 Back
BackEthers, Epoxides, and Thioethers: Structure, Properties, and Reactions
Study Guide - Smart Notes

Ethers, Epoxides, and Thioethers
Structure and Nomenclature of Ethers
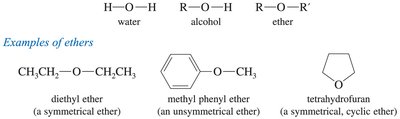
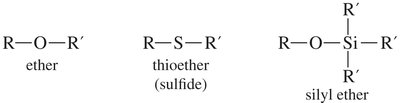
Ethers are organic compounds with the general formula R—O—R′, where R and R′ can be alkyl or aryl groups. Ethers can be symmetrical (both groups identical) or unsymmetrical (different groups). They are widely used as solvents due to their ability to dissolve both polar and nonpolar substances and their chemical inertness toward strong bases.
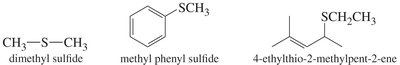
Symmetrical Ethers: Both R groups are the same (e.g., diethyl ether).
Unsymmetrical Ethers: R and R′ are different (e.g., methyl phenyl ether).
Cyclic Ethers: Oxygen is part of a ring structure (e.g., tetrahydrofuran, dioxane).
IUPAC Naming: The smaller group attached to oxygen is named as an "alkoxy" substituent on the larger alkane chain.
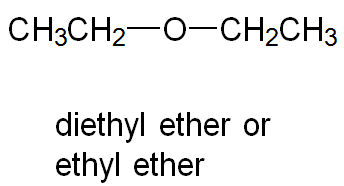
Physical Properties of Ethers
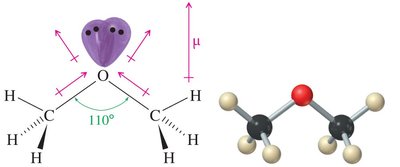
Ethers exhibit unique physical properties due to their molecular structure. The oxygen atom is sp3 hybridized, resulting in a bent geometry with a tetrahedral C—O—C angle of approximately 110°. Ethers possess polar C—O bonds but cannot form hydrogen bonds with themselves, leading to lower boiling points compared to alcohols of similar molecular weight.
Boiling Point: Lower than alcohols due to lack of hydrogen bonding between ether molecules.
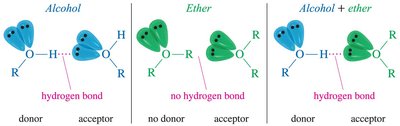
Hydrogen Bonding: Ethers are hydrogen bond acceptors, not donors. They can form hydrogen bonds with water and alcohols.
Solubility: Ethers can dissolve both polar and nonpolar substances.
Boiling Points and Density Comparison
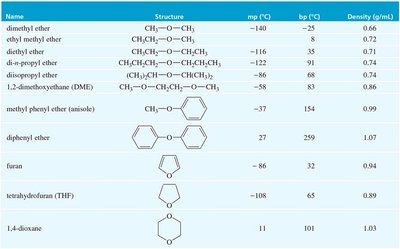
The boiling points and densities of various ethers, alcohols, and alkanes are compared in the table below:
Name | Structure | mp (°C) | bp (°C) | Density (g/mL) |
|---|---|---|---|---|
Dimethyl ether | CH3—O—CH3 | -140 | -25 | 0.66 |
Diethyl ether | CH3CH2—O—CH2CH3 | -116 | 35 | 0.71 |
1,2-dimethoxyethane (DME) | CH3—O—CH2CH2—O—CH3 | -58 | 82 | 0.87 |
Tetrahydrofuran (THF) | Cyclic | -108 | 65 | 0.89 |
1,4-dioxane | Cyclic | 11 | 101 | 1.03 |
Methyl phenyl ether (anisole) | Ph—O—CH3 | -37 | 154 | 0.99 |
Solvation and Solvent Properties
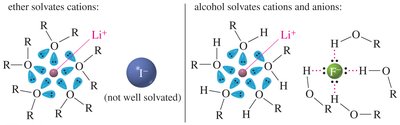
Ethers are excellent solvents for many organic reactions because they are relatively unreactive and can solvate cations effectively. However, they do not solvate anions as efficiently as alcohols due to their inability to donate hydrogen bonds.
