 Back
BackChemistry of Ethers, Epoxides, and Sulfides: Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Chemistry of Ethers, Epoxides, and Sulfides
Structure and Bonding of Ethers, Epoxides, and Sulfides
Ethers, epoxides, and sulfides are important classes of organic compounds characterized by the presence of oxygen or sulfur atoms bonded to carbon. Their structural features influence their chemical properties and reactivity.
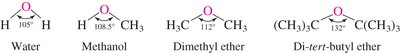
Ether C–O Bond Length: The C–O bond length in ethers is similar to that in alcohols (~142 pm).
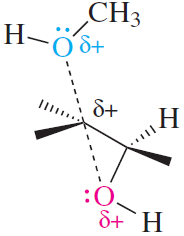
Epoxides: Epoxides are cyclic ethers with significant angle strain due to their three-membered ring structure.
Bond Angles: The C–O–C bond angle in ethers is influenced by steric strain and increases with bulkier substituents.
Sulfides: Sulfides are the sulfur analogs of ethers, with sulfur replacing oxygen.
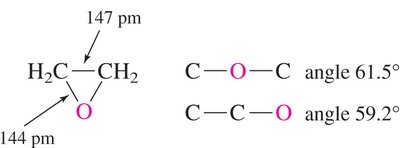
Physical Properties of Ethers
Ethers exhibit unique physical properties due to their molecular structure and lack of hydrogen bonding compared to alcohols.
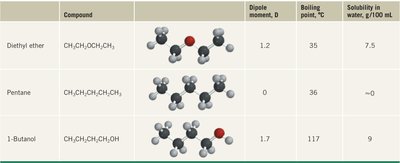
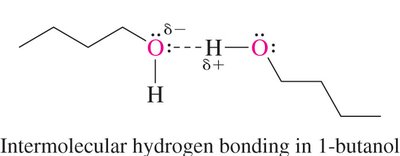
Boiling Point: Ethers generally have lower boiling points than alcohols of similar molecular weight due to the absence of intermolecular hydrogen bonding.
Polarity: Ethers are more polar than alkanes but less polar than alcohols.
Solubility: Ethers are moderately soluble in water, with solubility increasing with polarity.
Hydrogen Bonding: Alcohols can form intermolecular hydrogen bonds, leading to higher boiling points and solubility.
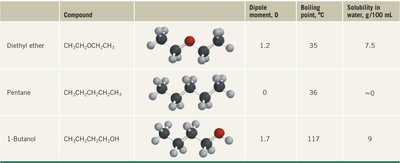
Compound | Dipole moment, D | Boiling point, °C | Solubility in water, g/100 mL |
|---|---|---|---|
Diethyl ether | 1.2 | 35 | 7.5 |
Pentane | 0 | 36 | ~0 |
1-Butanol | 1.7 | 117 | 9 |
Crown Ethers and Complex Formation
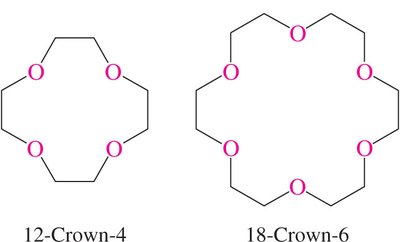
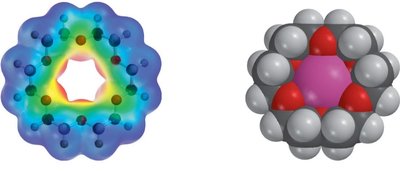
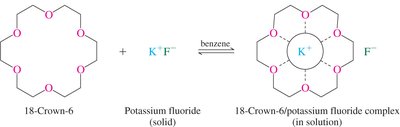
Crown ethers are cyclic polyethers that can strongly complex metal ions, especially alkali metals, due to their interior-facing oxygen atoms. This property is exploited in organic synthesis and phase transfer catalysis.
Lewis Acid/Base Complexes: Ethers act as Lewis bases, forming complexes with metal ions (Lewis acids).
Crown Ethers: Crown ethers are named by the total number of atoms in the ring and the number of oxygen atoms (e.g., 18-Crown-6).
Solubility: The hydrocarbon portion of crown ethers makes them soluble in organic solvents.
Applications: Crown ethers facilitate the solubility of ionic species in organic phases, enabling reactions such as nucleophilic substitutions.
