 Back
BackChapter 3: An Introduction to Organic Compounds – Nomenclature, Physical Properties, and Rotation About Single Bonds
Study Guide - Smart Notes

An Introduction to Organic Compounds
Overview of Organic Compounds
Organic compounds are primarily composed of carbon and hydrogen, often with other elements such as oxygen, nitrogen, and halogens. The study of their structure, nomenclature, and properties forms the foundation of organic chemistry.
Hydrocarbons: Compounds containing only carbon and hydrogen.
Alkanes: Hydrocarbons with only single bonds between carbon atoms.
Functional Groups: Specific groups of atoms within molecules that determine the chemical properties of those molecules.
Nomenclature of Alkanes
Straight-Chain Alkanes and Homologs
Alkanes are named according to the number of carbon atoms in the longest continuous chain. Homologs are compounds that differ by a single CH2 group.
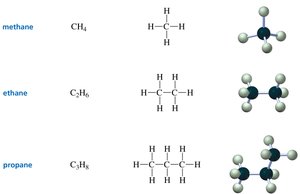
Methane, Ethane, Propane: Each has only one possible structure.
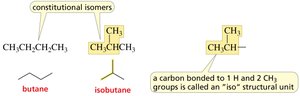
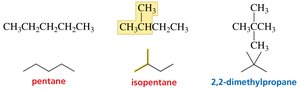
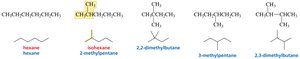
Butane, Pentane, Hexane: Can be arranged in multiple ways, leading to constitutional isomers.
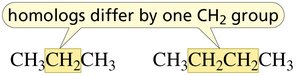
Constitutional Isomers
Constitutional isomers have the same molecular formula but different connectivity of atoms.
Butane and Isobutane: Example of constitutional isomers.
Iso structural unit: A carbon bonded to one hydrogen and two methyl groups.
Naming Alkyl Substituents
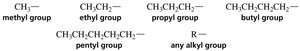
Alkyl substituents are formed by removing a hydrogen from an alkane. The suffix "-yl" replaces "-ane" in the parent alkane name.
Methyl, Ethyl, Propyl, Butyl, Pentyl: Common alkyl groups.
Common Names and Systematic Names
Organic compounds often have both common and systematic (IUPAC) names. Systematic names follow strict rules and include numbers to indicate the position of substituents.
Common names: Often used for simple compounds; do not include numbers.
Systematic names: Include numbers and follow IUPAC rules.

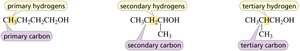
Classification of Carbons and Hydrogens
Primary, Secondary, and Tertiary Carbons
Carbons are classified based on the number of other carbons to which they are bonded.
Primary carbon: Bonded to one other carbon.
Secondary carbon: Bonded to two other carbons.
Tertiary carbon: Bonded to three other carbons.

Primary, Secondary, and Tertiary Hydrogens
Hydrogens are classified based on the type of carbon to which they are attached.
Primary hydrogen: Attached to a primary carbon.
Secondary hydrogen: Attached to a secondary carbon.
Tertiary hydrogen: Attached to a tertiary carbon.
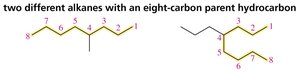
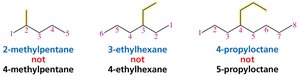
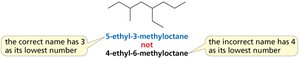
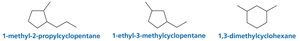
Naming Alkanes with Substituents
Rules for Naming Alkanes
To name an alkane with substituents:
Identify the longest continuous chain (parent hydrocarbon).
Number the chain to give the substituent the lowest possible number.
List substituents in alphabetical order, each preceded by a number.
Use prefixes (di-, tri-, tetra-) for multiple identical substituents.
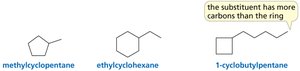
Cycloalkanes
Structure and Nomenclature of Cycloalkanes
Cycloalkanes are alkanes in which carbon atoms form a ring. The ring is the parent hydrocarbon unless a substituent has more carbons than the ring.
Monosubstituted cycloalkanes: No number needed.
Disubstituted cycloalkanes: Substituents are stated in alphabetical order; #1 goes to the first-listed substituent.

