 Back
BackEthers, Epoxides, and Sulfides: Structure, Synthesis, and Reactions
Study Guide - Smart Notes

Ethers, Epoxides, and Sulfides
Structure and Properties of Ethers
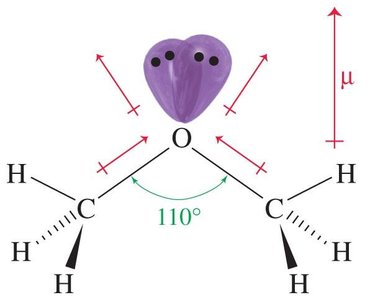
Ethers are organic compounds with the general formula R—O—R', where R and R' are alkyl or aryl groups. They can be symmetrical (both groups identical) or unsymmetrical. The oxygen atom in ethers is sp3 hybridized, resulting in a bent molecular geometry with a C—O—C bond angle of approximately 110°. Ethers possess polar C—O bonds and a dipole moment of about 1.3 D, contributing to their moderate polarity.
Symmetrical ethers: Both R groups are the same (e.g., diethyl ether).
Unsymmetrical ethers: R and R' are different (e.g., t-butyl methyl ether).
Polarity: Ethers are less polar than alcohols but more polar than alkanes.
Nomenclature of Ethers
Ethers are named using both common and IUPAC systems. In the common system, the two alkyl groups attached to oxygen are named in alphabetical order, followed by the word 'ether.' In the IUPAC system, the larger group is named as the parent hydrocarbon, and the smaller group attached to oxygen is named as an 'alkoxy' substituent.
Common names: diethyl ether, t-butyl methyl ether
IUPAC names: 2-methoxy-2-methylpropane, methoxycyclohexane
Spectroscopic Analysis of Ethers
IR Spectroscopy
The C—O stretch in ethers appears in the fingerprint region (1000–1200 cm-1), but this region is not unique to ethers, limiting its usefulness for identification.
Mass Spectrometry (MS)
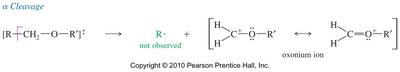
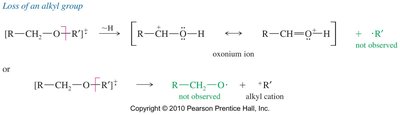
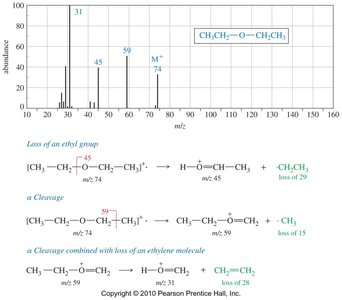
Ethers undergo characteristic fragmentation in MS, primarily through α-cleavage to form resonance-stabilized oxonium ions. The loss of alkyl groups and cleavage of C—O bonds are common fragmentation pathways.
NMR Spectroscopy
13C NMR: C—O signal between δ 65–90
1H NMR: H—C—O signal between δ 3.5–4
Synthesis of Ethers
Williamson Ether Synthesis
The Williamson ether synthesis is a classic method for preparing ethers. It involves an SN2 attack of an alkoxide ion on an unhindered primary (or secondary) alkyl halide or tosylate. The alkoxide is typically generated by treating an alcohol with sodium, potassium, or sodium hydride.
Reaction:
Limitations: Not suitable for tertiary halides due to elimination side reactions.
Phenyl Ethers
Phenoxide ions are easily produced due to the acidity of the phenol proton. However, phenyl halides or tosylates cannot be used in the Williamson synthesis because SN2 reactions do not occur on sp2 carbons.
Alkoxymercuration–Demercuration
This method uses mercuric acetate and an alcohol to form ethers. The alcohol attacks the more substituted carbon of the mercurinium ion intermediate, followed by reduction with sodium borohydride.
Reactivity and Cleavage of Ethers
Ethers are generally unreactive, making them ideal solvents. However, they can be cleaved by heating with strong acids such as HBr or HI. The reactivity order is HI > HBr. The cleavage mechanism involves protonation of the oxygen, followed by nucleophilic attack by the halide ion (SN2 or SN1, depending on the substrate).
Phenyl Ether Cleavage
Phenol cannot react further to become a halide because SN2 reactions cannot occur on sp2 carbons.
Autoxidation of Ethers
Ethers can slowly oxidize in the presence of atmospheric oxygen to form hydroperoxides and dialkyl peroxides, which are highly explosive. Precautions include not distilling to dryness and storing in brown bottles.
Sulfides (Thioethers) and Thiols
Sulfides (thioethers) are analogs of ethers with the formula R—S—R'. They are named similarly to ethers, replacing 'ether' with 'sulfide' in common names or 'alkoxy' with 'alkylthio' in IUPAC names. Sulfides are easily synthesized by the Williamson method, using thiolates as nucleophiles.

Sulfide Reactions
Oxidation: Sulfides are oxidized to sulfoxides and sulfones.
Alkylation: Sulfides react with alkyl halides to form sulfonium salts.
