 Back
BackCh. 16 Part 2
Study Guide - Smart Notes

Conjugation in Organic Molecules
Definition and Structural Requirements
Conjugation occurs when three or more adjacent atoms possess p orbitals that can overlap, allowing for electron delocalization. This phenomenon is central to the stability and reactivity of many organic molecules, especially dienes and allylic systems.
Conjugated system: A molecule with alternating single and double bonds, such as 1,3-diene, where p orbitals on adjacent atoms overlap.
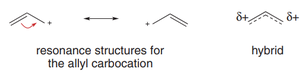
Allylic carbocation: A positively charged carbon atom adjacent to a double bond, enabling resonance stabilization.
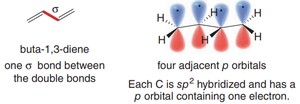
1,3-Dienes: Structure and Bonding
Hybridization and Orbital Overlap
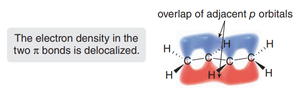
In 1,3-dienes, each carbon is sp2-hybridized, possessing three sp2 orbitals for σ-bonding and one unhybridized p orbital for π-bonding. The overlap of these p orbitals across four adjacent carbons leads to delocalized π electron density, which is a hallmark of conjugation.
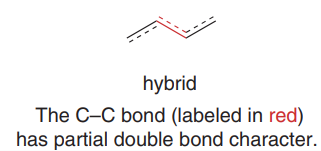
Each C–C bond between the double bonds is shorter than a typical single bond due to partial double bond character.
Delocalization of electrons across the π system increases molecular stability.
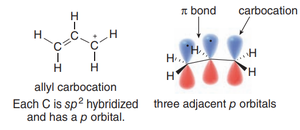
Allylic Carbocations and Resonance
Structure and Stability
An allylic carbocation is a carbocation where the positive charge is adjacent to a double bond. The structure allows for resonance stabilization, as the positive charge and π electrons can be delocalized over three atoms.
Each carbon in the allylic system is sp2-hybridized, with available p orbitals for delocalization.
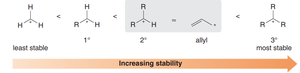
The resonance effect distributes the positive charge, making the allylic carbocation as stable as a secondary carbocation.
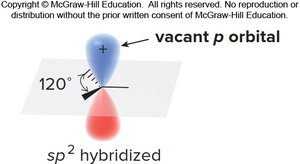
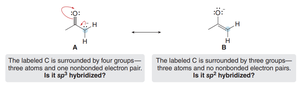
Atom Hybridization and Resonance
Requirements for Electron Delocalization
Electron delocalization through resonance requires overlapping p orbitals. The hybridization of atoms may differ from simple models, especially when lone pairs participate in resonance.
For resonance to occur, an atom must have a p orbital available (often sp2-hybridized with a lone pair in the p orbital).
Delocalization stabilizes the molecule by spreading out electron density.

