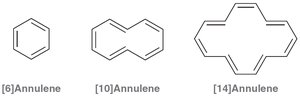
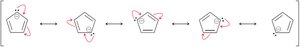
Aromaticity is not limited to benzene. Other fully conjugated rings, including annulenes, ions, and heterocycles, can be aromatic, antiaromatic, or nonaromatic depending on their electron count and structure.
Annulenes are fully conjugated rings; their aromaticity depends on π electron count.
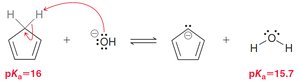
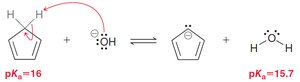
Carbanions and carbocations can be part of aromatic rings if they contribute to the π system.
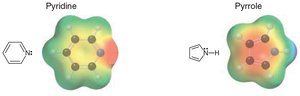
Heteroatoms (e.g., N, O, S) can participate in aromaticity if their lone pairs are part of the conjugated system.
Polycyclic aromatic hydrocarbons (PAHs) are also aromatic.
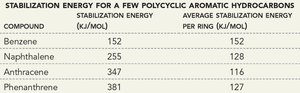
Compound | Stabilization Energy (kJ/mol) | Average Stabilization Energy per Ring (kJ/mol) |
|---|---|---|
Benzene | 152 | 152 |
Naphthalene | 255 | 128 |
Anthracene | 347 | 116 |
Phenanthrene | 381 | 127 |
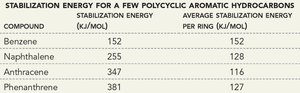
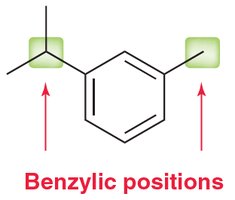
17.6 Reactions at the Benzylic Position
The benzylic position is the carbon directly attached to a benzene ring. This position is uniquely reactive, especially in oxidation and substitution reactions.
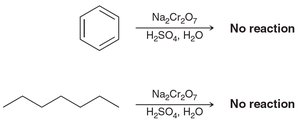
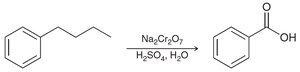
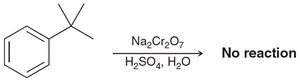
Aromatic rings and alkyl groups are not easily oxidized, but benzylic positions are.
Oxidation with chromic acid or permanganate requires at least one proton at the benzylic position.
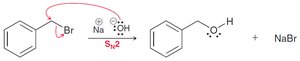
Benzylic positions undergo halogenation (chlorination, bromination) and substitution reactions (SN1 and SN2).
Benzylic bromides can be converted to alkenes.
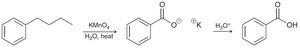
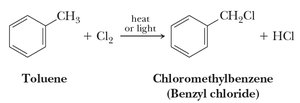
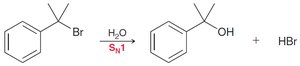
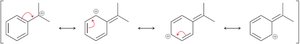
17.7 Reduction of Benzene
Benzene can be reduced under forceful conditions to cyclohexane, or partially reduced via Birch reduction. Regioselectivity in Birch reduction depends on the nature of substituents.
Alkenes can be selectively hydrogenated in the presence of benzene.
Birch reduction does not fully reduce the benzene ring.
Electron-donating and electron-withdrawing groups affect regioselectivity.
17.7 Buckyballs & Nanotubes
Fullerenes (buckyballs) and carbon nanotubes are unique forms of aromatic carbon structures with important applications in materials science.
Buckyballs (C60) are spherical aromatic molecules.
Nanotubes are cylindrical aromatic structures.
Summary Table: Key Concepts in Aromaticity
Type | Criteria | Example |
|---|---|---|
Aromatic | Fully conjugated ring, 4n+2 π electrons | Benzene |
Antiaromatic | Fully conjugated ring, 4n π electrons | Cyclobutadiene |
Nonaromatic | Not fully conjugated or not planar | Cyclooctatetraene (tub-shaped) |
Key Equations
Hückel's Rule: π electrons (where ) for aromaticity.
Stabilization Energy: (see energy diagram).
