 Back
BackElectrophilic Aromatic Substitution and Substituent Effects in Aromatic Chemistry
Study Guide - Smart Notes

Electrophilic Aromatic Substitution (EAS)
Introduction to EAS
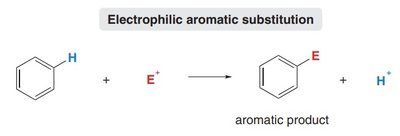
Electrophilic aromatic substitution (EAS) is a fundamental reaction in organic chemistry, allowing the functionalization of aromatic rings such as benzene. In EAS, an aromatic hydrogen is replaced by an electrophile, while the aromaticity of the ring is preserved.
Aromatic rings are highly stable due to delocalized π electrons.
These π electrons are nucleophilic and react with electrophiles (E+).
The general reaction: aromatic ring + E+ → substituted aromatic ring + H+
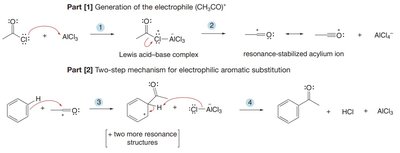
General Mechanism of EAS
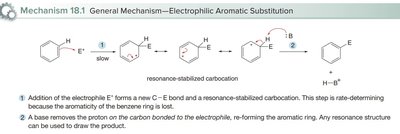
All EAS reactions proceed via a two-step mechanism:
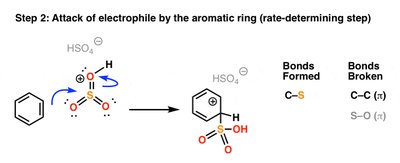
Addition of the electrophile (E+): The aromatic ring attacks the electrophile, forming a resonance-stabilized carbocation (arenium ion). This is the rate-determining step because aromaticity is temporarily lost.
Deprotonation: A base removes a proton from the carbon bonded to the electrophile, restoring aromaticity.
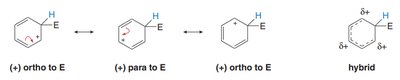
The positive charge in the intermediate is delocalized to the ortho and para positions relative to the new substituent.
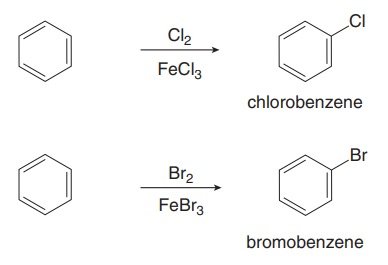
Halogenation of Benzene
Mechanism and Catalysis
Benzene reacts with chlorine or bromine in the presence of a Lewis acid catalyst (FeCl3 or FeBr3) to form aryl halides. Iodine and fluorine are not typically used due to reactivity issues.
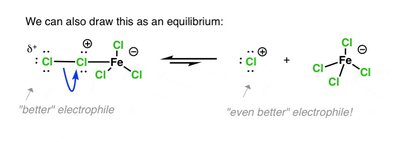
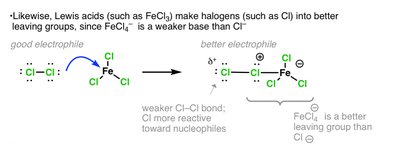
Lewis acid activates the halogen, making it a better electrophile.
The aromatic ring attacks the activated halogen, forming a resonance-stabilized carbocation.
Deprotonation restores aromaticity and regenerates the catalyst.
Activation of Halogen by Lewis Acid
The Lewis acid forms a complex with the halogen, polarizing the X–X bond and making one halogen atom more electrophilic.
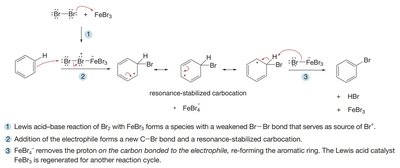
Detailed Mechanism Example: Bromination
The mechanism involves three main steps:
Lewis acid–base reaction forms a Br2–FeBr3 complex.
The aromatic ring attacks Br+, forming a resonance-stabilized carbocation.
Deprotonation by FeBr4– restores aromaticity and regenerates FeBr3.
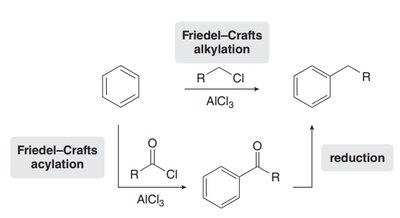
Friedel–Crafts Alkylation and Acylation
Friedel–Crafts Alkylation
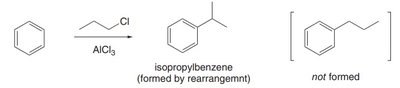
This reaction forms new C–C bonds by introducing an alkyl group onto the aromatic ring using an alkyl chloride and AlCl3 as a catalyst.
Primary and secondary alkyl halides may rearrange via carbocation shifts (1,2-hydride or 1,2-methyl shifts).
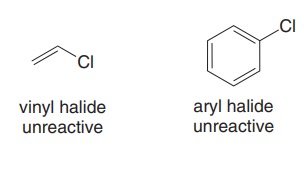
Vinyl and aryl halides are unreactive in this reaction.
Friedel–Crafts Acylation
Acylation introduces an acyl group (RCO–) onto the aromatic ring using an acyl chloride and AlCl3. The acylium ion (R–C≡O+) is the active electrophile.

