 Back
BackCarboxylic Acids: Structure, Properties, and Reactions
Study Guide - Smart Notes

Carboxylic Acids
Introduction to Carboxylic Acids
Carboxylic acids are a fundamental class of organic compounds characterized by the presence of a carboxyl group (–COOH). This group consists of a carbonyl (C=O) and a hydroxyl (–OH) bonded to the same carbon atom. Carboxylic acids are widely found in nature and play essential roles in biochemistry and industrial chemistry.
Carboxyl group: The functional group is usually written as –COOH.
Aliphatic acids: Contain an alkyl group bonded to the carboxyl group.
Aromatic acids: Contain an aryl group bonded to the carboxyl group.
Fatty acids: Long-chain aliphatic acids, important in biological systems.
Nomenclature of Carboxylic Acids
Carboxylic acids are named using both common and IUPAC systems. The IUPAC system is systematic and based on the parent alkane, while common names are often derived from historical sources.
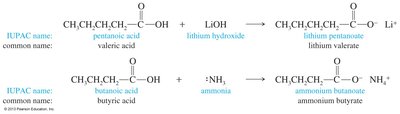
IUPAC Naming: Remove the final -e from the alkane name and add -oic acid. The carboxyl carbon is always carbon #1.
Common Names: Many aliphatic acids have traditional names. Substituent positions are labeled with Greek letters (α, β, γ, etc.), starting at the carbon adjacent to the carboxyl group.
Unsaturated Acids: Named by removing the -e from the alkene name and adding -oic acid. The position and stereochemistry (E/Z) of double bonds are specified.
Aromatic Acids: Named as derivatives of benzoic acid. Ortho-, meta-, and para- prefixes or numbers are used to indicate substituent positions.
Dicarboxylic Acids: Common names use Greek letters; IUPAC names number the chain from the end closest to a substituent.
Structure and Bonding
The carboxyl group is planar due to the sp2 hybridization of the carbonyl carbon, resulting in nearly trigonal bond angles. The O–H bond lies in the same plane, and the C–O–H angle is approximately 106°.
Bond Angles: Close to 120°, consistent with sp2 hybridization.
Bond Lengths: The C=O bond is shorter than the C–O bond due to double bond character.
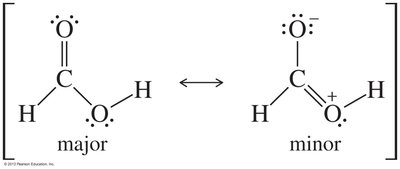
Resonance and Stability
Carboxylic acids exhibit resonance stabilization. One of the lone pairs on the hydroxyl oxygen is delocalized into the π system of the carbonyl, resulting in two resonance structures. This delocalization increases the stability of the carboxylate ion formed upon deprotonation.
Physical Properties
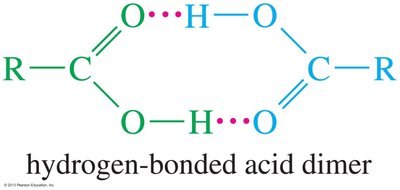
Boiling Points: Carboxylic acids have higher boiling points than alcohols, ketones, or aldehydes of similar molecular weight due to the formation of stable, hydrogen-bonded dimers.
Melting Points: Aliphatic acids with more than eight carbons are solids at room temperature. Double bonds, especially cis, lower the melting point.
Solubility: Water solubility decreases with increasing carbon chain length. Acids with more than 10 carbons are nearly insoluble in water but are soluble in alcohols and some nonpolar solvents due to hydrogen bonding.
Acidity of Carboxylic Acids
Carboxylic acids are weak acids that dissociate in water to give a proton and a carboxylate ion. The acid dissociation constant (Ka) and its logarithmic form (pKa) quantify acid strength. The carboxylate ion is stabilized by resonance, making carboxylic acids more acidic than alcohols.
Acid Dissociation:
Equilibrium Constant:
pKa:
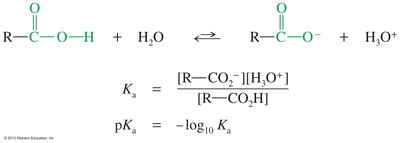
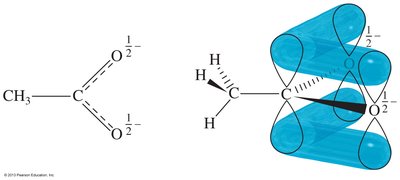
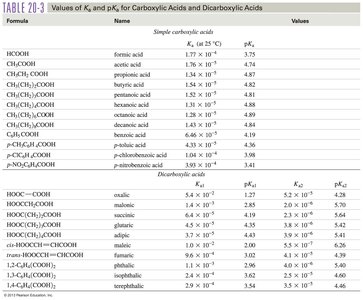
Table: pKa Values of Carboxylic Acids
Formula | Name | pKa |
|---|---|---|
HCOOH | formic acid | 3.75 |
CH3COOH | acetic acid | 4.74 |
PhCOOH | benzoic acid | 4.19 |
NO2PhCOOH | p-nitrobenzoic acid | 3.41 |
Substituent Effects on Acidity
The acidity of carboxylic acids is influenced by substituents. Electron-withdrawing groups (e.g., halogens, nitro) increase acidity by stabilizing the negative charge on the carboxylate ion, while electron-donating groups decrease acidity. The effect is stronger when the substituent is closer to the carboxyl group.