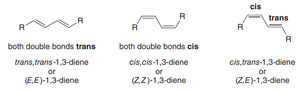
Conjugated Dienes: Stereochemistry and Conformations
Stereoisomers and Conformational Isomerism
Conjugated dienes, such as 1,3-butadiene, can exist as different stereoisomers and conformers due to rotation about the central single bond.
Stereoisomers: trans-trans, cis-cis, and cis-trans forms are possible.
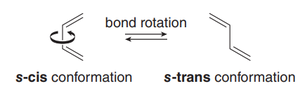
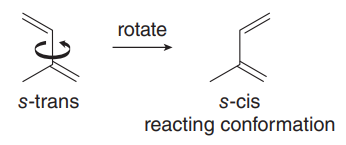
Conformations: Rotation about the central C–C bond gives rise to s-cis and s-trans conformations, which influence reactivity in cycloaddition reactions.
Bond Lengths and Resonance in Dienes
Percent s-Character and Bond Length
The bond between the two double bonds in a conjugated diene is shorter than a typical single bond due to increased s-character and resonance delocalization.
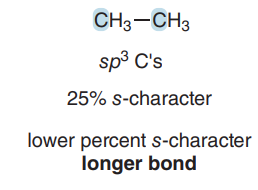
sp3 C–C bond: 25% s-character, longer bond.
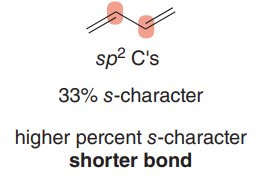
sp2 C–C bond: 33% s-character, shorter bond.
Resonance gives the central bond partial double bond character.
Stability of Conjugated Dienes
Resonance Stabilization
Conjugated dienes are more stable than isolated dienes due to the delocalization of π electrons, which lowers the overall energy of the molecule.
Delocalization distributes electron density over a larger framework, reducing electron-electron repulsion and increasing stability.
Electrophilic Addition to Dienes
Product Distribution: Isolated vs. Conjugated Dienes
Electrophilic addition reactions of dienes differ depending on whether the diene is isolated or conjugated.
Isolated diene: Addition yields a single product, following Markovnikov’s rule (H adds to less substituted carbon).
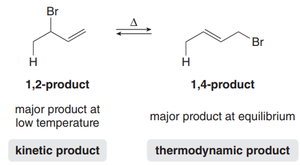
Conjugated diene: Addition yields two products: the 1,2-product (addition across C1 and C2) and the 1,4-product (addition across C1 and C4).
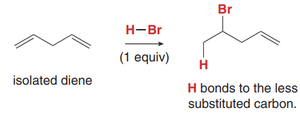
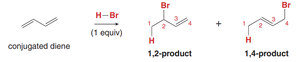
Mechanism of Electrophilic Addition to Conjugated Dienes
The reaction proceeds via the formation of an allylic carbocation intermediate, stabilized by resonance. The nucleophile can attack at two positions, leading to different products.
Step 1: Protonation at the terminal carbon forms an allylic carbocation (slow step).
Step 2: Nucleophilic attack by X– at either C2 (1,2-product) or C4 (1,4-product), dictated by resonance stabilization.
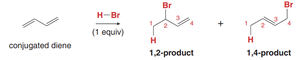
Kinetic vs. Thermodynamic Control
The product distribution depends on reaction temperature:
1,2-product (kinetic product): Forms faster due to proximity effect; major product at low temperature.
1,4-product (thermodynamic product): More stable due to greater alkene substitution; major product at high temperature.
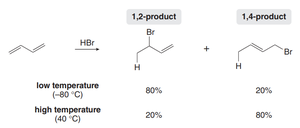
Diels-Alder Reaction
Overview and Mechanism
The Diels-Alder reaction is a [4+2] cycloaddition between a conjugated diene and a dienophile, forming a six-membered ring. This reaction is concerted, involving the simultaneous formation of two new σ bonds and one new π bond.
Requires heat to proceed.
Mechanism involves three curved arrows in a single step.
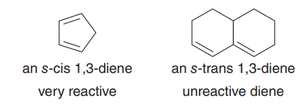
Only the s-cis conformation of the diene is reactive; s-trans conformers must rotate to s-cis to react.
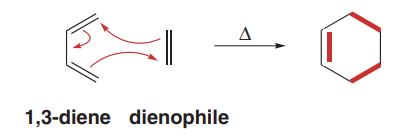
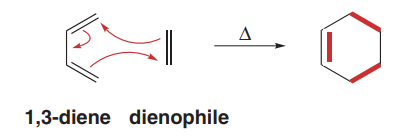
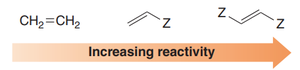
Reactivity of Dienes and Dienophiles
The reactivity of the diene depends on its conformation, while the reactivity of the dienophile is enhanced by electron-withdrawing groups (EWGs).
Cyclic dienes locked in s-cis are highly reactive; those locked in s-trans are unreactive.
Dienophiles with EWGs (e.g., carbonyls) react faster.
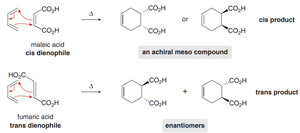
Cis/trans stereochemistry of the dienophile is preserved in the product.
Endo and Exo Addition in Diels-Alder Reactions
When the dienophile is cyclic or substituted, two possible stereochemical outcomes exist: endo and exo. The endo product is typically favored due to secondary orbital interactions.
Endo product: Dienophile substituent is oriented under the ring (pseudo-equatorial), closer to the electron-rich diene.
Exo product: Dienophile substituent is oriented above the ring (pseudo-axial).
Endo selectivity is enhanced by electron-withdrawing groups on the dienophile.
Summary Table: Hybridization and Bonding in Carbon Compounds
Number of groups bonded to C | Hybridization | Bond angle | Example | Observed bonding |
|---|---|---|---|---|
4 | sp3 | 109.5° | CH3CH3 (ethane) | σ bonds only |
3 | sp2 | 120° | CH2=CH2 (ethylene) | σ and π bonds |
2 | sp | 180° | HC≡CH (acetylene) | σ and two π bonds |

Practice Problems
Predict the products of electrophilic addition to conjugated dienes and identify which is favored at low or high temperature.
Draw mechanisms for the formation of both kinetic and thermodynamic products.
Draw the major product of each Diels-Alder reaction, considering stereochemistry and endo/exo selectivity.
