 Back
BackAtoms, Elements, and the Periodic Table: Foundations for Organic Chemistry
Study Guide - Smart Notes

Atoms and Subatomic Particles
The Structure of the Atom
The atom is the fundamental unit of matter, composed of a nucleus containing protons and neutrons, surrounded by electrons in an electron cloud. The nucleus is extremely small compared to the overall size of the atom, but it contains most of the atom's mass.
Protons: Positively charged particles found in the nucleus.
Neutrons: Neutral particles found in the nucleus.
Electrons: Negatively charged particles that orbit the nucleus in the electron cloud.
Electron Cloud: Much larger than the nucleus, not to scale in most diagrams.
Nuclear Force: Holds protons and neutrons together in the nucleus. Electrostatic Force: Repulsion between like charges (protons).
Key Concept: For a stable nucleus, the nuclear force must be stronger than the electrostatic force.
Subatomic Particle Properties
Subatomic particles differ in mass and charge. The atomic mass unit (amu) is used to express their masses.
1 amu = kg
Proton: 1.67262 x 10-27 kg, +1 charge
Neutron: 1.67493 x 10-27 kg, 0 charge
Electron: 0.00091 x 10-27 kg, -1 charge
Isotopes and Atomic Mass
Isotopes
Isotopes are atoms of the same element (same number of protons) but different numbers of neutrons. The atomic number (Z) identifies the element, while the mass number (A) is the sum of protons and neutrons.
Atomic Number (Z): Number of protons
Mass Number (A): Number of protons + neutrons
Isotope Notation:
Number of Neutrons:
Atomic Mass
The atomic mass of an element is the weighted average of all its isotopes, expressed in amu, Da (Dalton), or kg.
Most Abundant Isotope: The isotope whose mass number is closest to the atomic mass listed on the periodic table.
The Periodic Table
History and Organization
The periodic table organizes elements by increasing atomic number and recurring chemical properties (periodic law). Element symbols are usually derived from their English or Latin names.
Classifications
Elements are classified as metals, nonmetals, or metalloids based on their physical and chemical properties.
Metals: Largest classification; typically lustrous, conductive, malleable, and opaque.
Nonmetals: Second largest; typically brittle, poor conductors, non-ductile, and non-malleable.
Metalloids: Have properties of both metals and nonmetals; found along the 'staircase' border.

Groups and Periods
The periodic table is organized into horizontal rows (periods) and vertical columns (groups or families). There are 18 groups, each with unique properties. Elements in the same group have similar chemical properties due to similar electron arrangements.
Representative Elements and Transition Metals
Transition Metals: Groups 3-12, known for variable oxidation states.
Representative Elements: Groups 1-2 and 13-18, also called main group elements.
Elemental Forms
Monoatomic: Stable as single atoms (e.g., noble gases).
Diatomic: Stable as pairs (e.g., H2, O2, N2).
Polyatomic: Stable in groups larger than two (e.g., S8).
Phases of Elements
At room temperature and pressure, elements exist as solids, liquids, or gases.
Solids: Fixed shape and volume
Liquids: Fixed volume, variable shape
Gases: Variable shape and volume
Atomic Theory and Models
Dalton's Atomic Theory
John Dalton proposed that matter is composed of indivisible atoms, which combine in simple ratios to form compounds. Modern atomic theory recognizes subatomic particles and isotopes.
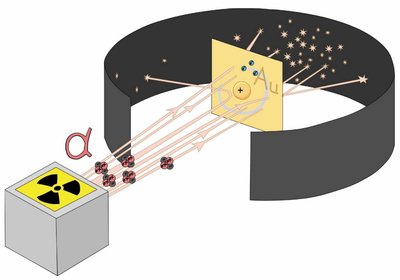
Rutherford's Gold Foil Experiment
Ernest Rutherford's experiment demonstrated the existence of a dense, positively charged nucleus at the center of the atom, surrounded by electrons.
Electromagnetic Spectrum and Atomic Models
Electromagnetic Spectrum
The electromagnetic spectrum includes all wavelengths and frequencies of electromagnetic radiation, from radio waves to gamma rays. Energy increases as wavelength decreases and frequency increases.
Photon: A quantum of electromagnetic energy.
Visible Light: The portion of the spectrum visible to the human eye.


Bohr Model
In the Bohr Model, electrons occupy discrete energy levels (shells) around the nucleus. Electrons can absorb or emit energy to move between shells.
Absorption: Electron moves to a higher shell (higher n).
Emission: Electron moves to a lower shell (lower n).
Energy difference:
Electron Arrangements and Valence Electrons
Electron Arrangement
The electron arrangement describes the distribution of electrons among energy levels. As n increases, the size and energy of the orbital increase.
Valence Electrons
Valence electrons are the outermost electrons involved in chemical bonding. For main group elements, the number of valence electrons equals the group number.
Inner Core Electrons: All electrons not in the outer shell.
Lewis Dot Symbols
Lewis Dot Symbols represent valence electrons as dots around the element symbol. Transition metals may have unpredictable patterns.
Periodic Trends
Metallic Character
Metallic character describes how easily an element loses electrons. It decreases across a period (left to right) and increases down a group.
Metals: Tend to lose electrons easily.
Nonmetals: Tend to gain electrons.
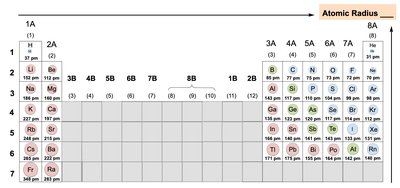
Atomic Radius
Atomic radius is the distance from the nucleus to the outermost electron shell. It increases down a group and decreases across a period.
Ionization Energy
Ionization energy is the energy required to remove an electron from an atom. It increases across a period and decreases down a group.
High IE: Electron is not easily lost (e.g., noble gases).
Low IE: Electron is easily lost (e.g., alkali metals).
Summary Table: Subatomic Particles
Subatomic Particle | Actual Mass (kg) | Relative Mass (amu) | Relative Charge |
|---|---|---|---|
Neutron | 1.67493 x 10-27 | 1 | 0 |
Proton | 1.67262 x 10-27 | 1 | +1 |
Electron | 0.00091 x 10-27 | ~0 | -1 |
Summary Table: Periodic Table Classifications
Classification | Physical Properties |
|---|---|
Metals | Lustrous, conductive, malleable, opaque |
Nonmetals | Brittle, poor conductors, non-ductile, non-malleable |
Metalloids | Intermediate properties, semi-conductors |
