 Back
BackDelocalization, Stability, Reactivity, and pKa in Organic Chemistry
Study Guide - Smart Notes

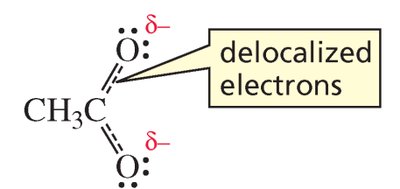
Delocalization of Electrons
Localized vs. Delocalized Electrons
Electrons in organic molecules can be either localized (confined to a single atom or shared between two atoms in a bond) or delocalized (spread over three or more atoms). Delocalization is a key concept for understanding resonance, stability, and reactivity in organic chemistry.
Localized electrons: Found in single bonds or as lone pairs on atoms not involved in resonance.
Delocalized electrons: Shared among three or more atoms, often represented by resonance structures.
Resonance hybrid: The true structure of a molecule with delocalized electrons, depicted as a composite of multiple resonance contributors.
Resonance structures differ only in the positions of π and nonbonding electrons; atoms and σ bonds remain unchanged.

Orbital Requirements for Delocalization
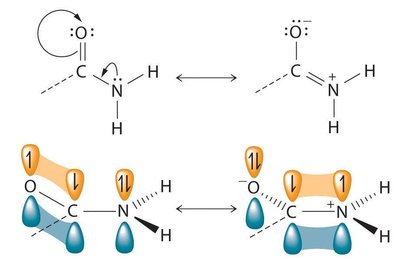
For resonance to occur, all atoms involved must have a p orbital (i.e., be sp2-hybridized). This allows for the overlap of p orbitals and the delocalization of π electrons.
Example: The amide functional group can be represented by two resonance structures, both involving p orbital overlap.
Resonance in Benzene
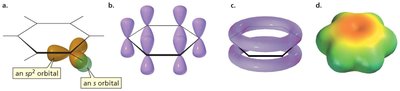
Benzene (C6H6) is a classic example of resonance. All carbon-carbon bonds in benzene are experimentally found to be the same length, indicating delocalization of π electrons over the entire ring.
The six p orbitals in benzene interact to form a continuous π system above and below the plane of the ring.
Drawing Resonance Structures
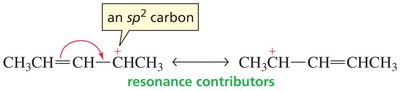
When drawing resonance structures, electrons typically flow from π bonds or lone pairs to sp2-hybridized atoms. Atoms with sp3 hybridization cannot participate in resonance without breaking a bond.
Resonance contributors are drawn using curved arrows to indicate electron movement.

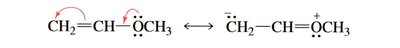
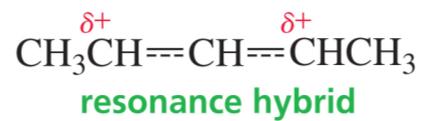
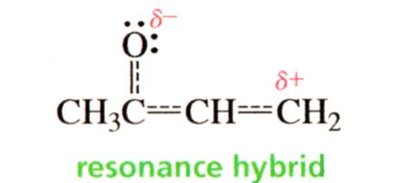
Delocalization and Stability
Resonance Stabilization
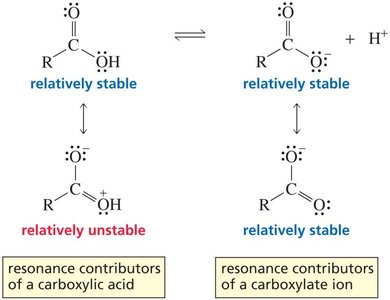
The presence of resonance generally increases the stability of a molecule. The actual structure (resonance hybrid) is more stable than any individual resonance contributor. The stabilization energy due to resonance is called delocalization energy or resonance energy.
Greater resonance stabilization occurs when there are more stable resonance contributors.
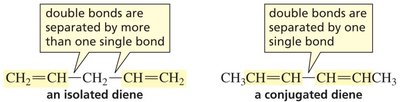
Stability of Dienes
Conjugated dienes (double bonds separated by one single bond) are more stable than isolated dienes (double bonds separated by more than one single bond) due to delocalization of π electrons.
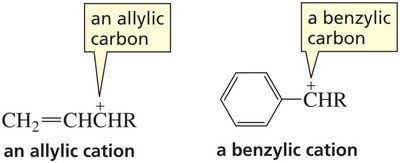
Stability of Carbocations
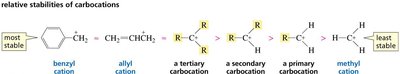
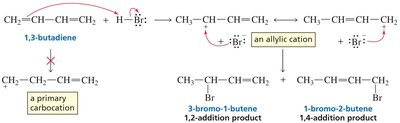
Carbocations are stabilized by resonance. Allylic and benzylic carbocations are especially stable because the positive charge is delocalized over multiple atoms.
Primary resonance-stabilized carbocations can be as stable as tertiary non-resonance-stabilized carbocations.
Delocalization and Acidity (pKa)
Resonance Effects on Acidity
Resonance can significantly affect the acidity of organic compounds. For example, acetic acid is much more acidic than ethanol because its conjugate base (acetate ion) is resonance-stabilized, while the ethoxide ion is not.

