 Back
BackChapter 1 Study Notes: Electronic Structure and Bonding in Organic Chemistry
Study Guide - Smart Notes

Remembering General Chemistry: Electronic Structure and Bonding
What Is Organic Chemistry?
Organic chemistry is the study of compounds primarily based on carbon. Historically, organic compounds were thought to originate only from living organisms, while inorganic compounds came from minerals. Modern definitions focus on the presence of carbon as the central element in organic compounds.
Early definition: Organic compounds were believed to possess a 'vital force' from living organisms.
Current definition: Organic compounds are carbon-based, regardless of their origin.
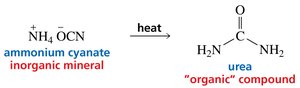
Example: The synthesis of urea from ammonium cyanate demonstrated that organic compounds can be formed from inorganic sources.
What Makes Carbon So Special?
Carbon is unique in its ability to form stable covalent bonds by sharing electrons. Unlike elements to its left (which lose electrons) or right (which gain electrons), carbon achieves stability through electron sharing, allowing for a vast diversity of organic molecules.
The Structure of an Atom
Atoms consist of a nucleus (protons and neutrons) surrounded by an electron cloud. The atomic number indicates the number of protons, which equals the number of electrons in a neutral atom.
Protons: Positively charged particles in the nucleus.
Neutrons: Neutral particles in the nucleus.
Electrons: Negatively charged particles in orbitals around the nucleus.
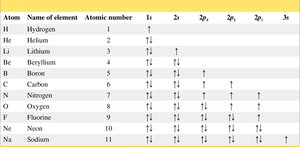
Example: Carbon has atomic number 6, so it has 6 protons and 6 electrons.
The Distribution of Electrons in an Atom
Electrons occupy shells around the nucleus, with the first shell being closest and lowest in energy. The arrangement of electrons follows specific principles:
Aufbau principle: Electrons fill the lowest energy orbitals first.
Pauli exclusion principle: Each orbital holds a maximum of two electrons with opposite spins.
Hund’s rule: Electrons fill degenerate orbitals singly before pairing.
Electron Shells and Stability
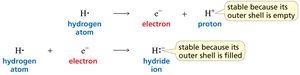
Atoms achieve stability by having a filled outer shell. Hydrogen can lose its electron to become a proton (empty shell) or gain an electron to become a hydride ion (filled shell).
Covalent Bonding: Sharing Electrons
Atoms can achieve filled outer shells by sharing electrons, forming covalent bonds. This is the basis for the structure of most organic molecules.
Example: Two fluorine atoms share electrons to form F2; two hydrogen atoms share electrons to form H2.
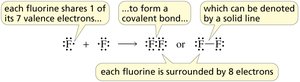
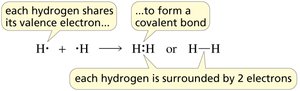
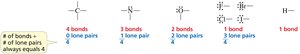
How Many Bonds Does an Atom Form?
The number of covalent bonds an atom forms depends on its valence electrons and the need to achieve a stable configuration:
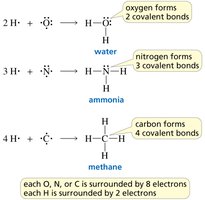
Carbon: Forms 4 bonds.
Nitrogen: Forms 3 bonds and has 1 lone pair.
Oxygen: Forms 2 bonds and has 2 lone pairs.
Halogens: Form 1 bond and have 3 lone pairs.
Hydrogen: Forms 1 bond.
Nonpolar and Polar Covalent Bonds
Covalent bonds can be nonpolar (equal sharing of electrons) or polar (unequal sharing due to differences in electronegativity). The direction of bond polarity is indicated by an arrow pointing toward the more electronegative atom.
Nonpolar covalent bond: Atoms have similar electronegativities (e.g., C–C, H–H).
Polar covalent bond: Atoms have different electronegativities (e.g., H–Cl, O–H).
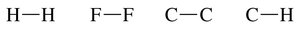
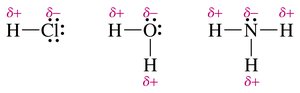

Electronegativity and Bond Polarity
The greater the difference in electronegativity between two atoms, the more polar the bond. If the difference is very large, the bond becomes ionic.
Dipole moment: A measure of bond polarity; increases with electronegativity difference.

Electrostatic Potential Maps
Electrostatic potential maps visually represent the distribution of electron density in molecules, highlighting regions of positive and negative charge.


