 Back
BackComprehensive Study Notes: Foundations and Early Chapters of Organic Chemistry
Study Guide - Smart Notes

Formulae, Equations, and Amount of Substance
Atoms, Elements, and Molecules
Understanding the basic building blocks of matter is essential for organic chemistry. Elements, atoms, molecules, and ions are fundamental concepts that underpin chemical reactions and molecular structures.
Element: The simplest substance that cannot be broken down by chemical reactions.
Atom: The smallest unit of an element retaining its properties.
Molecule: Two or more atoms bonded together.
Compound: Substance containing atoms of different elements combined.
Ion: Atom(s) with a positive or negative charge.
Monoatomic, Diatomic, Polyatomic ions: Ions made up of one, two, or three/more atoms respectively.
Writing Chemical Equations
Chemical equations represent the reactants and products in a reaction, using chemical symbols and balanced formulas.
Balancing Equations: Ensures equal numbers of each atom on both sides.
State Symbols: (s) solid, (l) liquid, (g) gas, (aq) aqueous.
Arrows: → for completion, ↔ for reversible reactions.
Writing Ionic Equations
Ionic equations show only the ions involved in a reaction, omitting spectator ions.
Write ionic compounds as ions if aqueous.
Remove ions present on both sides.
Reactions
Common reactions include those with metals, metal oxides/hydroxides, alkalis, carbonates, hydrogencarbonates, and displacement reactions.
Acid + Metal: Produces salt and hydrogen.
Acid + Metal Oxide/Hydroxide: Produces salt and water.
Acid + Alkali: Produces salt and water.
Acid + Carbonate: Produces salt, water, and CO2.
Displacement: More reactive element displaces less reactive one.

Precipitation and Chemical Tests
Precipitation reactions form solids from solutions. Tests for CO2 and sulphates involve precipitation.
Relative Masses and Moles
Relative atomic mass (RAM), molecular mass (Mr), and formula mass are used to compare and calculate quantities in reactions. The mole is a fundamental unit for counting particles.
Avogadro Constant: particles per mole.
Mole Calculation:
Yield and Atom Economy
Yield measures reaction efficiency; atom economy assesses how much reactant ends up in the desired product.
Percentage Yield:
Atom Economy:
Atomic Structure and the Periodic Table
Atoms and Isotopes
Atoms consist of protons, neutrons, and electrons. Isotopes are atoms of the same element with different numbers of neutrons.
Proton: Charge +1, mass 1.
Neutron: Charge 0, mass 1.
Electron: Charge -1, mass 1/1840.
Isotopes: Same atomic number, different mass number.

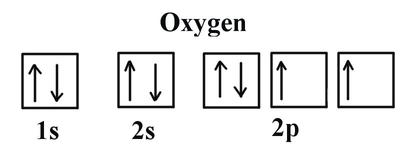
Atomic Orbitals and Electronic Configuration
Electrons occupy shells and subshells (s, p, d, f) around the nucleus. Electronic configuration describes the arrangement of electrons.
Shells: K (2), L (8), M (18), N (32).
Subshells: s (2), p (6), d (10), f (14).
Box Notation: Shows electron spins and Hund's Rule.

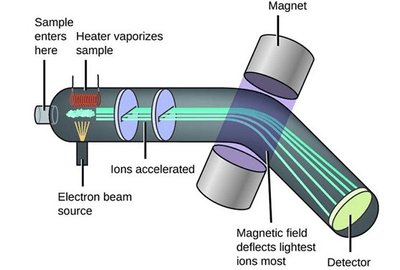
Mass Spectrometry
Mass spectrometry measures mass-to-charge ratios of ions, useful for identifying isotopes and calculating relative molecular mass.
Stages: Ionisation, Acceleration, Deflection, Detection.
Ionisation Energies
Ionisation energy is the energy required to remove an electron from an atom. Successive ionisation energies increase as electrons are removed closer to the nucleus.
First Ionisation Energy:
Second Ionisation Energy:

