 Back
BackAtomic and Molecular Structure: Foundations for Organic Chemistry
Study Guide - Smart Notes

Part 1: Penicillin – Structure, Mechanism, and Resistance
Introduction to Penicillins
Penicillins are a class of wide-spectrum bactericidal antibiotics, including drugs such as amoxicillin and phenoxymethylpenicillin. They are among the most important antibiotics in medical history and are included on the World Health Organization's list of essential medicines.
Discovery: The antibiotic action of penicillin was first observed by Alexander Fleming in 1929, who noticed inhibition of bacterial growth by a mold contaminant.
Development: The drug was later developed for clinical use by Howard Florey, Ernst Chain, and their team at Oxford in 1939.
Importance: Penicillins were the first wide-spectrum antibiotics and played a crucial role in treating infections, especially during World War II.

Chemical Structure of Penicillins
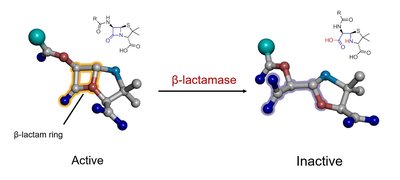
All penicillin antibiotics share a unique chemical feature: the beta-lactam ring. This four-membered cyclic amide is highly strained, making it reactive and essential for the antibiotic's function.
Beta-lactam ring: The ring's bond angles (90°) are much smaller than the ideal tetrahedral angle (109.5°), resulting in significant ring strain and reactivity.
Mechanism: The beta-lactam ring acts as a 'spring-loaded trap' that binds to and inactivates bacterial enzymes essential for cell wall synthesis.
Mechanism of Action: Inhibition of Transpeptidases
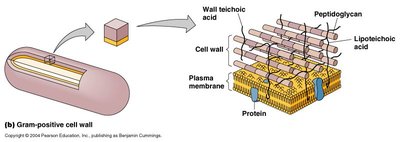
Penicillins target and irreversibly inhibit transpeptidase enzymes (also known as penicillin-binding proteins, PBPs), which are critical for cross-linking the peptidoglycan layers in bacterial cell walls.
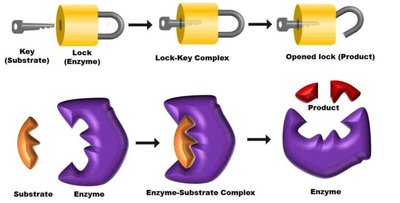
Lock and Key Model: The substrate (peptidoglycan precursor) fits into the enzyme's active site like a key into a lock. Penicillin mimics the substrate and binds irreversibly, inactivating the enzyme.
Result: Without cross-linking, the bacterial cell wall cannot withstand osmotic pressure, leading to cell lysis (osmolysis) and death.
Bacterial Resistance to Penicillins
Bacteria can develop resistance to penicillins through the production of beta-lactamase enzymes, which hydrolyze the beta-lactam ring, rendering the antibiotic inactive.
Beta-lactamase: Enzymes that break open the beta-lactam ring, conferring resistance to the bacterial organism.
Beta-lactamase inhibitors: Compounds such as clavulanic acid and tazobactam are often co-administered with penicillins to inhibit beta-lactamase enzymes and restore antibiotic efficacy.
Part 2: Atoms, Electrons, and Orbitals
Atomic Structure and Quantum Numbers
Understanding atomic structure is foundational for organic chemistry. Atoms consist of a nucleus (protons and neutrons) surrounded by electrons, which occupy regions of space called atomic orbitals.
Electrons: Subatomic particles with both wave-like and particle-like properties. Their behavior is described by quantum mechanics.
Orbitals: Mathematical functions (wavefunctions) that describe the probability distribution of an electron around the nucleus.
Quantum Numbers
Principal quantum number (n): Determines the size and energy of the orbital (n = 1, 2, 3, ...).
Angular momentum quantum number (l): Determines the shape of the orbital (l = 0 for s, 1 for p, 2 for d, 3 for f).
Magnetic quantum number (ml): Specifies the orientation of the orbital in space (ml = -l to +l).
Types of Atomic Orbitals
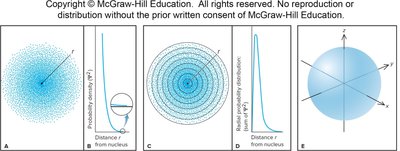
s orbitals (l = 0): Spherically symmetric; each shell has one s orbital.
p orbitals (l = 1): Dumbbell-shaped; each shell from n = 2 has three p orbitals (px, py, pz).
d orbitals (l = 2): Cloverleaf-shaped; each shell from n = 3 has five d orbitals.
Nodes in Orbitals
Radial nodes: Spherical regions where the probability of finding an electron is zero. Number of radial nodes = n – 1 – l.
Angular nodes: Planar regions where the probability is zero. Number of angular nodes = l.
Visualizing Orbitals
Orbitals can be visualized using contour surface plots (showing the boundary surface) and probability density plots (showing where electrons are most likely to be found).
Part 3: Electron Configurations and Lewis Dot Structures
Electron Configuration Principles
Aufbau Principle: Electrons fill orbitals in order of increasing energy.
Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons with opposite spins.
Hund’s Rule: Electrons occupy degenerate orbitals singly before pairing up.
Valence Electrons and Lewis Dot Structures
Valence shell: The outermost occupied electron shell of an atom.
Valence electrons: Electrons in the valence shell, important for chemical bonding and reactivity.
Lewis dot structure: A representation showing the element symbol and its valence electrons as dots.
Students should be able to draw Lewis dot structures for elements in the first three rows of the periodic table and for simple molecules, as well as calculate formal charges and convert between resonance structures using curved arrow notation.
Summary Table: Quantum Numbers and Orbitals
Quantum Number | Symbol | Possible Values | Physical Meaning |
|---|---|---|---|
Principal | n | 1, 2, 3, ... | Shell (size, energy) |
Angular Momentum | l | 0 to n-1 | Subshell (shape) |
Magnetic | ml | -l to +l | Orientation |
Key Equations
Number of radial nodes:
Number of angular nodes:
Example: For a 3p orbital (n = 3, l = 1): Radial nodes = 3 – 1 – 1 = 1 Angular nodes = 1
Additional info: Mastery of atomic structure, electron configuration, and the chemical basis of antibiotics like penicillin is foundational for understanding organic reaction mechanisms, molecular structure, and reactivity in subsequent chapters of organic chemistry.
