 Back
BackAromatic Compounds: Structure, Stability, and Reactions
Study Guide - Smart Notes

Aromatic Compounds
Introduction to Aromatic Compounds
Aromatic compounds are a class of organic molecules characterized by rings with conjugated π systems that exhibit unusual stability. The most well-known aromatic compound is benzene, and its derivatives are called arenes. Aromatic rings are prevalent in many pharmaceuticals and natural products, playing crucial roles in their chemical properties and biological activities.
Aromaticity refers to the special stability associated with certain conjugated ring systems.
Many aromatic compounds were originally named for their pleasant odors, but not all are fragrant.
Benzene rings are common in drugs such as Lipitor, Zyprexa, Norvasc, Prilosec, Prevacid, Plavix, Serevent, and Zoloft.
Nomenclature of Benzene Derivatives
Benzene derivatives are named based on the number and position of substituents on the ring. Both systematic and common names are used in practice.
Monosubstituted benzenes are named by prefixing the substituent to 'benzene' (e.g., chlorobenzene, nitrobenzene, ethylbenzene).
Common names accepted by IUPAC include toluene (methylbenzene), phenol (hydroxybenzene), anisole (methoxybenzene), aniline (aminobenzene), benzoic acid, benzaldehyde, styrene, and acetophenone.
If the substituent is larger than the benzene ring, the ring is called a phenyl group (Ph).
Disubstituted benzenes use the descriptors ortho (o-, 1,2-), meta (m-, 1,3-), and para (p-, 1,4-) to indicate relative positions (e.g., ortho-xylene, meta-xylene, para-xylene).
Polysubstituted benzenes are named by numbering the ring to give the lowest possible numbers to substituents and arranging them alphabetically.
Example: 5-bromo-2-chlorophenol is a trisubstituted benzene where the parent is phenol, and substituents are numbered for the lowest set of locants.
Structure of Benzene
Benzene (C6H6) is a planar, six-membered ring with alternating double and single bonds. However, resonance theory shows that all C–C bonds are equivalent, with a bond order of 1.5. The π electrons are delocalized over the entire ring, contributing to its stability.
Resonance structures do not represent electron movement but the delocalization of electrons.
Benzene is often depicted as a hexagon with a circle inside to indicate delocalized π electrons.


Stability of Benzene
Benzene is much more stable than expected for a compound with three double bonds. This is demonstrated by its resistance to addition reactions and its lower-than-expected heat of hydrogenation.
Expected ΔH° for hydrogenation of three double bonds:
Observed ΔH° for benzene:
The difference (152 kJ/mol) is the stabilization energy due to aromaticity.

Molecular Orbital (MO) Theory and Aromaticity
MO theory explains the stability of benzene by showing that its six p orbitals combine to form six molecular orbitals: three bonding and three antibonding. The six π electrons fill the bonding MOs, resulting in a closed-shell configuration and exceptional stability.
Hückel's Rule: A planar, cyclic, fully conjugated molecule is aromatic if it contains π electrons (where n is a non-negative integer).
Compounds with π electrons are antiaromatic and unusually unstable if planar.
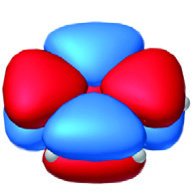
Criteria for Aromaticity
For a compound to be aromatic, it must:
Be cyclic and planar with a continuous ring of overlapping p orbitals (fully conjugated).
Contain a Hückel number of π electrons ().
Compounds that fail the first criterion are nonaromatic. Compounds that meet the first but have π electrons are antiaromatic.
Aromatic Compounds Other Than Benzene
Annulenes: Monocyclic compounds with conjugated π systems. [6]Annulene (benzene) is aromatic; [10]Annulene is nonaromatic due to nonplanarity; [14]Annulene and [18]Annulene can be aromatic if planar.
Aromatic Ions: The cyclopentadienyl anion (6 π electrons) and the tropylium cation (6 π electrons) are aromatic due to delocalization and planarity.
Heteroaromatic Compounds: Aromatic rings containing heteroatoms (N, O, S). Pyridine and pyrrole are classic examples, with lone pairs either localized (not part of aromaticity) or delocalized (contributing to aromaticity).

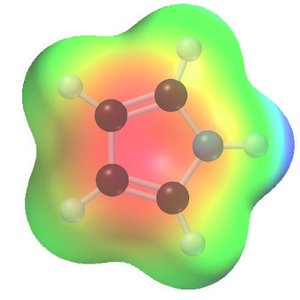
Polycyclic Aromatic Hydrocarbons (PAHs)
PAHs are compounds with fused aromatic rings, such as naphthalene, anthracene, and phenanthrene. Their stability can be measured by heats of hydrogenation, and the stabilization energy per ring is generally less than that of benzene.
Compound | Stabilization Energy (kJ/mol) | Average per Ring (kJ/mol) |
|---|---|---|
Benzene | 152 | 152 |
Naphthalene | 255 | 128 |
Anthracene | 347 | 116 |
Phenanthrene | 381 | 127 |

Reactions at the Benzylic Position
The benzylic position is the carbon directly attached to a benzene ring. This position is especially reactive in several types of reactions:
Oxidation: Alkylbenzenes are oxidized at the benzylic position by chromic acid (H2CrO4) or potassium permanganate (KMnO4) to give benzoic acid, provided the benzylic carbon is not quaternary.
Free-Radical Bromination: Benzylic positions undergo selective bromination with NBS (N-bromosuccinimide) due to resonance stabilization of the benzylic radical.
Substitution and Elimination: Benzylic halides undergo SN1, SN2, E1, and E2 reactions readily due to the stability of the benzylic carbocation and transition state.
Reduction of Benzene and Its Derivatives
Hydrogenation: Benzene can be hydrogenated to cyclohexane under high pressure and temperature. Under milder conditions, only alkenes are reduced, not aromatic rings.
Birch Reduction: Benzene and its derivatives can be partially reduced to 1,4-cyclohexadienes using sodium (or lithium), methanol, and ammonia. The regiochemistry depends on the nature of substituents (electron-donating or withdrawing).
Spectroscopy of Aromatic Compounds
IR Spectroscopy: Aromatic compounds show characteristic absorptions, including C–H stretching just above 3000 cm−1, combination bands (1700–2000 cm−1), and C–C stretching (1450–1600 cm−1).
1H NMR: Aromatic protons appear as multiplets between 6.5–8 ppm due to deshielding by the ring current. Benzylic protons appear between 2–3 ppm.
13C NMR: Aromatic carbons resonate between 110–170 ppm. The number and pattern of signals help determine substitution patterns.
Summary Table: Key Concepts in Aromaticity
Criterion | Description |
|---|---|
Planarity & Conjugation | Must have a planar, cyclic, fully conjugated π system |
Hückel's Rule | Must have π electrons (n = 0, 1, 2, ...) |
Stability | Exhibits unusual stability and low reactivity |
Reactivity | Undergoes substitution rather than addition reactions |
Applications and Biological Relevance
Aromatic rings are essential in pharmaceuticals (e.g., antihistamines), dyes, and materials science. Their unique stability and electronic properties make them central to organic chemistry and biochemistry.
