 Back
BackConjugated Pi Systems and Pericyclic Reactions: Study Notes
Study Guide - Smart Notes

Conjugated Pi Systems and Pericyclic Reactions
Conjugated Dienes
Conjugated dienes are organic compounds containing two double bonds separated by a single bond, allowing for delocalization of π electrons. This delocalization imparts unique stability and reactivity to these molecules.
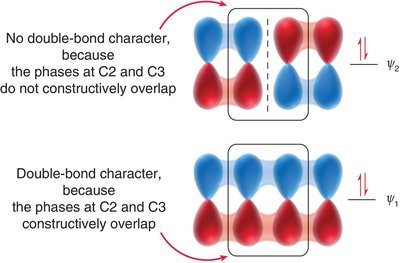
Stabilization Energy: Conjugated dienes are more stable than isolated dienes, as shown by heats of hydrogenation. The stabilization energy is approximately 15 kJ/mol when two π bonds are conjugated.
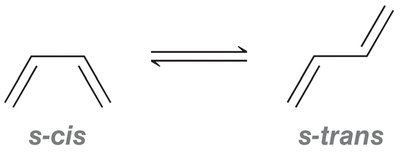
s-trans vs. s-cis Conformations: The s-trans conformation is lower in energy due to reduced steric hindrance compared to s-cis.
Example: Butadiene is a classic conjugated diene.
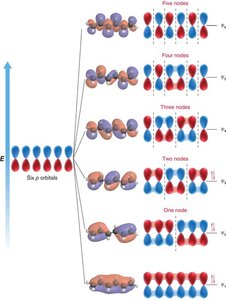
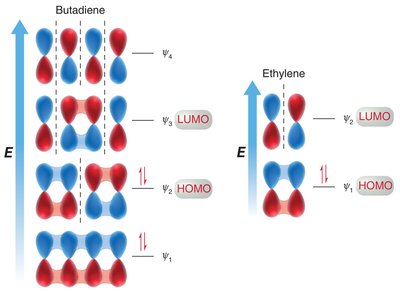
Molecular Orbital Theory of Conjugated Dienes
Molecular orbital (MO) theory explains the electronic structure of conjugated systems by combining atomic orbitals into molecular orbitals, which are occupied by electrons according to their energy levels.
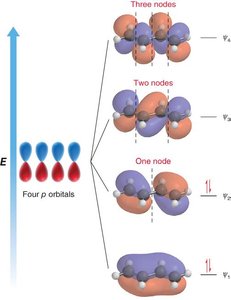
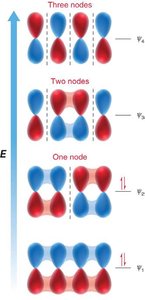
MO Diagram for Butadiene: Four p orbitals combine to form four molecular orbitals with increasing energy and nodes.
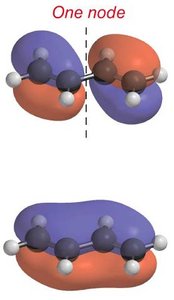
Electron Occupancy: The four π electrons in butadiene occupy the two lowest energy MOs.
Node Formation: The number of nodes increases with energy, affecting bonding and antibonding character.
Example: 1,3,5-Hexatriene has six p orbitals forming six MOs.
Introduction to Pericyclic Reactions
Pericyclic reactions are a class of organic reactions that proceed via a concerted mechanism, involving a cyclic transition state and the movement of electrons in a closed loop. They do not involve ionic or free radical intermediates.
Types of Pericyclic Reactions:
Cycloaddition Reactions: Two or more π systems combine to form a ring.
Electrocyclic Reactions: A conjugated polyene undergoes cyclization, converting a π bond to a σ bond.
Sigmatropic Rearrangement: A sigma bond migrates across a π system, changing the location of π bonds.
Characteristic Features:
The mechanism is concerted (no intermediates).
Electron movement occurs in a ring around a closed loop.
The transition state is cyclic.
Solvent polarity has little effect on reaction rate.
Comparison of Pericyclic Reaction Types
The three major types of pericyclic reactions differ in the changes in the number of sigma and pi bonds.
