 Back
BackElectronic Structure and Bonding: Foundations of Organic Chemistry
Study Guide - Smart Notes

Electronic Structure and Bonding (Part 1)
Introduction to Organic Chemistry
Organic chemistry is the study of carbon-containing molecules and their reactions. It explores how molecules interact, how bonds are broken and formed, and the importance of organic compounds in everyday life, including food, pharmaceuticals, and biological systems.
Atoms are the fundamental units of matter, consisting of a nucleus (protons and neutrons) and electrons in orbitals.
Valence electrons are the outermost electrons involved in bonding.
Bonding can be ionic (electron transfer) or covalent (electron sharing).
Atomic Structure and Orbitals
Atoms are composed of a nucleus (protons and neutrons) and electrons that reside in orbitals outside the nucleus. The arrangement of electrons in these orbitals determines the chemical properties of the atom.
Atomic orbitals (s, p, d, f) are regions of space where electrons are likely to be found.
Electron configuration describes the distribution of electrons among orbitals.
Valence electrons are crucial for chemical bonding and reactivity.
Bond Formation
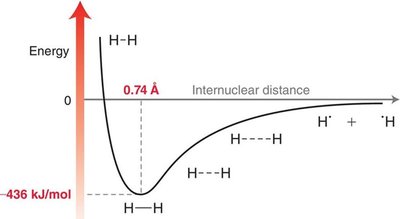
Covalent bonds form when two atoms share a pair of electrons, resulting in a stable arrangement. The optimal bond length is determined by the balance of attractive and repulsive forces between nuclei and electrons.
Attractive forces: Between nuclei and electrons.
Repulsive forces: Between like charges (nuclei-nuclei, electron-electron).
Bond energy: The energy required to break a bond.
The Structural Theory of Matter and Isomerism
The properties of a compound are determined not just by its formula, but by the specific arrangement of its atoms. Compounds with the same molecular formula but different connectivity are called constitutional isomers.
Constitutional isomers: Same formula, different connectivity.
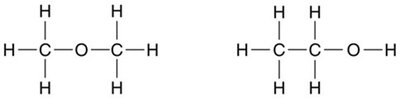
Lewis Structures and the Octet Rule
Lewis structures represent valence electrons as dots around atomic symbols. The octet rule states that main group elements tend to form bonds to achieve eight electrons in their valence shell.
Steps for drawing Lewis structures:
Write the skeletal structure (least electronegative atom central).
Count total valence electrons.
Distribute electrons to form bonds and complete octets.
Form multiple bonds if necessary to satisfy octet rule.
Formal charge calculation:
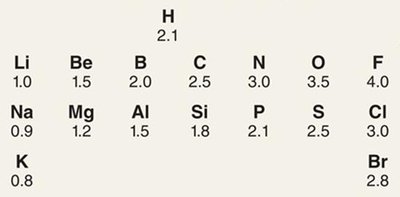
Electronegativity and Bond Polarity
Electronegativity is the ability of an atom to attract shared electrons. The difference in electronegativity between two atoms determines bond polarity:
Nonpolar covalent: Electronegativity difference < 0.5
Polar covalent: 0.5 < difference < 1.7
Ionic: Difference > 1.7
Bond-Line Structures
Bond-line structures are simplified representations where carbon atoms are implied at the ends and vertices of lines, and hydrogen atoms attached to carbon are not shown. Heteroatoms and their hydrogens are explicitly drawn.
Electronic Structure and Bonding (Part 2)
Atomic Orbitals and Quantum Mechanics
Quantum mechanics describes electrons as wavefunctions. Atomic orbitals (s, p, d, f) are regions where electrons are likely to be found, with specific shapes and orientations.
s orbital: Spherical shape.
p orbitals: Dumbbell-shaped, oriented along x, y, z axes.
Node: Region where the probability of finding an electron is zero.
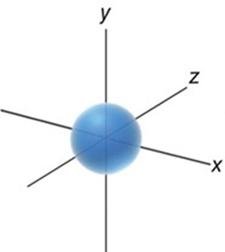
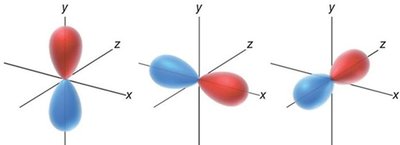

Filling Atomic Orbitals
Electrons fill orbitals according to three principles:
Aufbau principle: Fill lowest energy orbitals first.
Pauli exclusion principle: Maximum two electrons per orbital, with opposite spins.
Hund’s rule: One electron per degenerate orbital before pairing.
Valence Bond Theory
Covalent bonds form when atomic orbitals overlap. Constructive interference of wavefunctions leads to bond formation, while destructive interference leads to nodes and no bond.
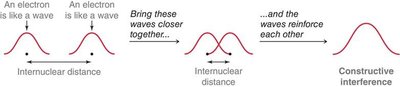