The Periodic Table
The periodic table organizes elements by increasing atomic number, grouping them by similar properties.
Groups: Vertical columns.
Periods: Horizontal rows.
Blocks: s-block, p-block, d-block.
Bonding and Structure
Ionic Bonds
Ionic bonds are strong electrostatic attractions between cations and anions, typically formed between metals and non-metals.
Octet Rule: Atoms strive for eight electrons in their outer shell.
Dot and Cross Diagrams: Visualize electron transfer.
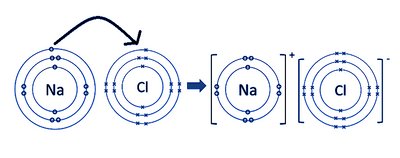
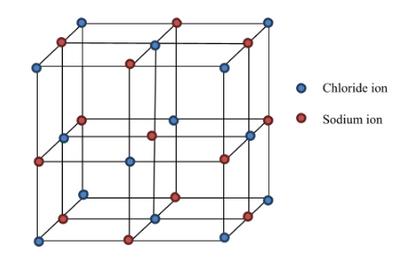
Ionic Lattice: Giant structure in solid state.
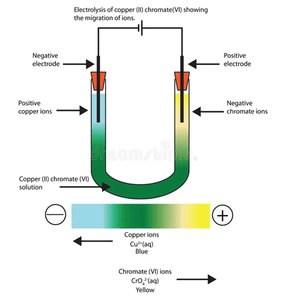
Migration of Ions: Evidence from electrolysis.
Ionic Radii and Polarisation
Ionic radii increase down a group and decrease across a period. Polarisation refers to distortion of electron density in anions by cations.
Polarising Power: Small, highly charged cations polarise anions.
Physical Properties of Ionic Compounds
High melting/boiling points.
Conduct electricity in molten/aqueous states.
Soluble in water due to hydration.
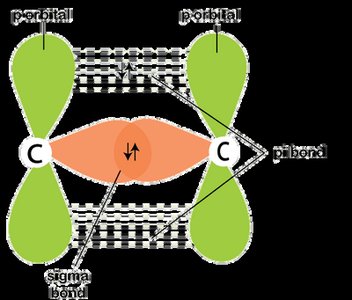
Covalent Bonding
Covalent bonds involve sharing electrons between atoms. Sigma (σ) bonds are formed by end-on overlap, pi (π) bonds by sideways overlap.
Bond Length: Distance between nuclei; shorter bonds are stronger.
Electronegativity: Atom's tendency to attract shared electrons.
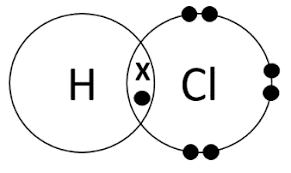
Dot and Cross Diagrams
Dot and cross diagrams represent covalent bonds and electron sharing.
Dative Covalent Bond
Dative bonds occur when both electrons in a shared pair come from one atom.
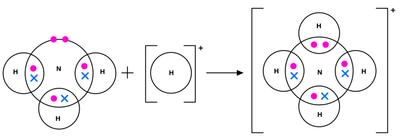
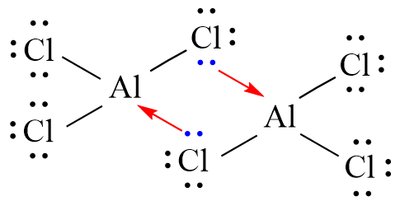
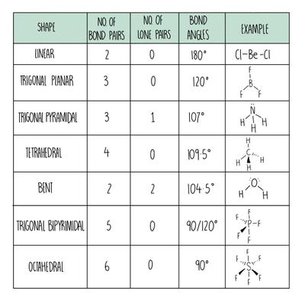
Shapes of Molecules and Ions
Electron-pair repulsion theory explains molecular shapes and bond angles.
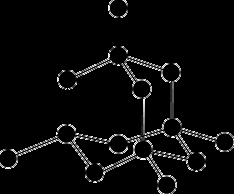
Giant Covalent Lattices
Diamond, graphite, and graphene are examples of giant covalent lattices with unique properties.
Diamond: Each C atom bonded to 4 others; rigid, hard, high melting point.
Graphite: Layered structure; good conductor; layers slide easily.
Graphene: Single layer; excellent electrical properties.
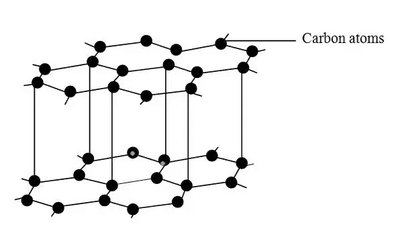
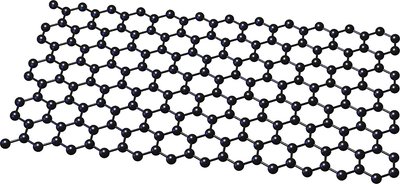
Metallic Bonding
Metallic bonds are strong attractions between positive ions and delocalised electrons, giving metals their characteristic properties.
High melting points, good conductivity, malleability, ductility.
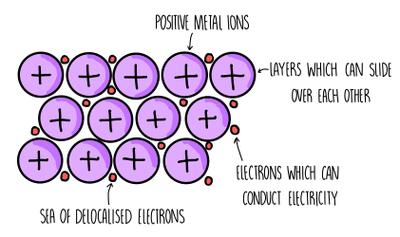
Structure and Properties Table
The properties of materials depend on their bonding and structure.
Type | Bonds Present | Melting/Boiling Points | Hardness | Electrical Conductivity | Solubility in Water | Solubility in Non-Polar Solvents |
|---|---|---|---|---|---|---|
Giant Ionic | Strong Ionic | High | Hard but brittle | Molten/aqueous | Often soluble | Insoluble |
Giant Covalent | Strong Covalent | Very High | Very Hard | Non-conductor | Insoluble | Insoluble |
Giant Metallic | Strong Metallic | Fairly High | Hard but malleable | Conductor | Insoluble | Insoluble |
Simple Molecular | Weak Covalent | Low | Soft | Non-conductor | Generally insoluble | Usually soluble |
Alkanes
Introduction to Organic Chemistry
Organic compounds contain carbon. Hydrocarbons are compounds of carbon and hydrogen. Alkanes are saturated hydrocarbons with only single bonds.
General Formula:
Methane:
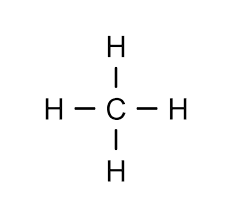
Nomenclature and Representation
IUPAC rules govern naming. Functional groups determine suffixes. Various formulae represent organic compounds: displayed, skeletal, structural, molecular, empirical.
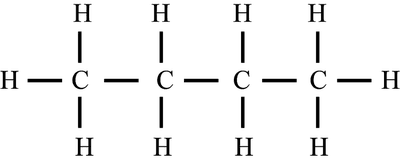
Cyclic Structures and Isomers
Cycloalkanes are ring structures with formula . Isomers have the same molecular formula but different arrangements.
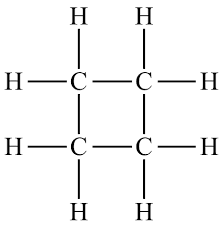
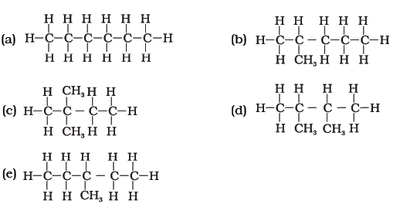
Chemical Reactions
Alkanes undergo substitution reactions, not addition due to lack of double bonds. Combustion is an oxidation reaction.
Free Radical Substitution Reaction
Bond fission can be homolytic (even) or heterolytic (uneven). Free radical substitution involves initiation, propagation, and termination steps.
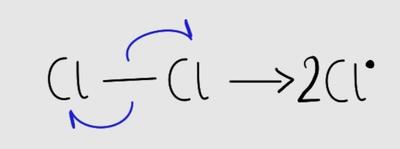
Hazards, Risks, and Risk Assessment
Hazards are inherent properties; risk depends on usage. Risk assessment minimizes potential dangers. Hazard symbols communicate risks.

