 Back
BackBenzene and Aromatic Compounds: Structure, Stability, and Aromaticity
Study Guide - Smart Notes

Benzene and Aromatic Compounds
Hydrocarbons: Aromatic vs. Aliphatic
Hydrocarbons are organic compounds composed solely of carbon and hydrogen. They are classified as aliphatic (open-chain or non-aromatic) or aromatic (containing benzene rings or similar structures).
Arenes are aromatic hydrocarbons based on the benzene ring.
Aromatic compounds meet the Hückel criteria for aromaticity, exhibiting special stability.
Benzene is the prototypical aromatic hydrocarbon.
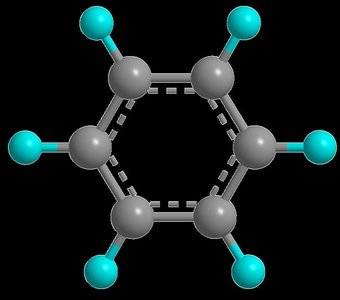
Benzene: Structure and Bonding
Benzene (C6H6) is a planar, cyclic molecule with six carbon atoms forming a regular hexagon. Each carbon is bonded to one hydrogen atom. The structure is often depicted with alternating single and double bonds, but in reality, all C–C bonds are of equal length due to electron delocalization.
Kekulé's model proposed rapid equilibrium between two cyclohexatriene structures, but experimental evidence shows all bonds are equivalent.
Bond angles in benzene are 120°, consistent with sp2 hybridization.
C–C bond length in benzene is 139 pm, intermediate between a single (153 pm) and double (134 pm) bond.
Electron delocalization: π-electrons are shared equally across all six carbons, forming a stable aromatic ring.
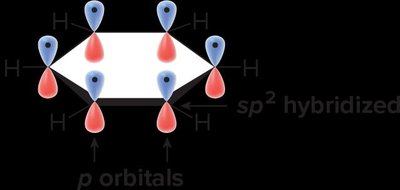

Electron Delocalization and Resonance
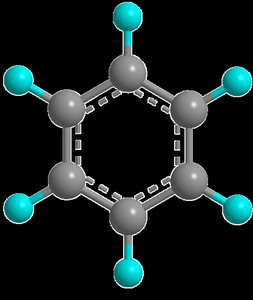
Benzene is best represented as a resonance hybrid of two equivalent Kekulé structures. The delocalization of π-electrons results in bonds that are neither purely single nor double, but intermediate.
Resonance stabilizes the molecule, making it less reactive than typical alkenes.
Delocalization is indicated by a circle inside the hexagonal ring in structural representations.
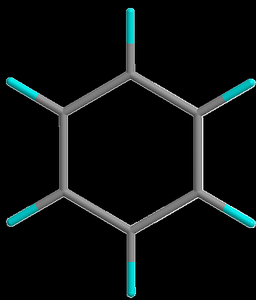
Nomenclature of Benzene Derivatives
Benzene derivatives are named based on the substituents attached to the ring. Monosubstituted derivatives use the parent name 'benzene' with the substituent.
Toluene: methylbenzene (CH3-benzene)
Styrene: ethenylbenzene (CH=CH2-benzene)
Anisol: methoxybenzene (OCH3-benzene)
Phenol: hydroxybenzene (OH-benzene)
Aniline: aminobenzene (NH2-benzene)
Benzoic acid: benzenecarboxylic acid (COOH-benzene)
Benzaldehyde: benzenecarbaldehyde (CHO-benzene)
Acetophenone: 1-phenylethanone (CCH3-benzene)
Disubstituted and Polysubstituted Benzene Derivatives
When two or more substituents are present, their positions are indicated by numbers or prefixes:
Ortho (o-): 1,2-substitution
Meta (m-): 1,3-substitution
Para (p-): 1,4-substitution
Substituents are listed alphabetically, and the ring is numbered to give the lowest possible numbers.
Spectroscopic Properties of Benzene
Benzene and its derivatives exhibit characteristic spectroscopic features:
IR absorption: Csp2-H at 3150–3000 cm-1; C=C (arene) at 1600, 1500 cm-1
1H NMR: Aryl-H at 6.5–8.0 ppm (highly deshielded); Benzylic-H at 1.5–2.5 ppm
13C NMR: Csp2 of arenes at 120–150 ppm
Benzene’s Stability and Aromaticity
Unusual Stability of Benzene
Benzene is significantly more stable than predicted by simple Lewis structures. This stability is termed aromaticity.
Measured by comparing the experimental heat of hydrogenation (ΔH°) to the predicted value for cyclohexatriene.
Observed ΔH° for benzene is –208 kJ/mol, much less than the expected –360 kJ/mol.
Stabilization energy is 152 kJ/mol, indicating benzene is less reactive and more stable.





Criteria for Aromaticity: Hückel’s Rule
Hückel’s rule defines the requirements for aromaticity in planar, monocyclic, fully conjugated polyenes:
Cyclic: Each p-orbital must overlap in a ring.
Planar: All p-orbitals must be aligned for π-electron delocalization.
Conjugated: A p-orbital on every atom in the ring.
(4n + 2) π-electrons: Only rings with this number of π-electrons are aromatic (n = 0, 1, 2, ...).
Systems with 4n π-electrons are antiaromatic and unstable.