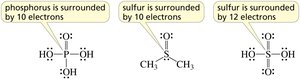
Lewis Structures and Formal Charge
Lewis structures depict the bonding between atoms and the arrangement of lone pairs. Formal charge is calculated to determine the most stable structure.
Steps to draw Lewis structures: Count valence electrons, arrange atoms, assign bonds and lone pairs, check for formal charges.
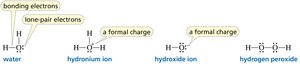
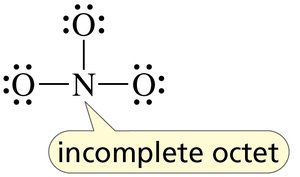
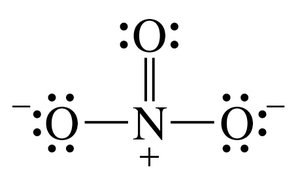
Carbon, Nitrogen, Oxygen, and Halogen Bonding Patterns
Each element has a characteristic bonding pattern in organic molecules:
Carbon: 4 bonds
Nitrogen: 3 bonds, 1 lone pair
Oxygen: 2 bonds, 2 lone pairs
Halogens: 1 bond, 3 lone pairs
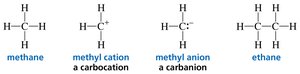
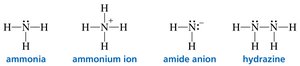
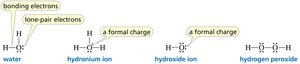

Bonding and Lone Pairs: The Rule of Four
The sum of the number of bonds and lone pairs around C, N, O, and F is always four, reflecting the octet rule.
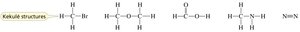
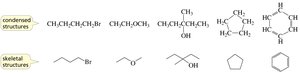
Structural Representations in Organic Chemistry
Organic compounds can be represented in several ways:
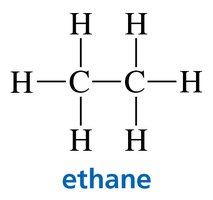
Kekulé structures: Show all bonds, omit lone pairs.
Condensed structures: Omit some or all bonds, show atom connectivity.
Skeletal structures: Show only carbon–carbon bonds as lines; carbons and hydrogens are implied.

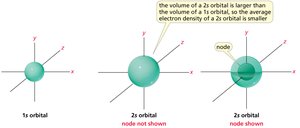
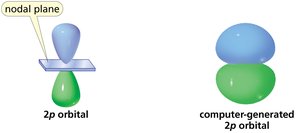
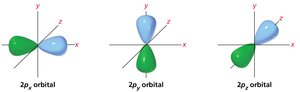
Atomic Orbitals: s and p Orbitals
Atomic orbitals are regions where electrons are most likely to be found. The s orbital is spherical, while p orbitals have two lobes with opposite phases.
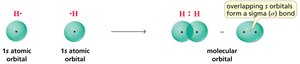
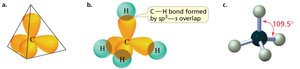
Sigma and Pi Bonds
Sigma (σ) bonds are formed by the head-on overlap of orbitals, while pi (π) bonds result from the side-to-side overlap of p orbitals. Sigma bonds are stronger and allow free rotation, while pi bonds restrict rotation.
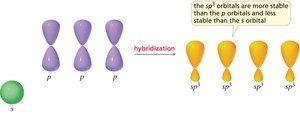
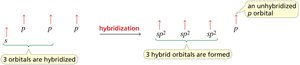
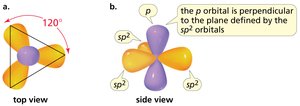
Hybridization and Molecular Geometry
Hybridization is the mixing of atomic orbitals to form new hybrid orbitals suitable for bonding. The type of hybridization determines molecular geometry and bond angles.
sp3 hybridization: Tetrahedral geometry, 109.5° bond angles (e.g., methane).
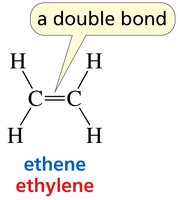
sp2 hybridization: Trigonal planar geometry, 120° bond angles (e.g., ethene).
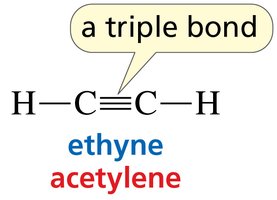
sp hybridization: Linear geometry, 180° bond angles (e.g., ethyne).

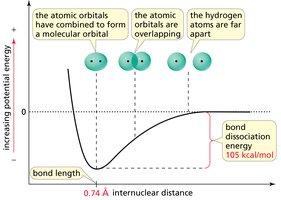
Bond Strength, Length, and Order
The strength and length of a bond depend on the number of shared electron pairs (bond order) and the type of orbitals involved. More bonds (higher bond order) mean shorter and stronger bonds.
Single bond: One shared pair (σ bond).
Double bond: Two shared pairs (one σ and one π bond).
Triple bond: Three shared pairs (one σ and two π bonds).
Summary of Key Concepts
The shorter the bond, the stronger it is.
The greater the electron density in the region of orbital overlap, the stronger the bond.
The more s character in a hybrid orbital, the shorter and stronger the bond, and the larger the bond angle.
Learning Objectives
Write ground-state electronic configurations for elements hydrogen through calcium.
Describe the relative polarity of bonds and determine the direction of dipoles.
Represent organic compounds using Lewis, Kekulé, condensed, and skeletal structures.
Assign lone pairs and calculate formal charges.
Determine the hybridization of carbon, oxygen, or nitrogen atoms from molecular formulas.
Describe how molecular geometry is determined by hybridization.
Explain how hybridization affects bond strength and length.
Describe how bond order affects bond length and strength.