Reaction Type | Change in σ Bonds | Change in π Bonds |
|---|---|---|
Cycloaddition | +2 | -2 |
Electrocyclic | +1 | -1 |
Sigmatropic | 0 | 0 |

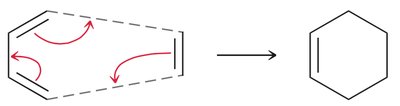
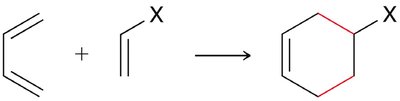
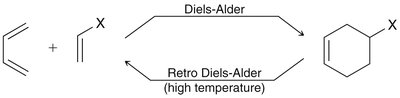
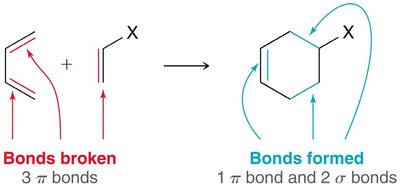
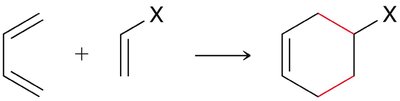
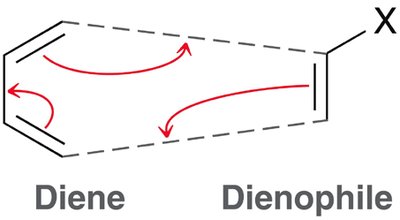
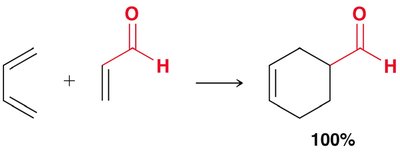
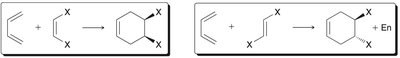
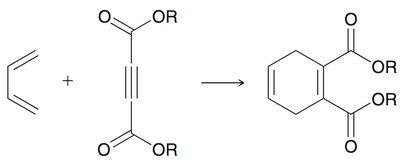
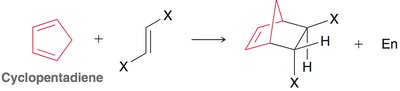
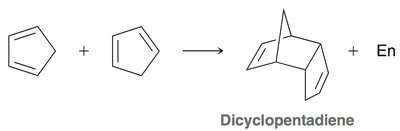
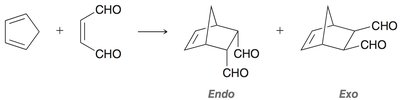
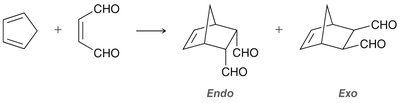
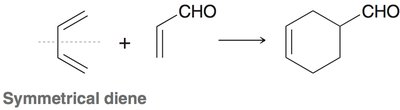
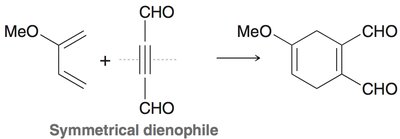
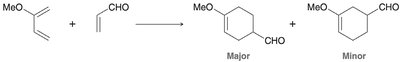
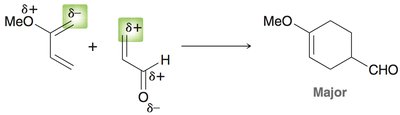
Diels-Alder Reaction ([4+2] Cycloaddition)
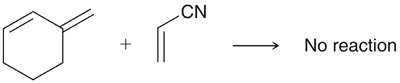
The Diels-Alder reaction is a classic cycloaddition between a conjugated diene and a dienophile, forming a six-membered ring. It is a concerted reaction, meaning all bond changes occur simultaneously.
Mechanism: The reaction proceeds via a cyclic transition state, with electron movement shown by curved arrows.
Thermodynamics: The reaction is favored at low to moderate temperatures due to product stability. At high temperatures, the retro Diels-Alder reaction (reverse process) is favored due to entropy.
Bond Changes: Conversion of π bonds to σ bonds provides a negative ΔH (enthalpy term).
Entropy: ΔS is negative, as two molecules combine to form one product.
Equation:
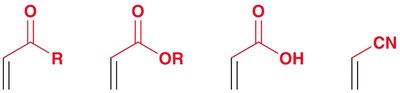
Reactants: Diene (must be in s-cis conformation) and dienophile (often with electron-withdrawing groups).
Stereospecificity: The reaction is stereospecific, depending on the geometry of the dienophile (E or Z).
Regioselectivity: If either reactant is unsymmetrical, two regioisomers can form, but one is usually favored.
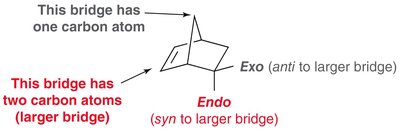
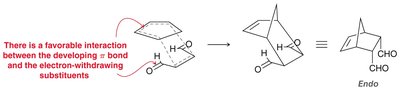
Endo vs. Exo Products: In bicyclic systems, the endo product is often favored due to favorable interactions between electron-withdrawing groups and the developing π bond.
