 Back
BackChapter 5: Alkenes – Introduction to Reactivity and Stability
Study Guide - Smart Notes

Alkenes: Structure and Reactivity
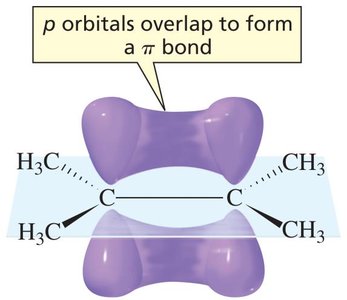
Structure of Alkenes and π Bonding
Alkenes are hydrocarbons containing at least one carbon–carbon double bond. The double bond consists of one sigma (σ) bond and one pi (π) bond. The π bond is formed by the sideways overlap of unhybridized p orbitals on adjacent sp2-hybridized carbons, resulting in electron density above and below the plane of the molecule. This makes alkenes electron-rich and reactive toward electrophiles.
sp2 Hybridization: Each carbon in the double bond is trigonal planar, with 120° bond angles.
π Bond: The π bond is weaker than the σ bond and is the site of most alkene reactivity.
Reactivity of Alkenes: Addition Reactions
General Features of Addition Reactions
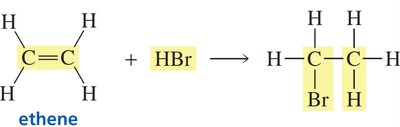
The most characteristic reactions of alkenes are addition reactions, where atoms or groups add across the double bond. The π bond is broken, and two new σ bonds are formed. These reactions are typically exothermic and occur readily at room temperature.
Example: Addition of hydrogen bromide (HBr) to ethene forms bromoethane.
Electrophilic Addition Mechanism
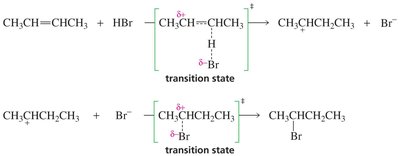
The addition of HBr to an alkene proceeds via a two-step electrophilic addition mechanism:
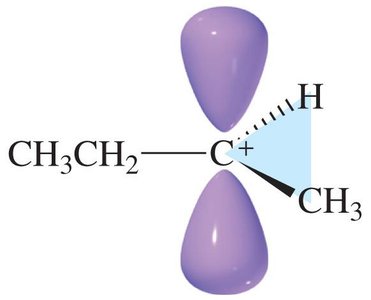
Step 1: Electrophilic Attack (Rate-Determining Step) The π electrons attack the electrophile (H+ from HBr), breaking the π bond and forming a carbocation intermediate. This step is endothermic and slow.
Step 2: Nucleophilic Attack The nucleophile (Br–) attacks the carbocation, forming a new σ bond. This step is fast and exothermic.
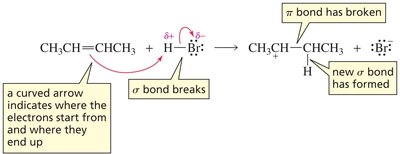
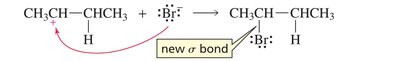
Carbocation Intermediate: The intermediate is a high-energy, positively charged species.
Transition States and Reaction Coordinate
The reaction proceeds through two transition states, corresponding to the two steps of the mechanism. The first transition state (formation of the carbocation) has the highest activation energy and determines the rate of the reaction.
Transition State: Bonds are partially formed and broken.
Rate-Determining Step: The step with the highest energy barrier (formation of the carbocation).
Catalytic Hydrogenation and Alkene Stability
Catalytic Hydrogenation
Catalytic hydrogenation is the addition of hydrogen (H2) across the double bond of an alkene in the presence of a metal catalyst (such as Pd/C). This reaction converts alkenes to alkanes and is exothermic.
Mechanism: The reaction occurs on the surface of the metal catalyst, where both the alkene and hydrogen are adsorbed and react.
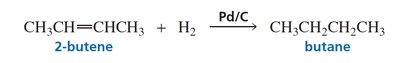
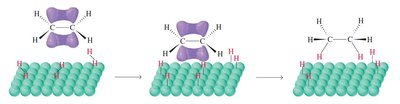
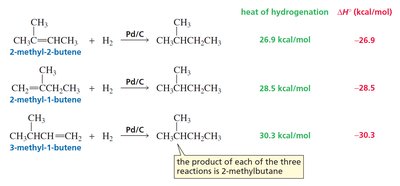
Heats of Hydrogenation and Alkene Stability
The heat of hydrogenation (ΔHo) measures the energy released when an alkene is hydrogenated. Lower heats of hydrogenation indicate greater alkene stability. The stability of alkenes increases with the number of alkyl substituents on the double bond due to hyperconjugation and inductive effects.
More substituted alkenes (more alkyl groups attached to the double bond) are more stable.
Heats of hydrogenation: Used to compare the relative stabilities of different alkenes.
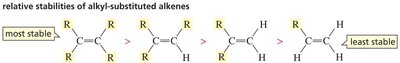
Alkyl Substitution and Stability
The stability of alkenes increases with the number of alkyl groups attached to the double-bonded carbons. This is due to the electron-donating effect of alkyl groups, which stabilizes the π bond.
Tetrasubstituted alkenes are the most stable, while monosubstituted are the least stable.
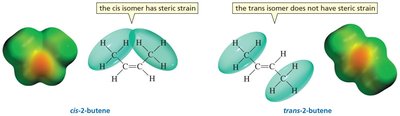
Cis-Trans Isomerism and Stability
Alkenes with the same number of alkyl groups can exist as cis (Z) and trans (E) isomers. Trans isomers are generally more stable than cis isomers due to reduced steric strain between substituents.
Trans isomers: Substituents are on opposite sides of the double bond, minimizing steric interactions.
Cis isomers: Substituents are on the same side, leading to increased steric strain and lower stability.

Summary Table: Alkene Stability
Alkene Type | Number of Alkyl Groups | Relative Stability | Heat of Hydrogenation (kcal/mol) |
|---|---|---|---|
Tetrasubstituted | 4 | Most stable | Lowest |
Trisubstituted | 3 | More stable | Lower |
Disubstituted (trans) | 2 | Stable | Lower |
Disubstituted (cis) | 2 | Less stable (steric strain) | Higher |
Monosubstituted | 1 | Least stable | Highest |
Example: Ranking Alkene Stability
Given the compounds: trans-3-hexene, cis-3-hexene, cis-2,5-dimethyl-3-hexene, and cis-3,4-dimethyl-3-hexene, rank them in order of decreasing stability:
cis-3,4-dimethyl-3-hexene (most substituted)
cis-2,5-dimethyl-3-hexene
trans-3-hexene
cis-3-hexene (least stable due to steric strain)
Key Takeaways:
Alkenes are electron-rich and undergo addition reactions, especially with electrophiles.
Stability increases with alkyl substitution and is greater for trans isomers than cis isomers.
Heats of hydrogenation provide a quantitative measure of alkene stability.