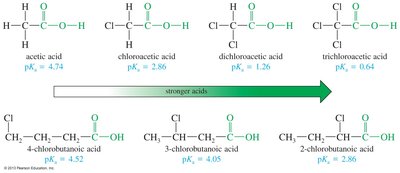
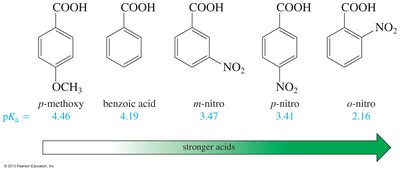
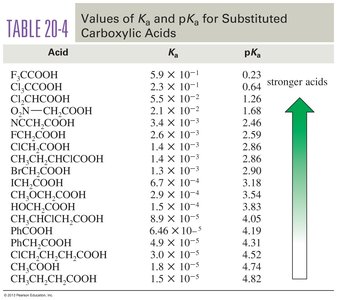
Table: pKa Values for Substituted Carboxylic Acids
Acid | pKa |
|---|---|
F3CCOOH | 0.23 |
Cl3CCOOH | 0.64 |
PhCOOH | 4.19 |
CH3CH2COOH | 4.82 |
Reactions of Carboxylic Acids
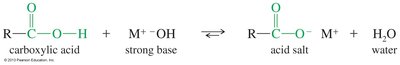
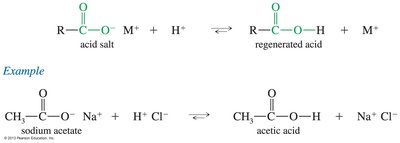
Deprotonation and Salt Formation
Carboxylic acids react with strong bases to form carboxylate salts, which are usually solids and often water-soluble. The reaction is reversible; adding a strong acid regenerates the carboxylic acid.
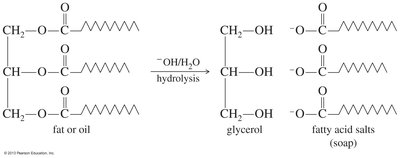
Saponification
The basic hydrolysis of fats and oils (triglycerides) produces soap and glycerol. This process is known as saponification and is an important industrial method for soap production.
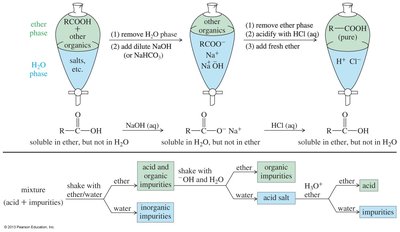
Extraction and Purification
Carboxylic acids can be separated from mixtures using acid-base extraction techniques. The acid is more soluble in organic solvents, while its salt is more soluble in water, allowing for efficient purification.
Spectroscopic Properties
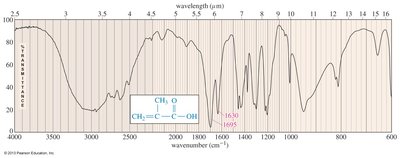
Infrared (IR) Spectroscopy
Carboxylic acids show two characteristic IR absorptions: a strong C=O stretch near 1710 cm–1 and a broad O–H stretch between 2500–3500 cm–1. Conjugation lowers the C=O stretching frequency.
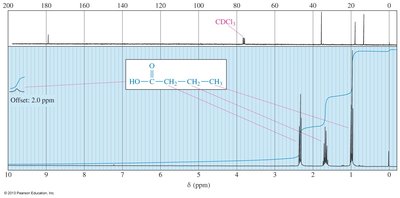
Nuclear Magnetic Resonance (NMR) Spectroscopy
Carboxylic acid protons are highly deshielded, appearing between δ 10 and δ 13 in 1H NMR spectra. Protons on the α-carbon absorb between δ 2.0 and δ 2.5.
Synthesis of Carboxylic Acids
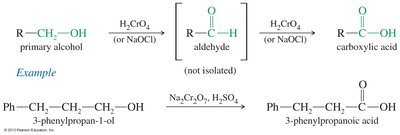
Oxidation of Primary Alcohols and Aldehydes: Chromic acid (H2CrO4) or potassium permanganate (KMnO4) oxidizes primary alcohols and aldehydes to carboxylic acids.
Cleavage of Alkenes: Hot, concentrated KMnO4 cleaves alkenes to give acids if a vinylic hydrogen is present.
Ozonolysis of Alkynes: Ozone or KMnO4 cleaves alkynes to carboxylic acids.
Side Chain Oxidation of Alkylbenzenes: Hot KMnO4 or chromic acid oxidizes alkyl side chains to benzoic acid.
Carboxylation of Grignard Reagents: Grignard reagents react with CO2 to form carboxylic acids after protonation, increasing the carbon count by one.
Hydrolysis of Nitriles: Acidic or basic hydrolysis of nitriles yields carboxylic acids, also increasing the carbon count by one.