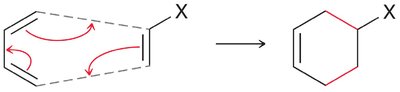
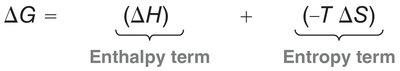
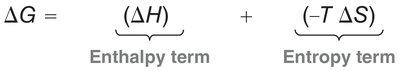

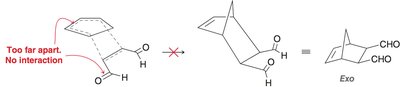
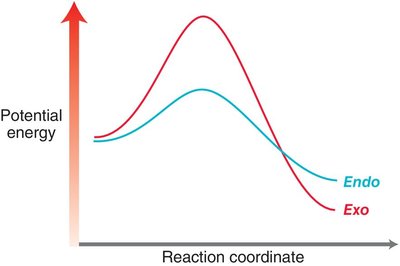
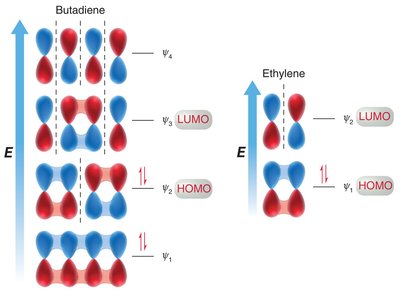
Molecular Orbital Descriptions of Cycloadditions
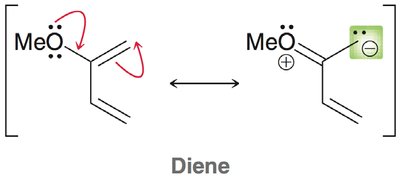
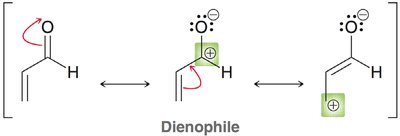
The molecular orbital approach explains cycloaddition reactions by analyzing the interaction between the highest occupied molecular orbital (HOMO) of one reactant and the lowest unoccupied molecular orbital (LUMO) of the other.
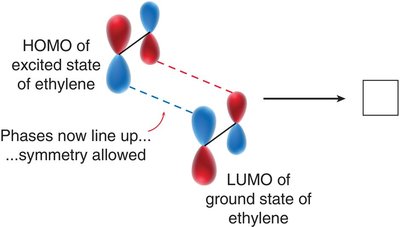
Symmetry Requirements: The phases of the reacting MOs must be symmetrical for the reaction to be allowed (conservation of orbital symmetry).
Diels-Alder: The HOMO of the diene interacts with the LUMO of the dienophile, especially when the dienophile has electron-withdrawing groups.
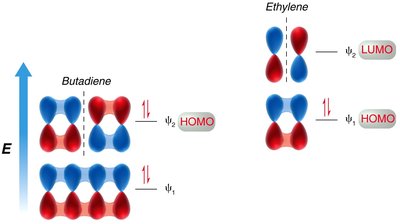
[2+2] Cycloaddition: This reaction is symmetry-forbidden under thermal conditions but allowed under photochemical conditions (light excites an electron).
![[2+2] cycloaddition MO analysis](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/76d96794_image_43.png)

![Symmetry forbidden [2+2] cycloaddition](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/76d96794_image_45.png)
![Photochemical excitation for [2+2] cycloaddition](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/76d96794_image_46.png)
![Symmetry allowed [2+2] cycloaddition under light](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/76d96794_image_47.png)
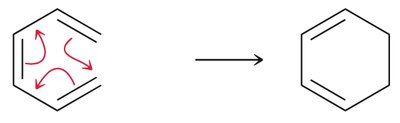
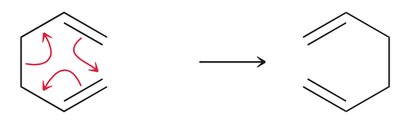
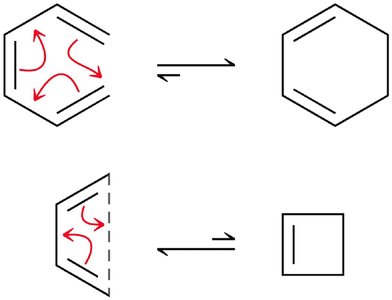
Electrocyclic Reactions
Electrocyclic reactions are pericyclic processes where a conjugated polyene undergoes cyclization, converting a π bond to a σ bond. The stereochemistry of the product depends on the symmetry of the HOMO and the conditions (heat vs. light).
Disrotatory vs. Conrotatory: Under thermal conditions, conjugated systems with 6 π electrons undergo disrotatory cyclization, while those with 4 π electrons undergo conrotatory cyclization.
Photochemical Conditions: Light excites an electron, changing the symmetry of the HOMO and altering the stereochemical outcome.
Woodward-Hoffmann Rules: These rules summarize the stereochemical outcomes for electrocyclic reactions based on electron count and conditions.
Sigmatropic Rearrangements
Sigmatropic rearrangements are pericyclic reactions in which a sigma bond migrates across a π system, resulting in a new sigma bond and a change in the location of π bonds. The notation [i,j] indicates the number of atoms involved on each side of the migrating bond.
[3,3] Sigmatropic Rearrangement: Both the Cope and Claisen rearrangements are [3,3] sigmatropic reactions.
Cope Rearrangement: All atoms in the cyclic transition state are carbon; equilibrium favors more substituted alkenes.
Claisen Rearrangement: The oxygen analogue of the Cope rearrangement, occurring with allylic vinylic ethers and aryl ethers; equilibrium favors formation of the C=O bond.
[1,5] Sigmatropic Rearrangement: Involves migration of a sigma bond over five atoms.
Biological Example: Pericyclic reactions are involved in the biosynthesis of Vitamin D.
Additional info: The Woodward-Hoffmann rules and molecular orbital symmetry are fundamental for predicting the outcomes of pericyclic reactions. Practice problems and checkpoints are recommended for mastery.
