 Back
BackChemical Quantities and Reactions: Foundations for Organic Chemistry
Study Guide - Smart Notes

Chemical Reactions and Chemical Equations
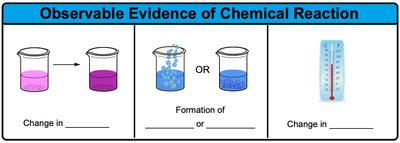
Definition and Evidence of Chemical Reactions
Chemical reactions are processes in which chemical bonds in reactants are broken and new bonds are formed in products, resulting in a chemical change. Observable evidence of a chemical reaction includes changes in color, formation of a precipitate or gas, and changes in temperature.
Chemical Equation: A symbolic representation using chemical formulas and symbols to describe a chemical reaction.
Example: The decomposition of sodium carbonate at high temperature produces sodium oxide and carbon dioxide gas.
Balancing Chemical Equations
Principles and Steps
Balancing chemical equations ensures the conservation of mass, meaning the number and type of atoms are equal on both sides of the equation. The numbers in front of compounds are called coefficients.
Step 1: List elements in reactants and products.
Step 2: Count the number of each element on both sides.
Step 3: Balance each element, adjusting coefficients as needed.
If a polyatomic ion appears unchanged on both sides, treat it as a single unit.
If fractional coefficients are used, multiply the entire equation to clear fractions.
Types of Chemical Reactions
Classification and Examples
Chemical reactions are classified based on the rearrangement of atoms and the types of products formed:
Combination: Multiple reactants form one product.
Decomposition: One reactant splits into multiple products.
Combustion: A hydrocarbon reacts with O2 to produce CO2 and H2O.
Single Displacement: One element replaces another in a compound.
Double Displacement: Ions from two compounds exchange places.
Redox Reactions: Involve the transfer of electrons between reactants.
Oxidation Numbers and Redox Reactions
Assigning Oxidation Numbers
Oxidation numbers indicate the degree of oxidation (loss of electrons) or reduction (gain of electrons) of an atom in a compound. Rules are used to assign oxidation numbers, especially when elements are in compounds.
For an atom in its natural state, the oxidation number is zero.
For a monoatomic ion, the oxidation number equals its charge.
In compounds, use known oxidation numbers to solve for unknowns.
Redox Reactions: Oxidation and Reduction
Redox (oxidation-reduction) reactions involve the transfer of electrons. The substance that loses electrons is oxidized (reducing agent), and the substance that gains electrons is reduced (oxidizing agent).
LEO the lion says GER: Lose Electrons = Oxidation; Gain Electrons = Reduction.

Mole Concept and Molar Mass
Definition and Calculations
The mole is a counting unit that connects mass, number of particles, and chemical reactions. One mole contains Avogadro's number (6.022 x 1023) of particles (atoms, molecules, ions, or formula units).
Molar Mass: The mass of one mole of a substance, calculated by summing the atomic masses of all atoms in a formula.
Formula:
Stoichiometry
Quantitative Relationships in Reactions
Stoichiometry involves using balanced chemical equations to calculate the amounts of reactants and products. The process includes converting between mass, moles, and number of particles using molar mass and Avogadro's number.
Map out the conversion path (mass ↔ moles ↔ particles).
Use coefficients from the balanced equation for mole-to-mole conversions.
Thermochemistry: Endothermic and Exothermic Reactions
Energy Changes in Chemical Reactions
Chemical reactions can absorb or release energy, usually in the form of heat.
Endothermic Reactions: Absorb heat from the surroundings; products have higher energy than reactants.
Exothermic Reactions: Release heat to the surroundings; products have lower energy than reactants.