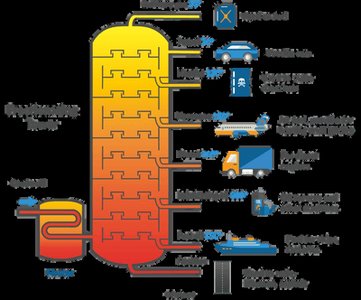
Fuels
Crude oil is processed by fractional distillation and cracking to produce alkanes. Reforming creates branched/cyclic hydrocarbons. Combustion produces CO2 and H2O; incomplete combustion produces CO.
Alkenes
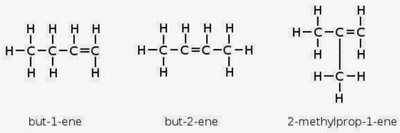
Introduction and Geometric Isomerism
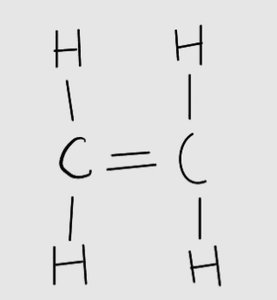
Alkenes have at least one C=C double bond, making them more reactive. They exhibit structural and stereoisomerism, including geometric (cis-trans/E-Z) isomerism due to restricted rotation about the double bond.
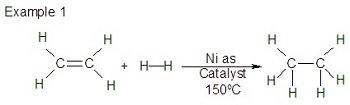
Addition Reactions
Alkenes undergo addition reactions, breaking the pi bond and forming new bonds. Tests for alkenes include decolourisation of bromine water and potassium permanganate.
Mechanism of Addition Reaction
Alkenes undergo electrophilic addition. Markovnikov's Rule determines major/minor products based on carbocation stability.
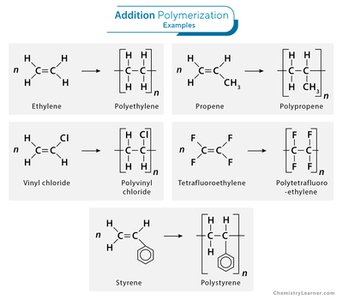
Addition Polymers
Alkenes polymerise to form addition polymers. The repeating unit is shown without the double bond. Polymerisation has 100% atom economy.
Dealing With Polymer Waste
Incineration and development of biodegradable polymers are methods to manage polymer waste.
*Additional info: These notes cover foundational concepts and early chapters relevant to organic chemistry, including atomic structure, bonding, periodic trends, and the chemistry of alkanes and alkenes. The content is structured to provide a comprehensive overview suitable for college-level organic chemistry students.*