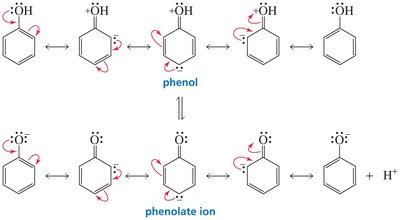
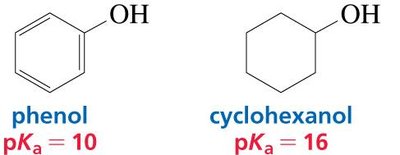
Phenol vs. Cyclohexanol
Phenol is more acidic than cyclohexanol because the phenolate ion (conjugate base of phenol) is resonance-stabilized, while the cyclohexanolate ion is not.
Protonated Aniline vs. Protonated Cyclohexylamine
Protonated aniline is a much stronger acid than protonated cyclohexylamine due to the resonance stabilization of the aniline conjugate base.

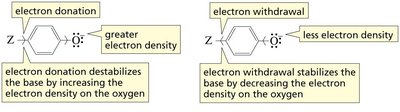
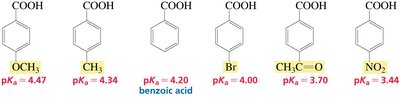
Electronic Effects and pKa
Electron-withdrawing groups stabilize a base (conjugate base), increasing the acidity of the parent acid. Electron-donating groups destabilize the base, decreasing acidity.
Electron donation increases electron density, destabilizing the base.
Electron withdrawal decreases electron density, stabilizing the base.
Delocalization and Reaction Products
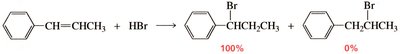
Product Distribution in Electrophilic Addition
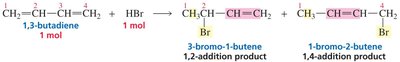
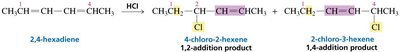
The distribution of products in reactions involving conjugated systems can be explained by resonance stabilization of intermediates. For example, in the addition of HX to conjugated dienes, both 1,2- and 1,4-addition products can form due to resonance-stabilized carbocation intermediates.
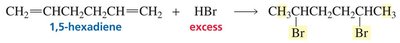
Benzene and Aromaticity
Definition and Criteria for Aromaticity
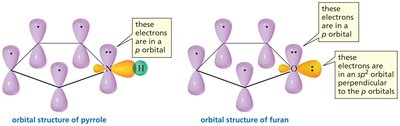
Aromatic compounds are cyclic, planar molecules with a continuous ring of p orbitals and an odd number of pairs of π electrons (4n+2 rule, where n is an integer). Aromatic compounds are exceptionally stable and do not undergo typical alkene reactions.
Every atom in the ring must have a p orbital (sp2-hybridized).
The ring must be planar to allow for continuous overlap of p orbitals.
Examples include benzene, pyrrole, and furan (which contain heteroatoms with lone pairs in p orbitals).
Electrophilic Aromatic Substitution (EAS)
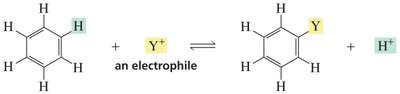
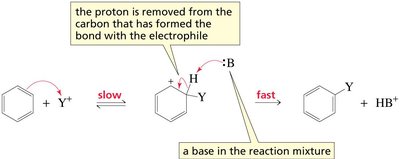
The characteristic reaction of benzene and other aromatic compounds is electrophilic aromatic substitution (EAS). In EAS, an electrophile replaces a hydrogen atom on the aromatic ring. The reaction proceeds via a two-step mechanism: addition of the electrophile to form a carbocation intermediate, followed by deprotonation to restore aromaticity.
Common Electrophilic Aromatic Substitution Reactions
Name | Reagents | Electrophile (or equivalent) | Product | |
|---|---|---|---|---|
Halogenation | X2 + FeX3 (cat.) | X+ | Ar-X | |
Nitration | HNO3 + H2SO4 | NO2+ | Ar-NO2 | |
Sulfonation | SO3 + H2SO4 | SO3H+ | Ar-SO3H | |
Friedel-Crafts Alkylation | RCl + AlCl3 (cat.) | R+ | Ar-R | |
Friedel-Crafts Acylation | RCOCl + AlCl3 (cat.) | RCO+ | Ar-COR |