Synthesis of Epoxides
Epoxidation of Alkenes
Epoxides are cyclic ethers formed by the reaction of alkenes with peroxyacids, most commonly meta-chloroperoxybenzoic acid (MCPBA). The reaction is carried out in aprotic solvents to prevent ring opening.
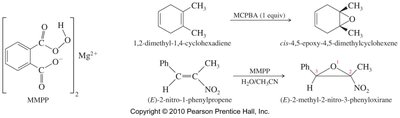
Halohydrin Cyclization
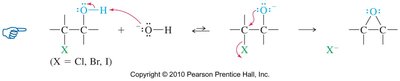
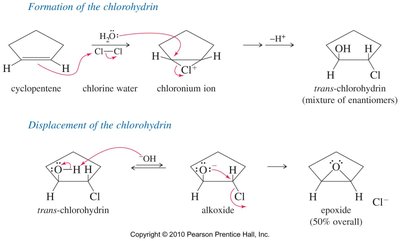
Epoxides can also be synthesized by treating halohydrins with base, resulting in internal SN2 ring closure.
Reactions of Epoxides
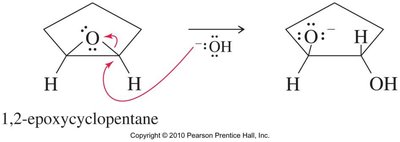
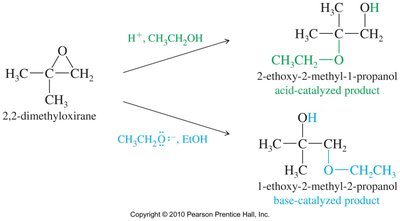
Acid-Catalyzed Ring Opening
Acid-catalyzed hydrolysis of epoxides yields glycols with anti stereochemistry due to backside attack by water on the protonated epoxide. Alcohols can also act as nucleophiles, producing alkoxy alcohols with anti stereochemistry.
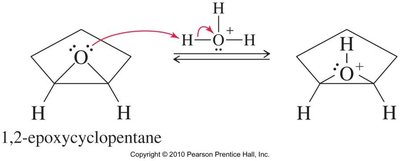
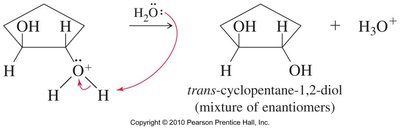
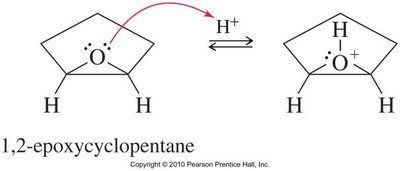
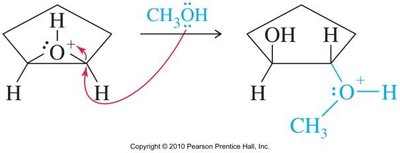
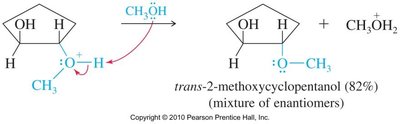
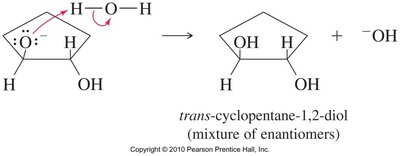
Base-Catalyzed Ring Opening
Hydroxide ions attack and open the epoxide ring, yielding diols after protonation. The release of ring strain makes this reaction thermodynamically favored.
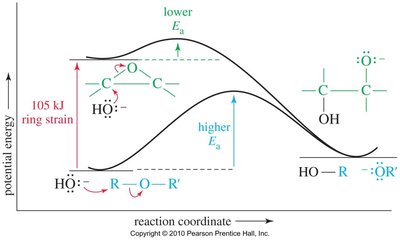
Ring Strain and Thermodynamics
Epoxides are higher in energy than acyclic ethers due to ring strain (~25 kcal/mol). Ring opening is favored thermodynamically.
Regioselectivity and Stereochemistry in Epoxide Reactions
Epoxidation is selective for the most electron-rich double bond. Nucleophilic ring opening occurs at the less hindered carbon, especially with Grignard and organolithium reagents.
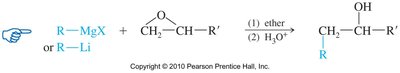
Epoxide Ring Opening with Grignard and Organolithium Reagents
These nucleophiles attack the less hindered carbon of the epoxide, resulting in alcohols after hydrolysis.
Summary Table: Ether, Epoxide, and Sulfide Reactions
Compound | Synthesis | Key Reactions | Notes |
|---|---|---|---|
Ether | Williamson, Alkoxymercuration | Acid cleavage, Autoxidation | Good solvent, limited reactivity |
Epoxide | Peroxyacid, Halohydrin cyclization | Acid/base ring opening, Grignard addition | High ring strain, anti stereochemistry |
Sulfide | Williamson with thiolate | Oxidation, Alkylation | Analog of ether, forms sulfoxides/sulfones |
Additional info: Academic context was added to clarify mechanisms, nomenclature, and spectroscopic details, and to ensure completeness for exam preparation.
