 Back
BackArrow Pushing Notation and Carbocation Rearrangements in Organic Reaction Mechanisms
Study Guide - Smart Notes

Arrow Pushing Notation for Reaction Mechanisms
Introduction to Curved Arrow Notation
Curved arrow notation is a fundamental tool in organic chemistry for illustrating the movement of electrons during chemical reactions. It is used to depict the making and breaking of chemical bonds, the flow of electron density, and the stepwise progression of reaction mechanisms.
Curved arrows show the movement of electron pairs.
They are essential for understanding reaction mechanisms and resonance structures.
Formal charges may change as electrons move; these must be tracked at each step.
Rules of Curved Arrow Notation
There are three legal moves for curved arrows in organic mechanisms:
Lone pair to bond: A lone pair of electrons forms a new bond.
Bond to lone pair: A bond breaks, resulting in a lone pair on an atom.
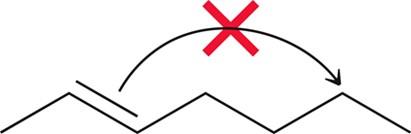
Bond to bond: A bond moves from one atom to another, often in rearrangements.
Each move must obey the octet rule and not result in atoms exceeding their allowed number of bonds.
Patterns of Arrow Pushing in Mechanisms
Four main patterns describe electron flow in polar organic reaction mechanisms:
Nucleophilic Attack
Loss of a Leaving Group
Proton Transfer (Acid/Base)
Rearrangements
Nucleophilic Attack
Nucleophilic attack involves a nucleophile (electron-rich species) donating a pair of electrons to an electrophile (electron-poor species).
The tail of the arrow starts at the nucleophile's electrons.
The head ends at the electrophile's nucleus.
Pi bonds can also act as nucleophiles.
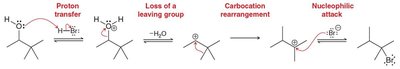
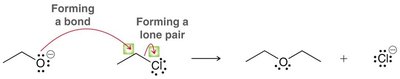
Loss of a Leaving Group
This pattern involves the breaking of a bond, with the leaving group taking the electron pair. Multiple arrows may be needed to show the complete process.
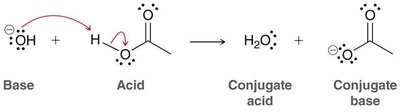
Proton Transfer (Acid/Base)
Proton transfer reactions (Bronsted acid/base) occur in one step, with electron density flowing from the base to the proton. The proton leaves behind its electrons as a lone pair on the conjugate base.
Usually requires two arrows: one for the base attacking the proton, and one for the bond breaking.
Rearrangements
Rearrangements involve changes to the connectivity of atoms, often to stabilize carbocations. These are common when carbocations are formed as intermediates.
Carbocations are unstable and will rearrange if a more stable carbocation can be formed.
Two main types: 1,2-hydride shift and 1,2-alkyl shift.
Carbocations: Structure and Stability
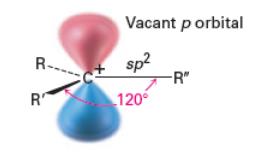
Carbocation Structure
Carbocations are positively charged carbon atoms with three bonds and no lone pairs, making them electron-deficient and highly reactive intermediates.
Carbocations are sp2 hybridized with a vacant p orbital.
The geometry is trigonal planar with bond angles of approximately 120°.
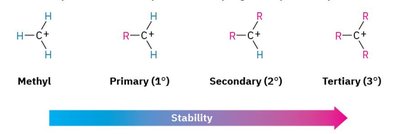
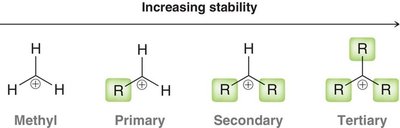
Carbocation Stability
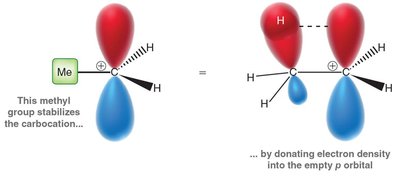
Carbocation stability increases with the number of alkyl groups attached to the positively charged carbon. This is due to hyperconjugation and inductive effects.
Methyl < Primary < Secondary < Tertiary
Hyperconjugation: Adjacent sigma bonds donate electron density into the empty p orbital, stabilizing the positive charge.
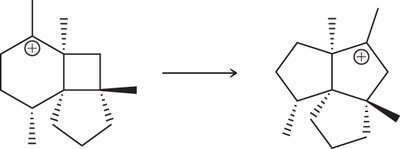
Carbocation Rearrangements
Types of Rearrangement
Carbocation rearrangements occur to form more stable carbocations. The two main types are:
1,2-Hydride Shift: A hydride (H-) migrates from an adjacent carbon to the carbocation.
1,2-Alkyl Shift: An alkyl group (R-) migrates from an adjacent carbon to the carbocation.
These shifts only occur if the resulting carbocation is more stable.

Combining Arrow Pushing Patterns in Multi-Step Mechanisms
Complex organic reactions often involve multiple steps, each corresponding to one or more of the four arrow pushing patterns. Understanding these patterns allows chemists to predict and rationalize reaction outcomes.

Common Mistakes in Arrow Drawing
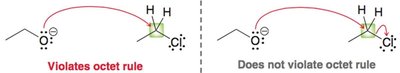
Violating the Octet Rule
Curved arrows must not result in atoms exceeding their allowed number of bonds (octet rule). The head of the arrow should always indicate the formation of a bond or a lone pair, never more than four bonds on carbon.
Practice and Application
Mastery of arrow pushing notation is essential for understanding organic reaction mechanisms. Practice is recommended through textbook problems, homework, and workshops.
Identify the four main patterns in mechanisms.
Draw arrows carefully, ensuring they follow the legal moves and do not violate the octet rule.
Summary Table: Carbocation Stability
Type | Structure | Stability |
|---|---|---|
Methyl | CH3+ | Least stable |
Primary | RCH2+ | Low stability |
Secondary | R2CH+ | Moderate stability |
Tertiary | R3C+ | Most stable |
