 Back
BackChapter 6: An Overview of Organic Reactions – Mechanisms, Radicals, and Energetics
Study Guide - Smart Notes

Organic Reaction Mechanisms
Electrophiles and Their Charges
Organic reactions often involve the interaction between an electrophile (electron-deficient species) and a nucleophile (electron-rich species). The electrophile can be either positively charged or neutral. When a positively charged electrophile accepts an electron pair, the atom bearing the charge becomes neutral.
Electrophile: A species that accepts an electron pair.
Nucleophile: A species that donates an electron pair.
Example: Addition of H2O+ to an alkene, where the oxygen becomes neutral after accepting electrons.
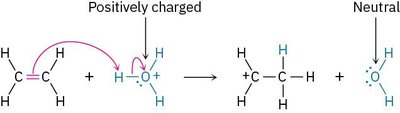
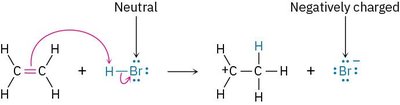
The Octet Rule in Organic Reactions
The octet rule states that second-row elements (such as carbon, nitrogen, and oxygen) cannot have more than eight electrons in their valence shell. If an electron pair moves to an atom that already has an octet, another electron pair must simultaneously move from that atom to maintain the octet.
Octet Rule: No second-row atom can be left with ten electrons.
Example: In the addition of H2O+ to an alkene, the electron pair in the H–O bond must leave when another pair moves to hydrogen.
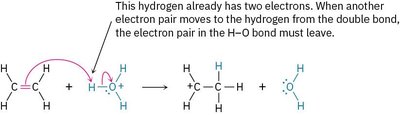
Radical Reactions
Definition and Properties of Radicals
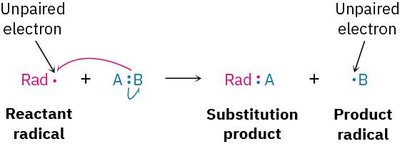
A radical is a highly reactive species containing an atom with an odd number of electrons (usually seven) in its valence shell, rather than a noble-gas octet. Radicals are important intermediates in many organic reactions, including industrial processes.
Radical: An atom or molecule with an unpaired electron.
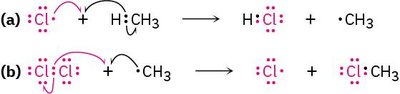
Example: Chlorine reacts with methane to yield chloromethane, a process used in solvent manufacturing.
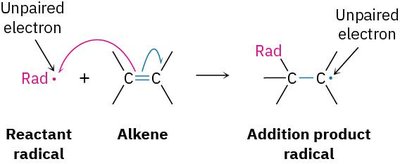
Types of Radical Reactions
Substitution: A reactant radical replaces an atom in another molecule, forming a new radical.
Addition: A reactant radical adds to a double bond, forming a new radical product.
Industrial Example: Chlorination of Methane
The reaction of chlorine with methane produces chloromethane, which is used to manufacture dichloromethane and chloroform. The mechanism involves radical intermediates.
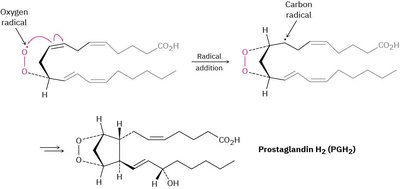
Biological Example: Radical Reactions in Arachidonic Acid and Prostaglandin H2
Radical reactions are also important in biological systems, such as the conversion of arachidonic acid to prostaglandin H2 (PGH2), a key step in inflammation and signaling pathways.
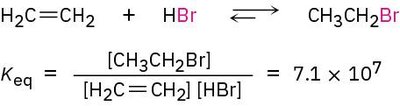
Describing a Reaction: Equilibria, Rates, and Energy Changes
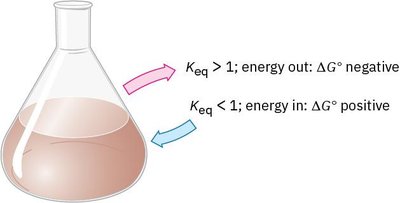
Chemical Equilibrium and the Equilibrium Constant (Keq)
A chemical equilibrium is reached when the concentrations of reactants and products remain constant over time. The equilibrium constant, Keq, is defined as:
Formula:
Interpretation: If Keq is near 1, both reactants and products are present in appreciable amounts. If Keq > 103, reactants are nearly consumed.
Energy Changes and Gibbs Free Energy (ΔG)
The Gibbs free-energy change (ΔG) determines whether a reaction is energetically favorable. It is calculated as:
Formula:
Exergonic Reaction: ΔG is negative; energy is released.
Endergonic Reaction: ΔG is positive; energy is absorbed.

Relationship Between ΔG° and Keq
The standard Gibbs free-energy change (ΔG°) is related to the equilibrium constant by:
Formula:
R: Gas constant (8.314 J/(K·mol) or 1.987 cal/(K·mol))
T: Temperature in Kelvin
Interpretation: If ΔG° is negative, Keq > 1 and the reaction favors products.
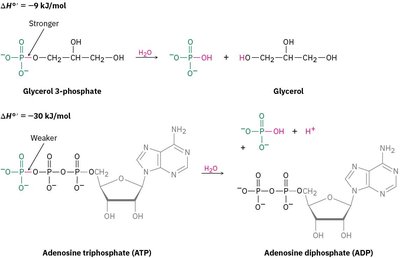
Bond Dissociation Energies
The bond dissociation energy (D) is the energy required to break a specific bond in a molecule, producing two radical fragments. It is a measure of bond strength and is typically reported for the gas phase at 25°C.
Example: The C–H bond in methane has D = 439.3 kJ/mol (105.0 kcal/mol).
Stronger bonds: Require more energy to break.
Weaker bonds: Require less energy to break.
Summary Table: Key Concepts in Organic Reaction Mechanisms
Concept | Definition | Example |
|---|---|---|
Electrophile | Electron pair acceptor | H2O+ in alkene addition |
Nucleophile | Electron pair donor | Alkene in addition reaction |
Radical | Species with unpaired electron | Cl· in chlorination of methane |
Bond Dissociation Energy | Energy to break a bond | C–H in methane: 439.3 kJ/mol |
Equilibrium Constant (Keq) | Ratio of product/reactant concentrations | Alkene + HBr ⇌ Alkyl bromide |
Gibbs Free Energy (ΔG) | Energy change in reaction | ΔG = Gproducts − Greactants |
Additional info: Academic context was added to clarify definitions, mechanisms, and energetics for self-contained study notes.
