 Back
BackConjugated Systems and Diels–Alder Reactions: Study Notes
Study Guide - Smart Notes

Conjugated Systems
Structure and Stability of Conjugated Double Bonds
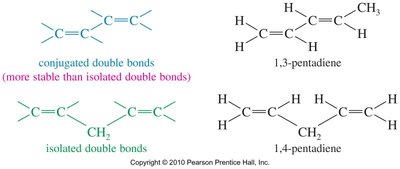
Conjugated systems are organic molecules where double bonds are separated by a single bond, allowing for electron delocalization across the molecule. This delocalization imparts unique stability and reactivity compared to isolated double bonds.
Conjugated double bonds are more stable than isolated double bonds due to electron delocalization.
In 1,3-butadiene, the central single bond is shorter than a typical single bond (less than 1.54 Å), indicating partial double bond character.
Electrons are delocalized over the entire conjugated system, contributing to stability.
Conjugated systems are common in organic molecules such as dienes and aromatic compounds.
Example: 1,3-pentadiene (conjugated) vs. 1,4-pentadiene (isolated).
Heat of Hydrogenation and Relative Stabilities
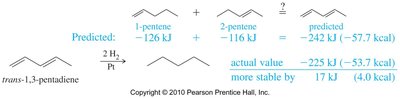
The heat of hydrogenation is a measure of the stability of a molecule. Conjugated dienes have a lower heat of hydrogenation than predicted by the sum of individual double bonds, indicating greater stability.
For trans-1,3-pentadiene, the actual heat of hydrogenation is less than the predicted value for two isolated double bonds.
This difference (about 17 kJ or 4.0 kcal/mol) demonstrates the stabilizing effect of conjugation.
Equation:
Conformations of 1,3-Butadiene
1,3-Butadiene exists in two main conformations: s-trans and s-cis. The s-trans conformer is more stable by 12 kJ/mol, but both can interconvert at room temperature.
s-trans is more stable than s-cis by 12 kJ/mol (2.8 kcal/mol).
Interconversion is possible due to rotation around the central single bond.
Allylic Cations and Carbocation Stability
Allylic cations are stabilized by resonance, with the positive charge delocalized over two carbons. This makes them more stable than nonconjugated carbocations.
Stability order: 1° allylic ≈ 2° carbocation; 2° allylic ≈ 3° carbocation.
Resonance stabilization is key to their reactivity.
SN2 Reactions of Allylic Halides and Tosylates
Allylic halides and tosylates are highly reactive in SN2 reactions due to stabilization of the transition state by conjugation with the π orbitals.
These substrates react readily with Grignard reagents and organolithiums.
Transition state is stabilized by conjugation.
Diels–Alder Reaction
Overview and Mechanism
The Diels–Alder reaction is a [4+2] cycloaddition between a conjugated diene and a dienophile (an electron-deficient alkene), resulting in the formation of a cyclohexene ring. This reaction is widely used in organic synthesis for constructing six-membered rings.
Named after Otto Diels and Kurt Alder (Nobel Prize, 1950).
Produces a cyclohexene ring via a concerted mechanism.
Diene must be electron-rich; dienophile must be electron-poor.
Example: Diene + Dienophile → Cyclohexene ring.

Electron Donor and Electron Withdrawing Groups
Substituents on the diene and dienophile affect the reactivity and selectivity of the Diels–Alder reaction.
Electron Donor Groups (EDG): Donate electrons via sigma or π bonds or resonance, increasing nucleophilicity. Examples: alkyl (-CH3), alkoxy (-OCH3), amines (-NH2).
Electron Withdrawing Groups (EWG): Withdraw electrons, increasing electrophilicity. Examples: carbonyls (aldehydes, ketones, esters), carboxylic acids, nitriles, nitro groups.
Stereochemical Requirements
The diene must be in the s-cis conformation for the Diels–Alder reaction to occur. The p orbitals of the diene and dienophile must overlap to form new sigma bonds.
s-cis conformation is required for orbital overlap.
s-trans is more stable but cannot participate in the reaction.
Rotation around the single bond allows interconversion.
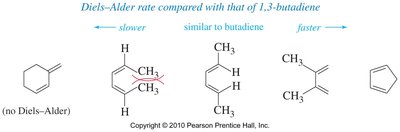
Rate of Diels–Alder Reaction for Dienes
The rate of the Diels–Alder reaction depends on the conformation and steric hindrance of the diene. Cyclopentadiene reacts rapidly due to its fixed s-cis conformation, while sterically hindered dienes react more slowly.
Cyclopentadiene: fast reaction due to fixed s-cis conformation.
Sterically hindered dienes: slower reaction.
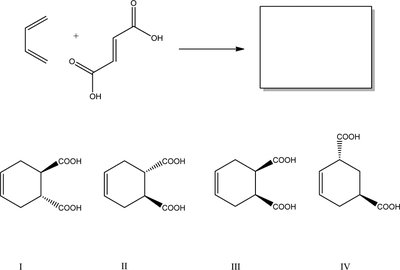
Examples of Diels–Alder Reactions
Various examples illustrate the stereochemical outcomes and product formation in Diels–Alder reactions.
Pay attention to the stereochemistry of the product.
Major product is determined by the orientation of substituents and the conformation of the diene.
Stereochemistry and the Endo Rule
The endo rule states that electron-withdrawing groups on the dienophile prefer the endo position due to secondary orbital overlap with the diene. This influences the major product formed.
Endo products are favored when electron-withdrawing groups are present.
Secondary overlap between p orbitals stabilizes the transition state.
Exo products are less favored.
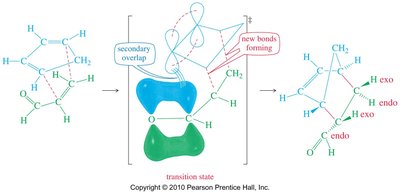
Examples of Endo Rule
Specific examples demonstrate the application of the endo rule in predicting major products of Diels–Alder reactions.
Major product is often the endo isomer.
Stereochemistry is crucial in determining the outcome.

Unsymmetrical Reagents: 1,2 and 1,4 Products
When unsymmetrical reagents are used, the electron-donating groups on the diene and electron-withdrawing groups on the dienophile influence whether the product is a 1,2 or 1,4 adduct.
EDG on diene and EWG on dienophile favor specific regioselectivity.
Products can be classified as 1,2 or 1,4 adducts based on substituent positions.
Diels–Alder: Retrosynthetic Analysis
Retrosynthetic analysis is used to plan the synthesis of complex molecules by breaking them down into simpler precursors. The Diels–Alder reaction is a key step in constructing six-membered rings.
Identify possible diene and dienophile precursors.
Predict the product based on the structure and substituents.
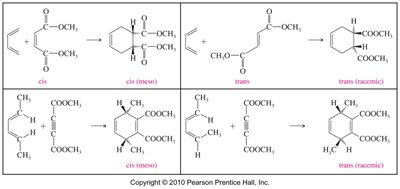
Summary Table: Diels–Alder Stereochemistry
The stereochemistry of the Diels–Alder reaction depends on the configuration of the diene and dienophile. The following table summarizes the outcomes:
Diene | Dienophile | Product | Stereochemistry |
|---|---|---|---|
cis | cis | cyclohexene | meso |
trans | trans | cyclohexene | racemic |
cis | trans | cyclohexene | racemic |
Additional info: Academic context was added to clarify the mechanism, stereochemistry, and practical applications of the Diels–Alder reaction, as well as the stability and properties of conjugated systems.