Solvation of Cations: Ethers solvate cations (e.g., Li+) via lone pairs on oxygen.
Solvation of Anions: Alcohols can solvate both cations and anions due to hydrogen bonding.
Complexes with Ethers
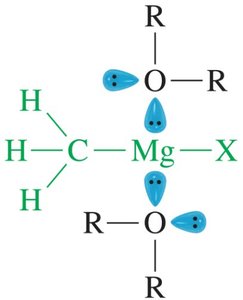
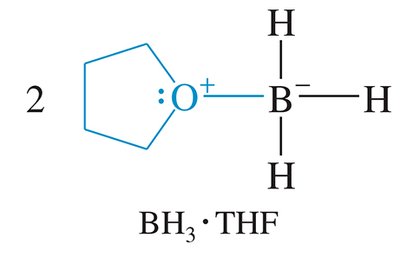
Ethers stabilize reactive species in solution, such as Grignard reagents and borane complexes, by coordinating through their lone pairs.
Grignard Reagents: Ethers stabilize the reagent and keep it in solution.
Borane Complexes: Ethers stabilize borane (BH3).
Crown Ethers
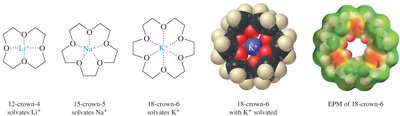
Crown ethers are cyclic polyethers that can selectively complex metal cations in the center of their ring. The size of the crown ether determines which cation it can solvate most effectively, enabling the dissolution of polar inorganic salts in nonpolar organic solvents.
12-crown-4: Solvates Li+
15-crown-5: Solvates Na+
18-crown-6: Solvates K+
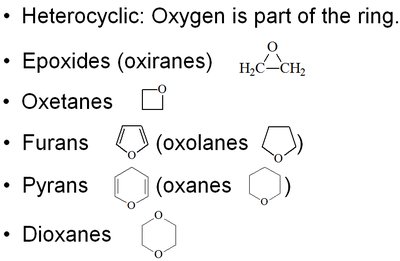
Cyclic Ethers and Epoxides
Cyclic ethers include various ring sizes, such as epoxides (three-membered rings), oxetanes (four-membered), furans (five-membered), pyrans (six-membered), and dioxanes (six-membered with two oxygens). Epoxides are highly reactive due to ring strain.
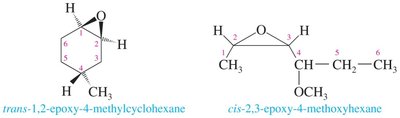
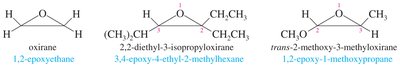
Nomenclature of Epoxides
Epoxides are named by adding "oxide" to the parent alkene or by treating the oxygen as an "epoxy" substituent. The three-membered oxirane ring is the parent structure, with oxygen as position 1 and carbons as 2 and 3.
Spectroscopic Properties of Ethers
Infrared (IR) Spectroscopy
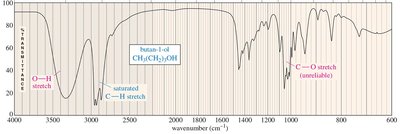
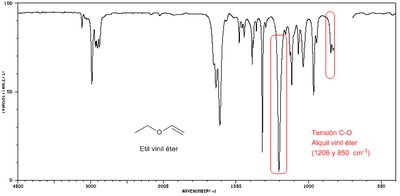
The C—O stretch in ethers appears in the fingerprint region around 1000–1200 cm–1. The absence of C=O or O—H stretches suggests the compound is an ether.