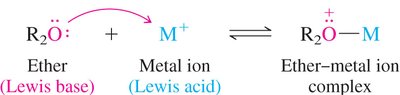

Synthesis of Ethers: Williamson Ether Synthesis
The Williamson Ether Synthesis is a classic method for preparing ethers via nucleophilic substitution (SN2) reactions. It is particularly effective for making unsymmetrical ethers.
Mechanism: An alkoxide ion reacts with a primary or methyl alkyl halide to form an ether and a salt.
Limitations: Secondary and tertiary alkyl halides are not suitable due to competing elimination (E2) reactions.
Reaction Equation:



Reactivity and Cleavage of Ethers
Ethers are generally unreactive, making them useful as solvents. However, they can be cleaved under acidic conditions, and are susceptible to oxidation.
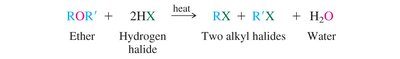
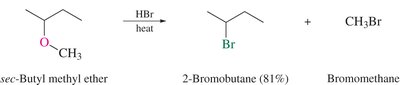
Acid-Catalyzed Cleavage: Ethers react with strong acids (e.g., HI, HBr) to yield alkyl halides and water.
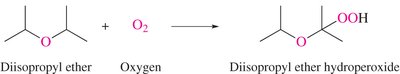
Oxidation: Ethers can form hydroperoxides upon exposure to oxygen, which are unstable and shock-sensitive.
Reaction Equation:
Epoxides: Structure, Preparation, and Reactivity
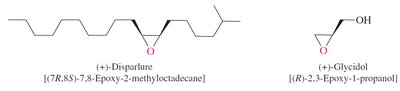
Epoxides are three-membered cyclic ethers with significant ring strain, making them highly reactive toward nucleophiles. They are important intermediates in organic synthesis and are found in nature.
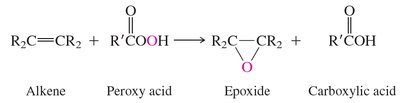
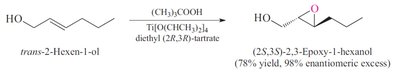
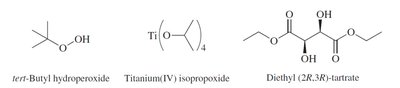
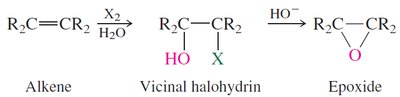
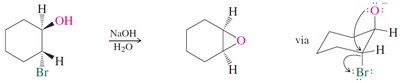
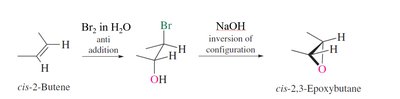
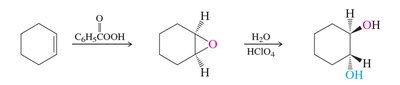
Preparation: Epoxides can be synthesized by epoxidation of alkenes using peroxy acids or by intramolecular SN2 reactions of vicinal halohydrins.
Sharpless Epoxidation: A method for enantioselective epoxidation of allylic alcohols using chiral catalysts.
Reactivity: Epoxides undergo ring-opening reactions with nucleophiles, with regioselectivity depending on reaction conditions (acidic or basic).
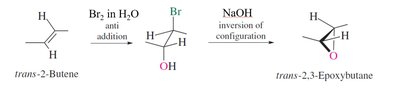
Reactions of Epoxides
Epoxides are versatile electrophiles, reacting with a variety of nucleophiles. The regioselectivity of ring opening depends on whether the conditions are acidic or basic.
Basic Conditions: Nucleophiles attack the less substituted carbon (SN2-like mechanism).
Acidic Conditions: Nucleophiles attack the more substituted carbon due to increased positive character (SN2-like, but acid-assisted).
Biological Processes: Epoxides are involved in various biochemical pathways.
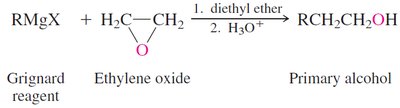
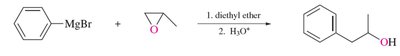
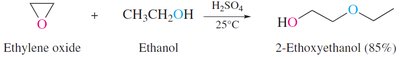
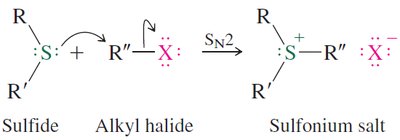
Sulfides: Synthesis, Oxidation, and Alkylation
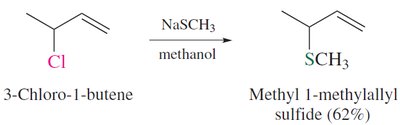
Sulfides are prepared by nucleophilic substitution of thiolates with alkyl halides. They can be oxidized to sulfoxides and sulfones, and alkylated to form sulfonium salts.