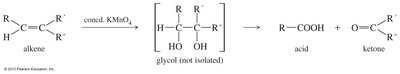
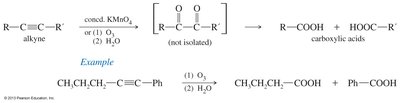
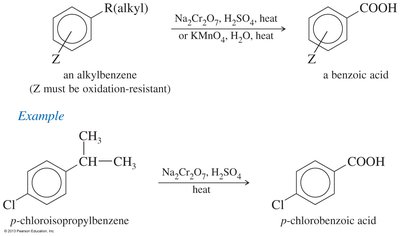
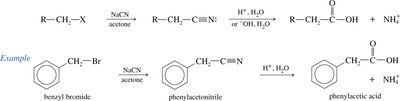
Reactions of Carboxylic Acids: Derivatives and Transformations
Acid Derivatives and Nucleophilic Acyl Substitution
The group bonded to the acyl carbon determines the class of acid derivative: –OH (acid), –Cl (acid chloride), –OR' (ester), –NH2 (amide). These derivatives interconvert via nucleophilic acyl substitution, where one nucleophile replaces another on the acyl carbon.
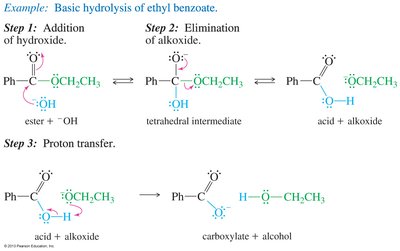
Hydrolysis of Esters (Saponification)
Esters can be hydrolyzed under basic conditions to yield carboxylate salts and alcohols. This is a key reaction in soap production.
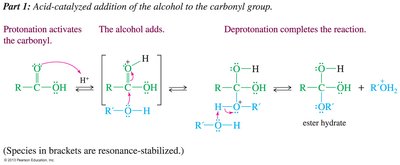
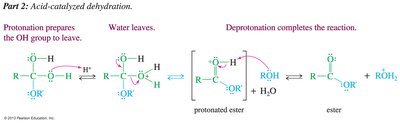
Fischer Esterification
Carboxylic acids react with alcohols under acidic conditions to form esters. The reaction is reversible and driven to completion by using excess alcohol or removing water.
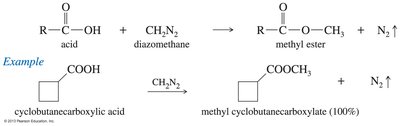
Esterification Using Diazomethane
Diazomethane converts carboxylic acids to methyl esters in high yield. The reaction is efficient but diazomethane is toxic and explosive.
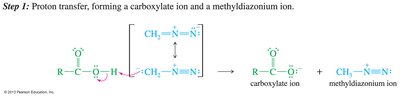

Synthesis of Amides
Carboxylic acids react with amines to form ammonium carboxylate salts, which upon heating yield amides and water.
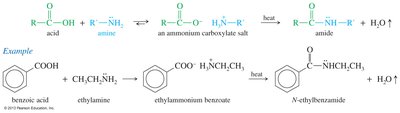
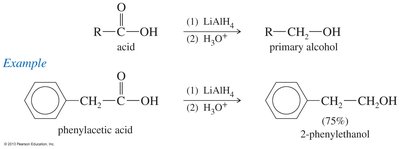
Reduction of Carboxylic Acids
Carboxylic acids can be reduced to primary alcohols using lithium aluminum hydride (LiAlH4) or borane (BH3).
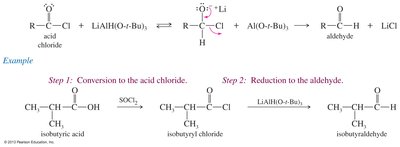
Reduction of Acid Chlorides to Aldehydes
Lithium aluminum tri(tert-butoxy)hydride is a selective reducing agent that converts acid chlorides to aldehydes without further reduction to alcohols.
Conversion to Ketones
Carboxylic acids react with two equivalents of an organolithium reagent to form ketones after hydrolysis.
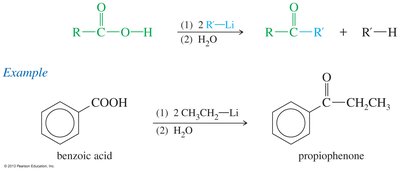
Synthesis of Acid Chlorides
Carboxylic acids are converted to acid chlorides using thionyl chloride (SOCl2) or oxalyl chloride (COCl2), producing gaseous by-products for easy purification.
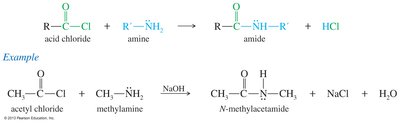
Amide Synthesis from Acid Chlorides
Acid chlorides react with ammonia or amines to form amides. A base such as NaOH, pyridine, or a second equivalent of amine is used to neutralize the HCl formed.
Additional info: This guide covers the structure, nomenclature, properties, acidity, synthesis, and reactions of carboxylic acids, as well as their derivatives. It is suitable for exam preparation in a college-level organic chemistry course.