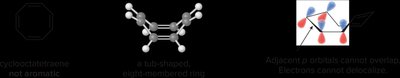
Antiaromatic and Non-Aromatic Compounds
Antiaromatic compounds are planar, monocyclic, fully conjugated, but have 4n π-electrons, making them unstable. Non-aromatic compounds fail one or more criteria for aromaticity.
Antiaromatic: Cyclobutadiene (4 π-electrons)
Non-aromatic: Cyclooctatetraene (adopts a non-planar shape)

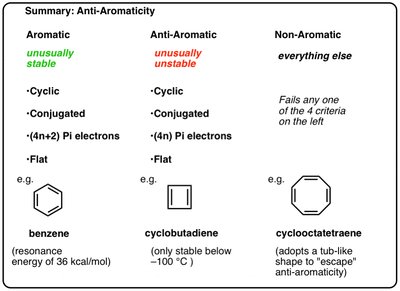


Aromatic | Anti-Aromatic | Non-Aromatic |
|---|---|---|
Unusually stable Cyclic Conjugated (4n+2) π-electrons Flat e.g., benzene | Unusually unstable Cyclic Conjugated (4n) π-electrons Flat e.g., cyclobutadiene | Everything else Fails any one of the 4 criteria e.g., cyclooctatetraene |

Examples of Aromatic Compounds and Ions
Annulenes and Polycyclic Aromatics
Annulenes are single-ring aromatics with alternating single and double bonds. Polycyclic aromatics, such as naphthalene (10 π-electrons) and phenanthracene (14 π-electrons), also exhibit aromaticity.
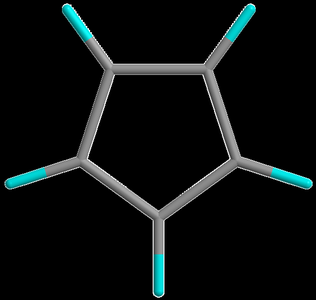
Aromatic Ions
Cycloheptatrienyl cation (tropylium): 6 π-electrons, cyclic, planar, conjugated, very stable.
Cyclopentadienyl anion: 6 π-electrons, planar, conjugated, stabilized by delocalization.
Cyclopropenyl cation: 2 π-electrons, aromatic.
Heterocyclic Aromatic Compounds
Five- and Six-Membered Rings
Aromaticity is not limited to hydrocarbons; it also occurs in heterocyclic compounds containing atoms like N, O, or S.
Furan: Five-membered ring with oxygen
Thiophene: Five-membered ring with sulfur
Pyrrole: Five-membered ring with nitrogen
Pyridine: Six-membered ring with nitrogen
Pyrimidine, Pyrazine: Six-membered rings with two nitrogens

Bonding and Molecular Orbital Theory
Valence Bond vs. Molecular Orbital Theory
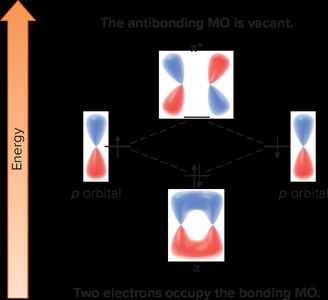
Valence Bond Theory explains σ and π bonds but is inadequate for aromatic compounds due to extensive p-orbital overlap. Molecular Orbital (MO) Theory describes the combination of atomic orbitals to form molecular orbitals, explaining aromatic stability.
In benzene, six p-orbitals combine to form six MOs: three bonding and three antibonding.
All bonding MOs are filled, resulting in aromatic stability.
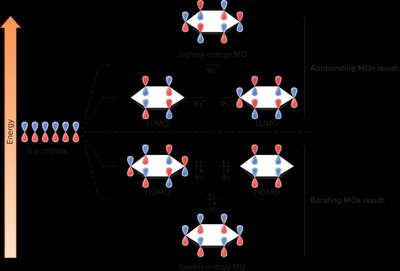
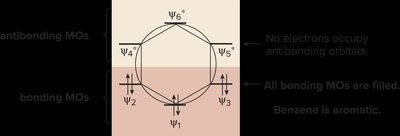
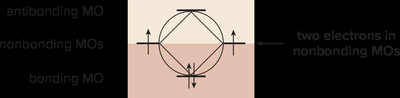
Inscribed Polygon Method for Predicting Aromaticity
This method helps visualize the relative energies of MOs in cyclic systems:
Draw the polygon with vertices pointing down.
Fill orbitals with electrons, starting from the lowest energy.
All bonding MOs filled = aromatic; any electrons in antibonding MOs = not aromatic.
Summary
Benzene is the archetype of aromatic compounds, exhibiting unique stability due to electron delocalization.
Hückel’s rule defines aromaticity: cyclic, planar, conjugated, and (4n+2) π-electrons.
Antiaromatic compounds are unstable; non-aromatic compounds fail one or more criteria.
Aromaticity extends to ions and heterocyclic compounds.
Molecular Orbital Theory provides a robust explanation for aromatic stability.
