 Back
BackAromatic Compounds: Structure, Properties, and Nomenclature
Study Guide - Smart Notes

Chapter 16: Aromatic Compounds
16.1 Introduction: The Discovery of Benzene
Aromatic compounds are a class of cyclic, conjugated molecules that exhibit unique stability and reactivity due to their electronic structure. Benzene is the prototypical aromatic compound, and many fragrant natural products are derivatives of benzene. Compounds lacking aromatic properties are termed aliphatic.
Aromatic: Cyclic, conjugated, and unusually stable compounds (e.g., benzene).
Aliphatic: Non-aromatic, "fat-like" compounds.
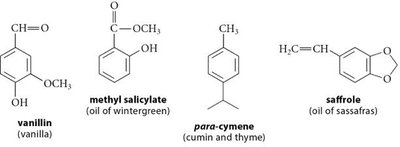
16.2 The Structure and Properties of Benzene
Benzene's structure was first proposed by Kekulé in 1865 as a cyclic conjugated triene. However, benzene's chemical reactivity differs significantly from typical alkenes, as it does not undergo addition reactions with bromine unless a Lewis acid is present, favoring substitution instead.
Kekulé Structure: Benzene as a six-membered ring with alternating double bonds.
Reactivity: Benzene resists addition reactions (e.g., with Br2), unlike alkenes such as cyclohexene or cyclooctatetraene.
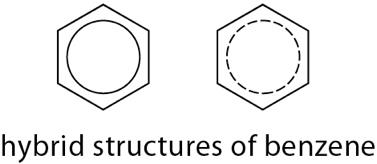
Resonance: Benzene is a single compound with six equivalent C–C bonds, intermediate between single and double bonds, due to delocalized π electrons.
Resonance Energy: The difference between predicted and observed heats of hydrogenation quantifies benzene's extra stability.
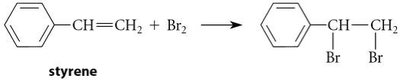
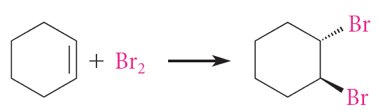
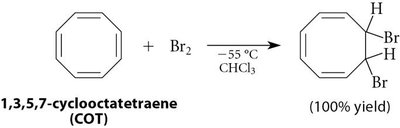
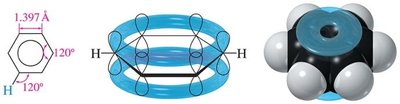
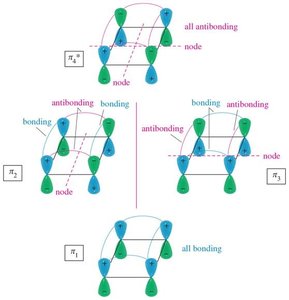
16.3 Molecular Orbital Picture of a Conjugated System
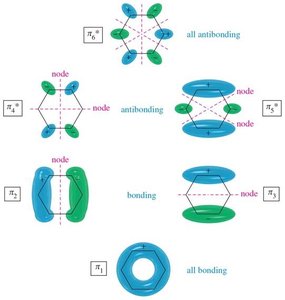
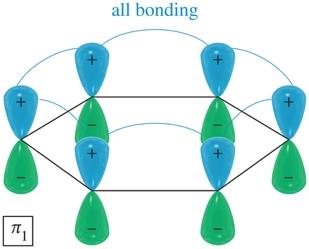
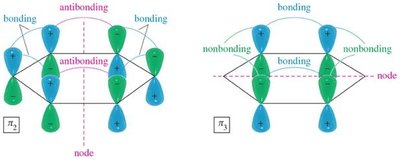
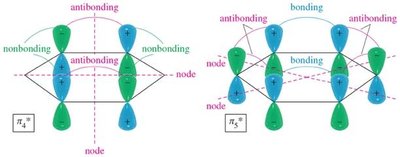
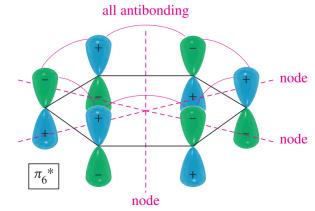
Benzene's six p orbitals combine to form six molecular orbitals (MOs): three bonding and three antibonding. The six π electrons fill the three bonding MOs, resulting in a closed-shell, highly stable system.
Bonding MOs: Lower in energy, filled with electrons.
Antibonding MOs: Higher in energy, unfilled in benzene.
Delocalization: All six π electrons are delocalized over the ring, contributing to aromatic stability.
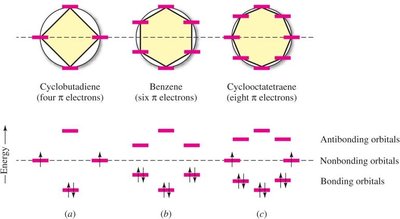
16.4 The Molecular Orbital Picture of Cyclobutadiene
Cyclobutadiene, with four π electrons, has two electrons in nonbonding MOs, making it a diradical and highly reactive. It does not exhibit aromatic stability.
