 Back
BackBonding and Electronic Structure in Organic Molecules
Study Guide - Smart Notes

Bonding in Organic Molecules
Wave Nature of Electrons and Atomic Orbitals
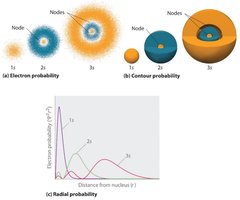
The quantum mechanical model of the atom, based on the wave nature of electrons, describes electrons as occupying regions of space called atomic orbitals. These orbitals are solutions to the Schrödinger wave equation and are represented by wavefunctions (ψ). The probability of finding an electron in a particular region is given by the square of the wavefunction, ψ2.
Atomic orbitals differ in size, shape, and orientation depending on their energy level.
Electrons in second-row elements (e.g., carbon, nitrogen, oxygen) occupy the five lowest energy atomic orbitals: 1s, 2s, 2px, 2py, and 2pz.
The spatial distribution and probability of finding electrons in these orbitals can be visualized using electron probability maps and contour diagrams.
Energy Levels and Electron Configuration
The energy of an electron in an atom is determined by its orbital. Lower energy orbitals (e.g., 1s) are filled first, followed by higher energy orbitals (e.g., 2s, 2p). The arrangement of electrons in these orbitals is called the electron configuration.
For carbon (atomic number 6): 1s2 2s2 2p2
For nitrogen (atomic number 7): 1s2 2s2 2p3
For oxygen (atomic number 8): 1s2 2s2 2p4
Bond Energy and Bond Length
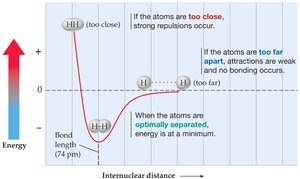
The strength of a chemical bond is related to the distance between the nuclei of the bonded atoms. The bond energy is the energy required to break a bond, and the bond length is the optimal distance where the energy is minimized.
If atoms are too close, repulsive forces dominate; if too far, attractive forces are weak and no bond forms.
The optimal bond length corresponds to the minimum energy point on the energy vs. distance curve.
Hybridization and Molecular Geometry
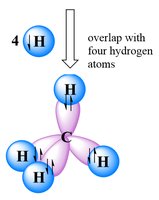
sp3 Hybridization in Methane and Ethane
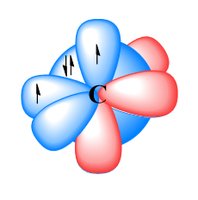
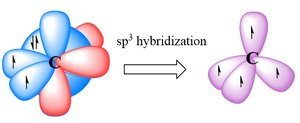
To explain the observed geometry of molecules like methane (CH4), Linus Pauling proposed hybridization: the mixing of atomic orbitals to form new, equivalent hybrid orbitals. In methane, carbon forms four equivalent sp3 hybrid orbitals, each forming a σ bond with hydrogen.
sp3 hybridization involves mixing one s and three p orbitals.
The resulting geometry is tetrahedral, with bond angles of 109.5°.
sp2 and sp Hybridization
Other hybridizations explain the bonding in molecules with double or triple bonds:
sp2 hybridization: Mixing one s and two p orbitals forms three sp2 hybrids (trigonal planar, 120° bond angles). The unhybridized p orbital forms a π bond in alkenes (e.g., ethene).
sp hybridization: Mixing one s and one p orbital forms two sp hybrids (linear, 180° bond angles). Two unhybridized p orbitals form two π bonds in alkynes (e.g., ethyne).
Valence Bond Theory and Sigma Bonding
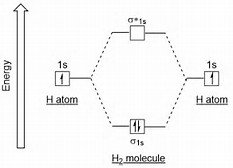
Sigma Bonding in H2
According to valence bond theory, a σ bond forms when two atomic orbitals overlap head-on, creating a region of high electron density between the nuclei. In H2, two 1s orbitals overlap to form a σ bond.

Molecular Orbital (MO) Theory and Conjugation
MO Theory Basics
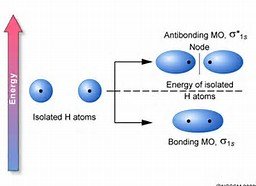
Molecular orbital theory describes bonding by combining atomic orbitals to form molecular orbitals that are delocalized over the entire molecule. The linear combination of atomic orbitals (LCAO) produces bonding and antibonding molecular orbitals.
The number of molecular orbitals formed equals the number of atomic orbitals combined.
Bonding MOs are lower in energy; antibonding MOs are higher in energy.
Conjugation and Delocalization
Conjugation occurs when three or more adjacent atoms have parallel p orbitals, allowing π electrons to be delocalized over multiple atoms. This delocalization stabilizes the molecule and is a key feature in many organic compounds (e.g., 1,3-butadiene, benzene).
Conjugated systems have alternating single and multiple bonds.
Delocalized electrons occupy molecular orbitals that extend over several atoms.
Resonance and Resonance Structures
Resonance in Conjugated Systems
When a molecule has a conjugated system, it is often possible to draw two or more valid Lewis structures, called resonance structures. The actual electronic structure is a resonance hybrid of these contributors, which stabilizes the molecule through resonance stabilization.
Resonance contributors differ only in the arrangement of electrons, not atom positions.
Curved arrows are used to show the movement of electron pairs when drawing resonance structures.
All resonance structures must obey the octet rule for second-row elements.
Rules for Resonance Contributors
Octet Principle: Structures with complete octets are more stable.
Minimize Formal Charge: Fewer formal charges increase stability.
Location of Formal Charge: Negative charges are more stable on electronegative atoms; positive charges are more stable on less electronegative atoms.
Insignificant contributors have incomplete octets or introduce unnecessary formal charges.
Recognizing and Drawing Resonance Structures
Common patterns for resonance include allylic lone pairs, allylic carbocations, and multiple bonds to heteroatoms. Curved arrows indicate the movement of electrons to generate alternative resonance contributors.
Application: Resonance in Free Radicals and Carbocations
Resonance can also stabilize free radicals and carbocations, especially when the unpaired electron or positive charge is delocalized over a conjugated system.
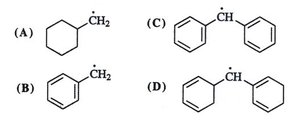

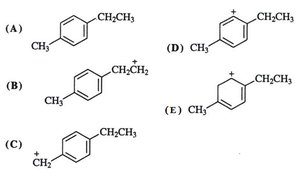
Example: The benzyl carbocation is highly stabilized by resonance because the positive charge can be delocalized over the aromatic ring.
Additional info: Mastery of resonance and hybridization is essential for understanding reactivity, stability, and structure in organic chemistry.