Classification and Nomenclature of Alkyl Halides, Ethers, Alcohols, and Amines
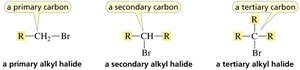
Alkyl Halides
Alkyl halides are classified based on the carbon to which the halogen is attached.
Primary alkyl halide: Halogen on a primary carbon.
Secondary alkyl halide: Halogen on a secondary carbon.
Tertiary alkyl halide: Halogen on a tertiary carbon.
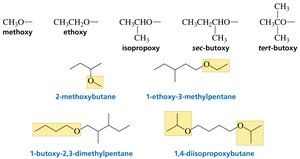
Ethers
Ethers are compounds with an oxygen atom connected to two alkyl or aryl groups. They can be symmetrical or unsymmetrical.
Common names: List substituents in alphabetical order.
Systematic names: Replace "yl" with "oxy" for the RO substituent.

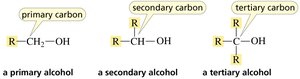
Alcohols
Alcohols are classified based on the carbon to which the hydroxyl group (OH) is attached.
Primary alcohol: OH attached to a primary carbon.
Secondary alcohol: OH attached to a secondary carbon.
Tertiary alcohol: OH attached to a tertiary carbon.
Amines
Amines are classified by the number of alkyl groups attached to the nitrogen atom.
Primary amine: One R group bonded to N.
Secondary amine: Two R groups bonded to N.
Tertiary amine: Three R groups bonded to N.
Physical Properties of Organic Compounds
Boiling Points
The boiling point of organic compounds depends on intermolecular forces and molecular structure.
Greater attractive forces (e.g., hydrogen bonds, dipole-dipole interactions) increase boiling point.
Greater surface area increases boiling point.
Branching lowers boiling point.
Intermolecular Forces
Organic compounds exhibit various intermolecular forces:
London dispersion forces: Present in all molecules, especially nonpolar ones.
Dipole-dipole interactions: Stronger than dispersion forces; present in polar molecules.
Hydrogen bonds: Strongest intermolecular force; present when H is bonded to N, O, or F.
Solubility
Solubility is governed by the principle "like dissolves like." Polar compounds dissolve in polar solvents, and nonpolar compounds dissolve in nonpolar solvents.
Polar compounds: Dissolve in water.
Nonpolar compounds: Dissolve in hexane.
Rotation About Single Bonds and Conformers
Rotation and Conformers
Rotation occurs about carbon–carbon single bonds, leading to different conformers. The staggered conformer is more stable than the eclipsed conformer due to reduced steric strain and hyperconjugation.
Staggered conformer: More stable, lower energy.
Eclipsed conformer: Less stable, higher energy.
Hyperconjugation: Stabilizing interaction between bonding and antibonding orbitals.
Cyclohexane Conformations
Cyclohexane adopts a chair conformation to minimize strain. Substituents prefer the equatorial position to reduce 1,3-diaxial interactions.
Chair conformer: Completely free of strain.
Axial and equatorial bonds: Substituents are more stable in equatorial positions.
Ring flip: Interconversion between chair conformers.
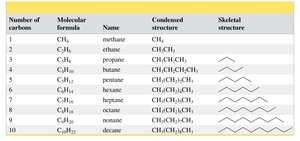
Summary Table: Straight-Chain Alkanes
Number of Carbons | Molecular Formula | Name | Condensed Structure | Skeletal Structure |
|---|---|---|---|---|
1 | CH4 | methane | CH4 | • |
2 | C2H6 | ethane | CH3CH3 | — |
3 | C3H8 | propane | CH3CH2CH3 | — — |
4 | C4H10 | butane | CH3CH2CH2CH3 | — — — |
5 | C5H12 | pentane | CH3(CH2)3CH3 | — — — — |
6 | C6H14 | hexane | CH3(CH2)4CH3 | — — — — — |
7 | C7H16 | heptane | CH3(CH2)5CH3 | — — — — — — |
8 | C8H18 | octane | CH3(CH2)6CH3 | — — — — — — — |
9 | C9H20 | nonane | CH3(CH2)7CH3 | — — — — — — — — |
10 | C10H22 | decane | CH3(CH2)8CH3 | — — — — — — — — — |

Learning Objectives
3.1 Name alkanes, cycloalkanes, alkyl halides, ethers, alcohols, and amines using both systematic (IUPAC) nomenclature and common nomenclature.
3.2 Draw condensed and skeletal structures of alkanes, alkyl halides, ethers, alcohols, and amines from their systematic and/or common name.
3.3 Distinguish primary, secondary, and tertiary carbons and hydrogens.
3.4 Draw all possible constitutional isomers for a given molecular formula.
3.5 Predict relative physical properties of compounds based on their structure, polarity, and ability to form hydrogen bonds.
3.6 Draw Newman projections for rotation about a given bond and predict which projection is more stable.
3.7 Draw chair conformers of a substituted cyclohexane and predict which conformer is more stable.
3.8 Draw chair conformers for the geometric isomers of disubstituted cyclohexanes and predict their relative stabilities.