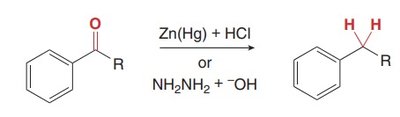
Overcoming Carbocation Rearrangement
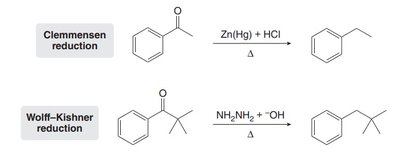
To avoid carbocation rearrangement in alkylation, a two-step process is used: acylation followed by reduction (Clemmensen or Wolff–Kishner reduction).
Reactions at the Benzylic Position
Oxidation
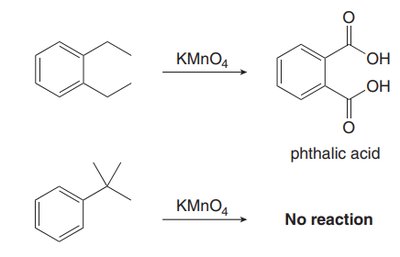
Alkyl groups on the benzene ring can be oxidized to carboxylic acids using KMnO4, provided there is at least one benzylic hydrogen.
Bromination
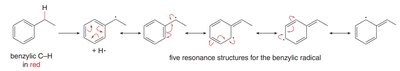
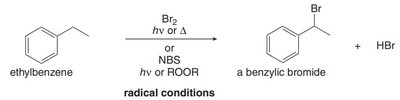
Bromination at the benzylic position occurs under radical conditions (Br2 or NBS with light or peroxide), not under ionic conditions.
Nitration and Sulfonation of Benzene
Nitration
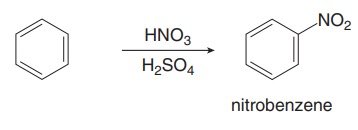
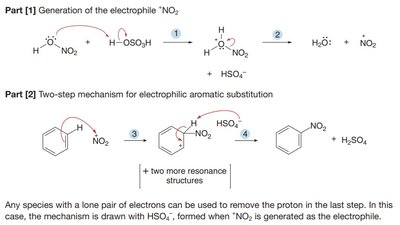
Nitration introduces a nitro group (NO2) onto the aromatic ring using HNO3 and H2SO4. The active electrophile is the nitronium ion (NO2+).
Reduction of Nitro Groups
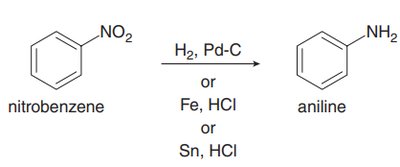
Nitro groups can be reduced to amines (NH2) using catalytic hydrogenation or metal/acid reduction.
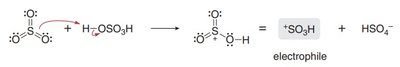
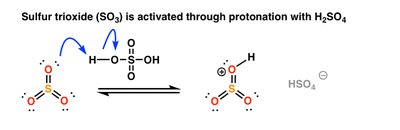
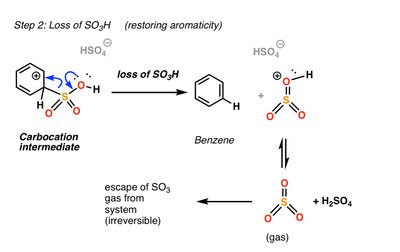
Sulfonation
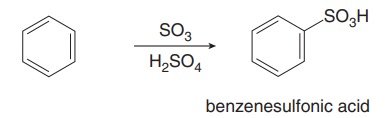
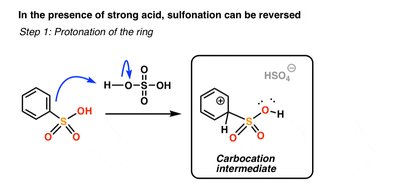
Sulfonation introduces a sulfonic acid group (SO3H) onto the aromatic ring using SO3 and H2SO4. The reaction is reversible under acidic conditions with heat and water.
Substituent Effects in Electrophilic Aromatic Substitution
Inductive and Resonance Effects
Substituents on the benzene ring influence both the reactivity and the regioselectivity of EAS reactions through inductive and resonance effects.
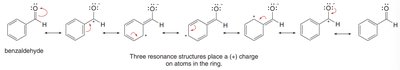
Electron-withdrawing groups (EWG) decrease electron density, deactivating the ring and often directing new substituents to the meta position.
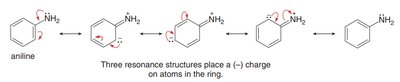
Electron-donating groups (EDG) increase electron density, activating the ring and directing new substituents to the ortho and para positions.


Classification of Substituents
Substituents are classified based on their effects:
EDG: Alkyl, neutral N or O with lone pairs (activate, ortho/para-directing)
EWG: Halogens, groups with positive charge or partial positive charge (deactivate, meta-directing except halogens, which are ortho/para-directing but deactivating)
Summary Table: Substituent Effects
The following table summarizes the effects of common substituents on reactivity and regioselectivity in EAS:
Substituent | Inductive effect | Resonance effect | Reactivity | Directing effect |
|---|---|---|---|---|
R = alkyl | donating | none | activating | ortho, para |
Z = N or O | withdrawing | donating | activating | ortho, para |
X = halogen | withdrawing | donating | deactivating | ortho, para |
Y (δ+ or +) | withdrawing | withdrawing | deactivating | meta |
Additional info: Understanding the interplay of inductive and resonance effects is crucial for predicting the outcome of EAS reactions and for designing synthetic routes in aromatic chemistry.