Kinetics: Rate of Reaction
Factors Affecting Reaction Rate
The rate of a chemical reaction is influenced by several factors:
Concentration: Higher concentration increases collision frequency.
Surface Area: Greater surface area increases reaction rate.
Temperature: Higher temperature increases kinetic energy and collision frequency.
Catalyst: Lowers the activation energy, increasing the reaction rate.
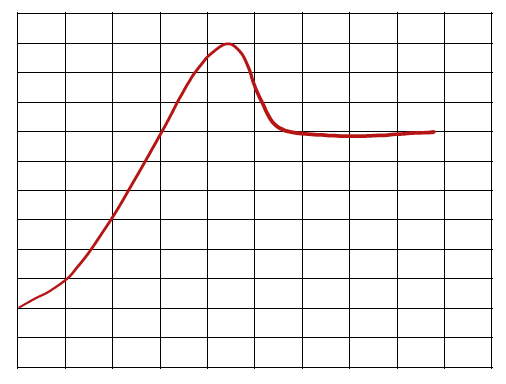
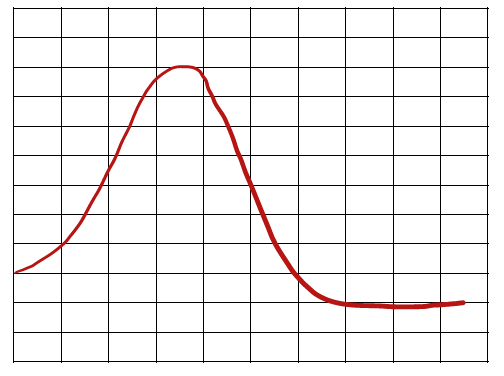
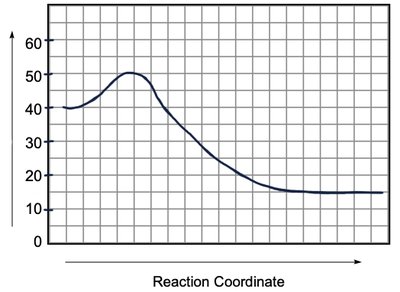
Energy Diagrams
Understanding Reaction Pathways
Energy diagrams plot the energy changes during a reaction, showing reactants, products, and the transition state (highest energy point). The difference in energy between reactants and products determines if the reaction is exothermic or endothermic.
Activation Energy (Ea): The minimum energy required for a reaction to occur.
Overall Energy Change (ΔH or ΔG): The difference in energy between products and reactants.
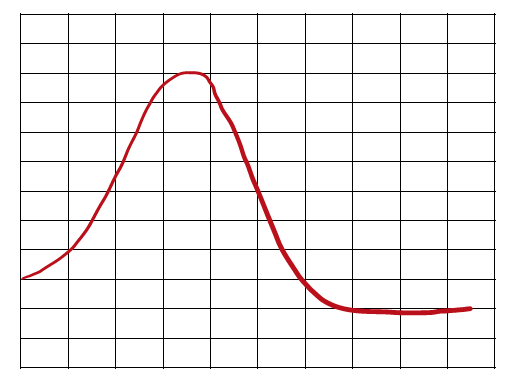
Speed of Reactions and Stability
The speed of a reaction depends on the activation energy. Lower activation energy means a faster reaction. The overall energy difference (ΔH or ΔG) indicates the thermodynamic favorability of the reaction.
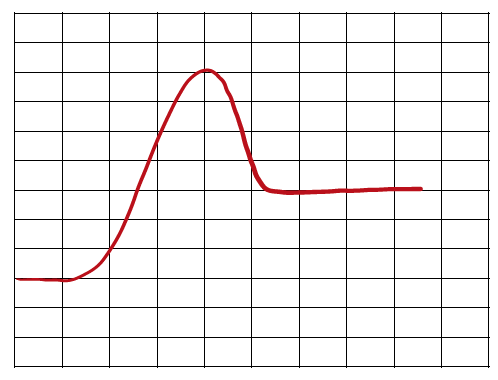
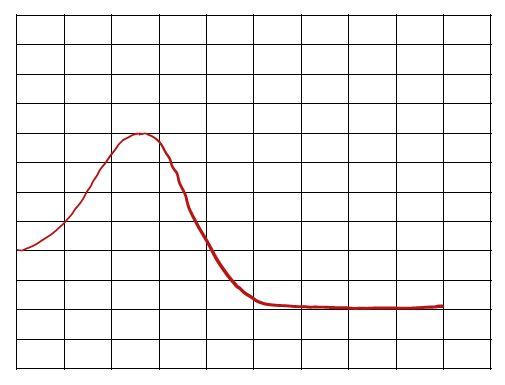
Comparing Reaction Pathways
Multiple energy diagrams can be compared to determine which reaction is fastest (lowest activation energy) or most thermodynamically favorable (largest negative ΔH or ΔG).
Additional info: These foundational concepts are essential for understanding organic chemistry, as they underpin reaction mechanisms, energy changes, and quantitative analysis in organic transformations.