Synthesis: Primary and secondary alkyl halides react with thiolates via SN2 to form sulfides.
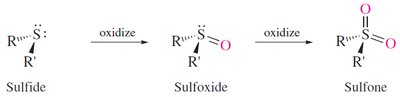
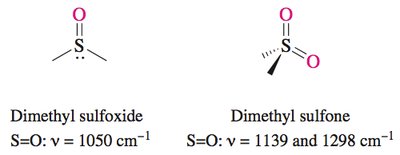
Oxidation: Sulfides can be oxidized to sulfoxides (R2S=O) and sulfones (R2S(=O)2).
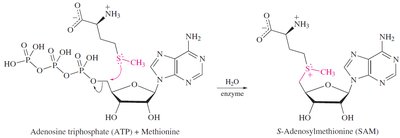
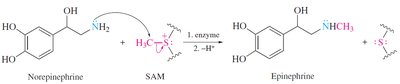
Alkylation: Sulfides react with alkyl halides to form sulfonium salts, which are important in biological methylation reactions.
Spectroscopic Techniques for Ethers, Epoxides, and Sulfides
Spectroscopy is essential for identifying and characterizing ethers, epoxides, and sulfides.
Infrared (IR) Spectroscopy: Ethers show a characteristic C–O–C stretch around 1100 cm−1. Sulfides have weak bands near 600 cm−1. Sulfoxides and sulfones exhibit strong S–O bands at higher wavenumbers.
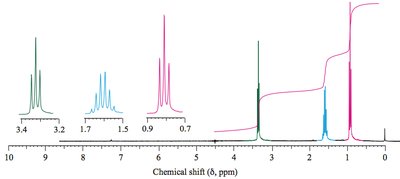
1H NMR Spectroscopy: Ether protons (H–C–OR) appear at δ 3.2–4.0. Sulfide protons (H–C–SR) are further upfield (δ 2.0–3.0). Epoxide protons are shielded (δ 2.5).
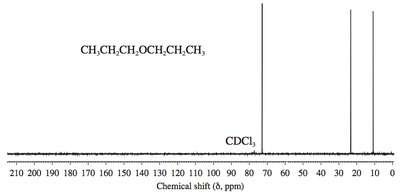
13C NMR Spectroscopy: Ether carbons (C–O–C) are less shielded (δ 57–87). Sulfide carbons (C–S–C) are more shielded.
Nomenclature of Ethers, Epoxides, and Sulfides
The nomenclature of ethers, epoxides, and sulfides follows IUPAC conventions, with both substitutive and functional class names used.
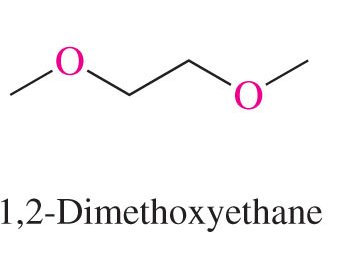
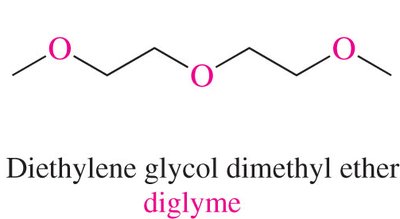
Ethers: Named as alkoxy derivatives or by functional class (e.g., diethyl ether).
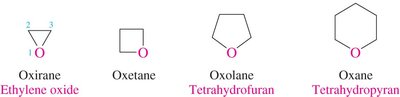
Epoxides: Named as oxiranes or by specifying the epoxide group.
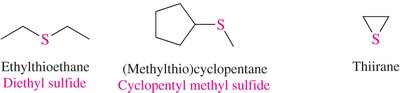
Sulfides: Named as alkylthio derivatives or by functional class (e.g., diethyl sulfide).
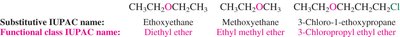
Additional info: These notes cover the essential chemistry of ethers, epoxides, and sulfides, including their structure, properties, synthesis, reactivity, and spectroscopic identification, as relevant to a college-level organic chemistry course.