Diradical Character: Two unpaired electrons in nonbonding orbitals.
Polygon Rule (Frost Circle): A graphical method to predict the relative energies of π orbitals in cyclic conjugated systems.
16.5 & 16.6 Aromatic, Antiaromatic, and Nonaromatic Compounds & Hückel’s Rule
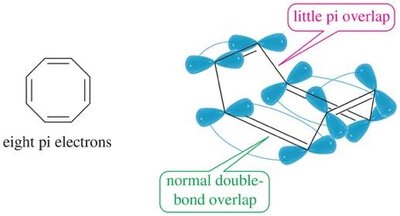
Hückel’s rule defines aromaticity: a planar, cyclic, fully conjugated polyene with (4n+2) π electrons is aromatic. Compounds with 4n π electrons are antiaromatic (destabilized), while those lacking continuous conjugation or planarity are nonaromatic.
Aromatic: Planar, cyclic, conjugated, (4n+2) π electrons (n = 0, 1, 2, ...).
Antiaromatic: Planar, cyclic, conjugated, 4n π electrons.
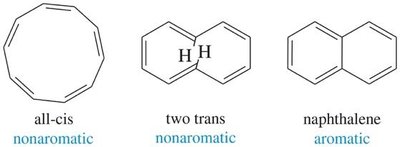
Nonaromatic: Not fully conjugated or not planar.

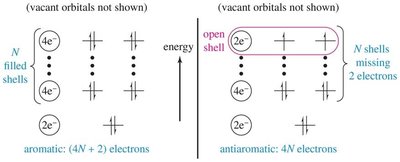
16.7 Molecular Orbital Derivation of Hückel’s Rule
The molecular orbital approach shows that aromatic compounds have filled bonding orbitals (closed shells) with (4n+2) electrons, while antiaromatic compounds have unpaired electrons in degenerate orbitals (open shells) with 4n electrons.
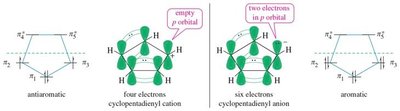
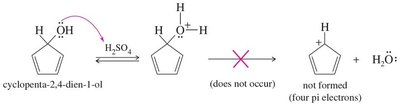
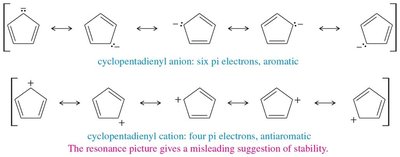
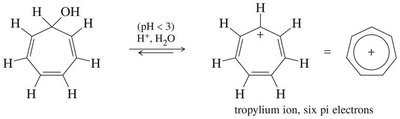
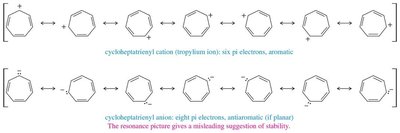
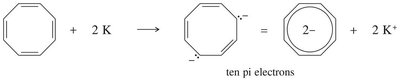
16.8 Aromatic Ions
Certain ions can also be aromatic or antiaromatic, depending on their electron count and structure. The cyclopentadienyl anion (6 π electrons) is aromatic, while the cation (4 π electrons) is antiaromatic. The tropylium ion (cycloheptatrienyl cation) is aromatic with 6 π electrons.
Cyclopentadienyl Anion: Aromatic, 6 π electrons, delocalized negative charge.
Cyclopentadienyl Cation: Antiaromatic, 4 π electrons, highly reactive.
Tropylium Ion: Aromatic, 6 π electrons, stable cation.
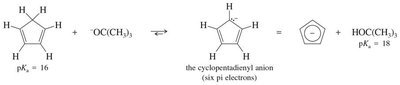
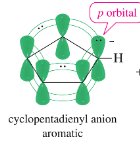
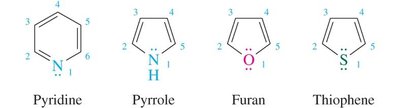
16.9 Heterocyclic Aromatic Compounds
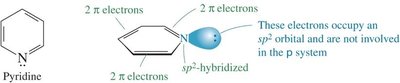
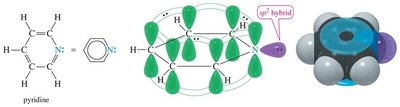
Heterocyclic aromatic compounds contain at least one non-carbon atom in the ring. The heteroatom may contribute zero or two electrons to the π system, depending on its hybridization and lone pairs. Pyridine and pyrrole are classic examples.
Pyridine: Six π electrons; nitrogen's lone pair is in an sp2 orbital, not part of the π system; basic.
Pyrrole: Six π electrons; nitrogen's lone pair is in a p orbital, part of the π system; weak base.
Other Heterocycles: Furan, thiophene, etc., can also be aromatic if they satisfy Hückel’s rule.