Sigma (σ) and Pi (π) Bonds
There are two main types of covalent bonds:
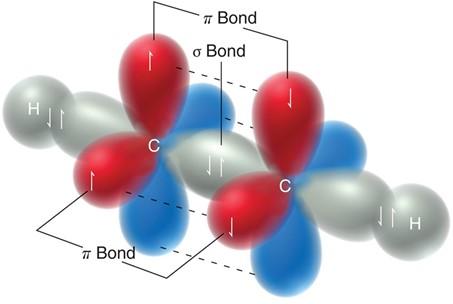
Sigma (σ) bond: Formed by head-on overlap of orbitals; present in all single bonds.
Pi (π) bond: Formed by side-by-side overlap of p orbitals; present in double and triple bonds.
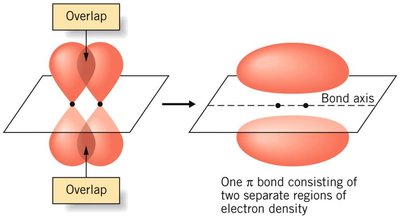
Hybridization
Hybridization explains the observed shapes and bond angles in molecules by mixing atomic orbitals to form new, equivalent hybrid orbitals.
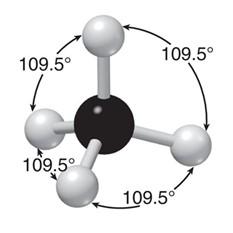
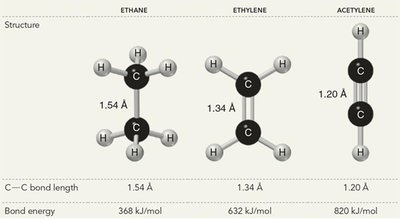
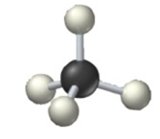
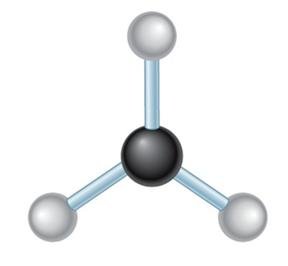
sp3 hybridization: Four equivalent orbitals, tetrahedral geometry, 109.5° bond angles (e.g., methane, CH4).
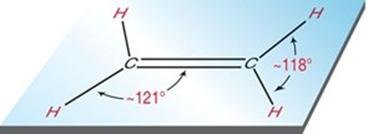
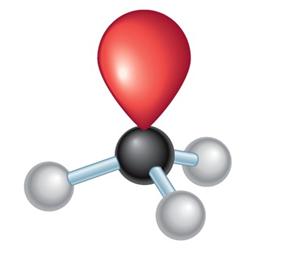
sp2 hybridization: Three equivalent orbitals, trigonal planar geometry, 120° bond angles (e.g., ethene, C2H4).
sp hybridization: Two equivalent orbitals, linear geometry, 180° bond angles (e.g., acetylene, C2H2).
Bond Strength and Bond Length
The strength and length of a bond depend on the type of bond and the s-character of the hybrid orbitals involved:
Sigma bonds are stronger than pi bonds.
Triple bonds (one σ, two π) are stronger and shorter than double or single bonds.
Higher s-character (sp > sp2 > sp3) leads to shorter, stronger bonds.
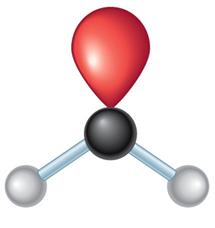
Molecular Geometry: VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the 3D shapes of molecules based on the repulsion between electron pairs around a central atom.
Steric Number | Hybridization | Electron Pair Geometry | Molecular Geometry | Bond Angles |
|---|---|---|---|---|
4 | sp3 | Tetrahedral | Tetrahedral | 109.5° |
3 | sp2 | Trigonal planar | Trigonal planar | 120° |
2 | sp | Linear | Linear | 180° |
Electronic Structure and Bonding (Part 3)
Dipole Moments and Molecular Polarity
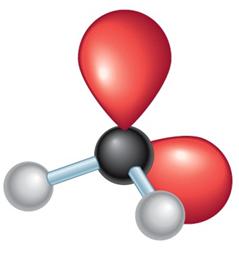
Electronegativity differences in bonds create dipole moments, which are vectors indicating the direction and magnitude of charge separation. The overall molecular dipole is the vector sum of all bond dipoles and lone pair contributions.
Polar molecules have a net dipole moment.
Nonpolar molecules have bond dipoles that cancel out.
Intermolecular Forces and Physical Properties
The physical properties of organic compounds are determined by intermolecular forces:
Dipole-dipole interactions: Attraction between polar molecules.
Hydrogen bonding: Strong dipole-dipole interaction involving H bonded to N, O, or F.
London dispersion forces: Weak, transient attractions due to temporary dipoles in all molecules, especially significant in large, nonpolar molecules.
Hydrogen Bonding: Protic and Aprotic Solvents
Solvents are classified based on their ability to form hydrogen bonds:
Protic solvents: Contain H bonded directly to O or N (e.g., water, ethanol).
Aprotic solvents: Do not have H bonded to O or N (e.g., acetone, DMSO).
London Dispersion Forces: Polarizability and Surface Area
London forces increase with:
Polarizability: Larger atoms/molecules are more easily polarized, leading to stronger dispersion forces.
Surface area: Larger, less branched molecules have more surface area for interactions, resulting in higher boiling points.
Solubility: "Like Dissolves Like"
Polar compounds dissolve well in polar solvents, and nonpolar compounds dissolve well in nonpolar solvents. This principle is essential for understanding solubility and extraction in organic chemistry.
Summary Table: Bond Types and Properties
Bond Type | Number of σ-bonds | Number of π-bonds | Bond Length | Bond Strength |
|---|---|---|---|---|
Single (C–C) | 1 | 0 | 1.54 Å | 368 kJ/mol |
Double (C=C) | 1 | 1 | 1.34 Å | 632 kJ/mol |
Triple (C≡C) | 1 | 2 | 1.20 Å | 820 kJ/mol |

Additional info: This guide covers foundational concepts in organic chemistry, including atomic structure, bonding, molecular geometry, and intermolecular forces, providing a basis for understanding reactivity and properties of organic molecules.
