 Back
BackCh. 16 Part 1
Study Guide - Smart Notes

Conjugation in Organic Molecules
Definition and Structural Requirements
Conjugation in organic chemistry refers to the overlap of p orbitals across three or more adjacent atoms, allowing for the delocalization of electrons. This phenomenon is central to the stability and reactivity of many organic compounds, especially those containing alternating double and single bonds.
Conjugated system: A molecule with alternating single and double bonds, such as a 1,3-diene.
p Orbitals: Each carbon in a conjugated system is sp2-hybridized, possessing one unhybridized p orbital that can participate in π-bonding.
Delocalization: The π electrons are not confined between two atoms but are spread over several adjacent atoms, increasing molecular stability.
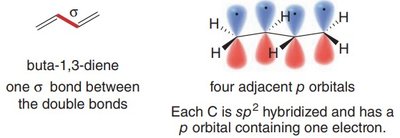
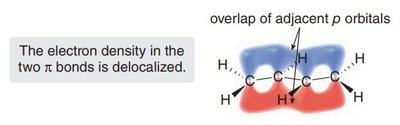
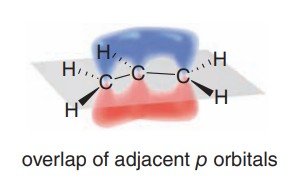
1,3-Dienes: Structure and Bonding
1,3-Dienes are the simplest conjugated systems, featuring two double bonds separated by a single bond. Their unique bonding leads to distinct physical and chemical properties compared to isolated double bonds.
Bond Lengths: The central C–C bond in a conjugated diene is shorter than a typical single bond due to partial double bond character from delocalization.
Hybridization: All carbons are sp2-hybridized, allowing for continuous overlap of p orbitals.
Examples: Buta-1,3-diene (conjugated) vs. penta-1,4-diene (isolated).
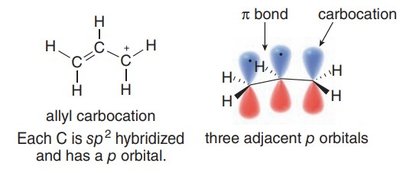
Allylic Carbocations and Their Stability
Structure and Delocalization
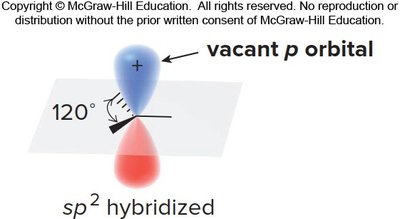
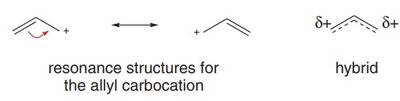
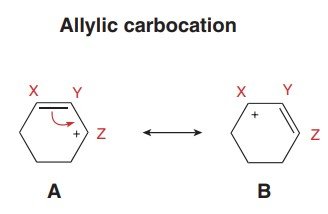
An allylic carbocation is a positively charged carbon atom adjacent to a double bond. The positive charge is delocalized over multiple atoms due to resonance, which greatly enhances its stability compared to typical primary carbocations.
Hybridization: All carbons in the allylic system are sp2-hybridized, with three adjacent p orbitals available for delocalization.
Vacant p Orbital: The carbocation center has an empty p orbital, which participates in resonance stabilization.
Carbocation Stability: Allylic vs. Alkyl
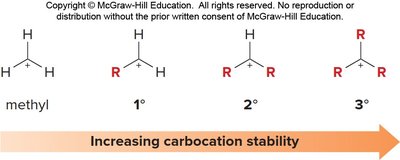
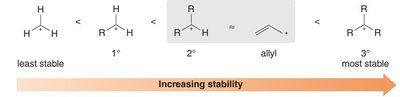
Carbocation stability increases with the degree of alkyl substitution and resonance stabilization. Allylic carbocations are unusually stable due to resonance, often comparable to secondary carbocations.
Order of Stability: Methyl < 1° < 2° ≈ Allyl < 3°
Reason: Resonance allows the positive charge to be spread over multiple atoms, reducing electron deficiency at any one center.
Resonance: Delocalization of Electrons
Principles of Resonance
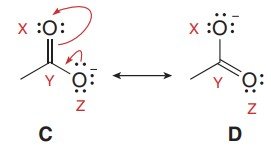
Resonance describes the delocalization of electrons in molecules where multiple valid Lewis structures can be drawn. Only π electrons and lone pairs adjacent to π systems or charges are moved; σ bonds remain unchanged.
Rules: Move only π electrons or lone pairs; maintain overall charge; do not exceed the octet rule for second-row elements.
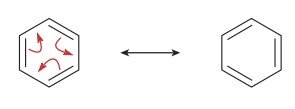
Common Systems: Allylic carbocations, carbanions, radicals, acetate, amidate, conjugated dienes, and aromatic rings.
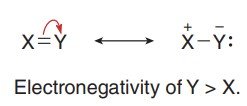
Resonance Hybrid and Contributors
The actual structure of a molecule is a resonance hybrid, which is a weighted average of all possible resonance contributors. The best contributors have full octets, minimal charge separation, and place negative charges on more electronegative atoms.
Major Contributor: Structure with the most atoms having full octets, least formal charges, and negative charges on electronegative atoms.
Minor Contributor: Structures with incomplete octets or more charge separation.
Practice: Draw all resonance structures for a given system and classify each as a major or minor contributor to the resonance hybrid.
Atom Hybridization and Resonance
Hybridization and Electron Delocalization
Electron delocalization in resonance requires overlapping p orbitals, which is only possible if the involved atoms are sp2 or sp hybridized. The hybridization of an atom may differ from simple predictions if resonance is possible.
Hybridization Table: The following table summarizes the relationship between the number of groups bonded to carbon, hybridization, bond angle, and observed bonding.
Number of groups bonded to C | Hybridization | Bond angle | Example | Observed bonding |
|---|---|---|---|---|
4 | sp3 | 109.5° | CH3CH3 (ethane) | One σ bond |
3 | sp2 | 120° | CH2=CH2 (ethylene) | One σ bond, one π bond |
2 | sp | 180° | HC≡CH (acetylene) | One σ bond, two π bonds |
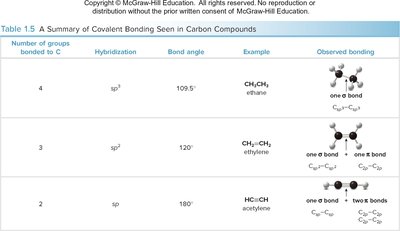
Additional info: Understanding hybridization is crucial for predicting molecular geometry and the possibility of resonance stabilization in organic molecules.