16.10 Polynuclear Aromatic Hydrocarbons (PAHs)
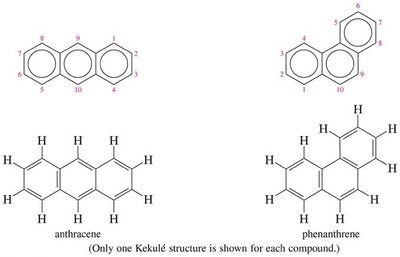
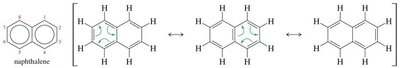
Polynuclear aromatic hydrocarbons are compounds with two or more fused benzene rings. Naphthalene is the simplest PAH. Larger PAHs, such as anthracene and phenanthrene, are found in combustion products and can be carcinogenic.
Naphthalene: Two fused benzene rings, aromatic.
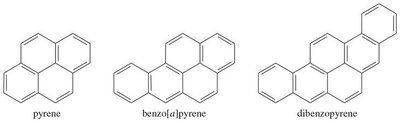
Larger PAHs: Anthracene, phenanthrene, pyrene, etc.
Health Effects: Some PAHs form epoxides that can react with DNA, leading to carcinogenicity.
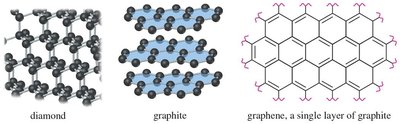
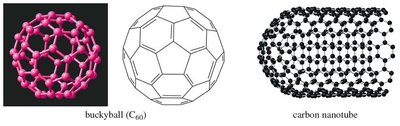
16.11 Aromatic Allotropes of Carbon
Carbon exists in several allotropes, some of which are aromatic in nature. These include graphite (layers of fused benzene rings), fullerenes (C60 "buckyballs"), and carbon nanotubes.
Diamond: Tetrahedral lattice, electrical insulator.
Graphite: Planar layers of fused benzene rings, conducts electricity parallel to layers.
Fullerenes: Spherical molecules made of 5- and 6-membered rings.
Nanotubes: Cylindrical structures of fused aromatic rings.
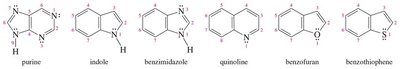
16.12 Fused Heterocyclic Compounds
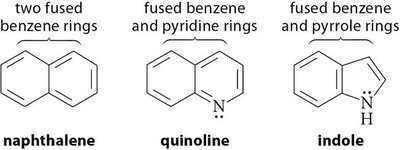
Fused heterocyclic aromatic compounds are common in nature and pharmaceuticals. Examples include purine, indole, quinoline, and benzimidazole.
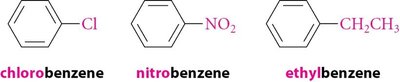
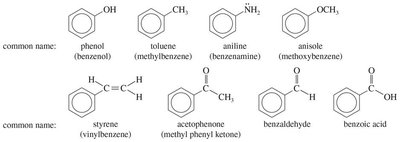
16.13 Nomenclature of Benzene Derivatives
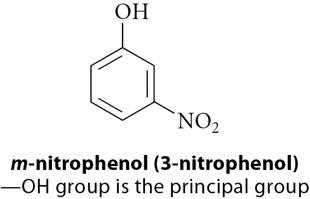
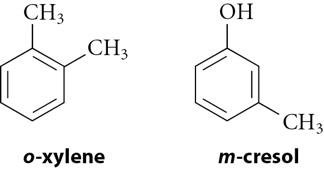
Benzene derivatives are named using IUPAC rules, with benzene as the parent. Common names are often used for simple derivatives. Disubstituted benzenes use ortho (1,2-), meta (1,3-), and para (1,4-) prefixes. When more than two substituents are present, numbers are used exclusively.
Principal Group: The highest priority substituent is at C-1.
Aryl Group: Substituted benzene ring (C6H5–).
Phenyl Group (Ph): C6H5–.
Benzyl Group (Bn): C6H5CH2–.
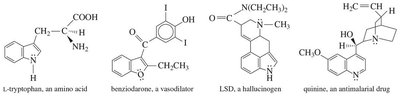
16.14 Physical Properties of Benzene and Its Derivatives
Aromatic compounds are generally more symmetrical than alkanes, resulting in higher melting points. Their boiling points depend on dipole moments, and they are typically more dense than nonaromatics but less dense than water. Aromatics are generally insoluble in water.
16.15 Spectroscopy of Aromatic Compounds
Spectroscopic techniques are essential for identifying aromatic compounds:
IR Spectroscopy: C=C stretch at 1600 cm–1; sp2 C–H stretch just above 3000 cm–1.
NMR Spectroscopy: 1H NMR at δ 7–8 ppm for aromatic hydrogens; 13C NMR at δ 120–150 ppm.
Mass Spectrometry: Useful for determining molecular weight and structure.
UV Spectroscopy: Benzene and derivatives show characteristic UV absorptions due to π→π* transitions.