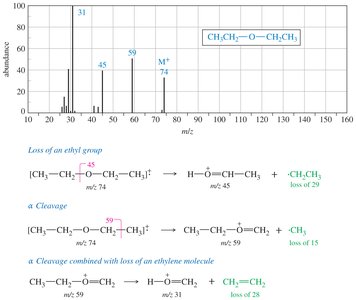
Mass Spectrometry (MS)
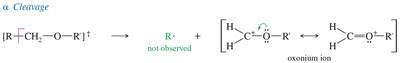
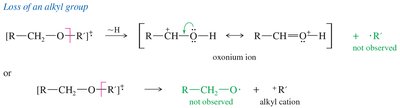
Ethers undergo α-cleavage to form resonance-stabilized oxonium ions. The C—O bond can also be cleaved to produce carbocations.
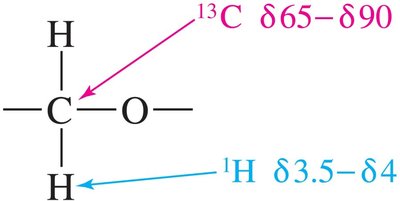
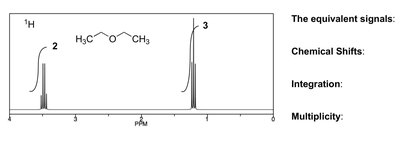
Nuclear Magnetic Resonance (NMR) Spectroscopy
Typical chemical shifts for ethers:
13C—O: δ 65–90
1H—C—O: δ 3.5–4
Synthesis of Ethers
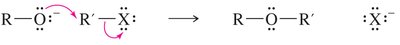
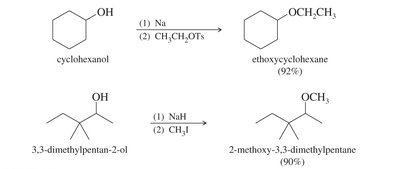
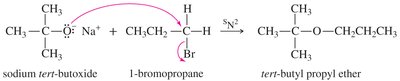
Williamson Ether Synthesis
This laboratory method involves an SN2 attack of an alkoxide ion on an unhindered primary halide or tosylate. Alkoxides are generated by treating alcohols with Na, K, or NaH.
Phenyl Ethers
Phenoxide ions are easily produced due to the acidity of the alcohol proton. However, phenyl halides or tosylates cannot be used in this synthesis.
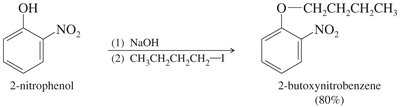
Limitations and Alternative Methods
SN2 reactions cannot occur on tertiary alkyl halides, leading to elimination instead of ether formation. A better synthesis uses the less hindered alkyl group as the SN2 substrate and the alkoxide of the more hindered group.
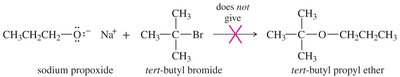
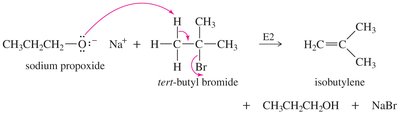
Alkoxymercuration–Demercuration
Mercuric acetate reacts with an alcohol and an alkene to form an ether, with the alcohol attacking the more substituted carbon of the mercurinium ion intermediate.

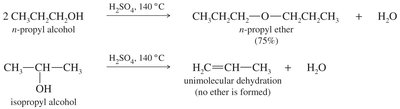
Industrial Synthesis
Bimolecular condensation of alcohols is used industrially but is not suitable for laboratory synthesis. High temperatures favor alkene formation instead of ether.
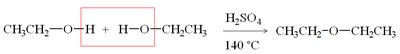
Reactions of Ethers
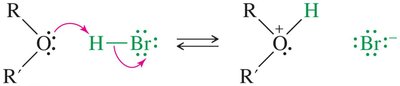
Cleavage by HBr and HI
Ethers are generally unreactive but can be cleaved by heating with concentrated HBr or HI. The mechanism involves protonation of the oxygen, followed by SN2 attack by the halide.
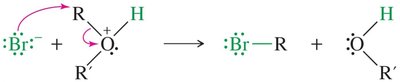
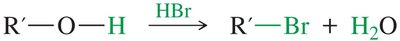
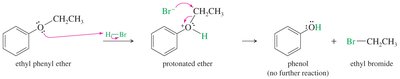
Phenyl Ether Cleavage
Phenol cannot react further to become a halide because SN2 reactions cannot occur on sp2 carbons.
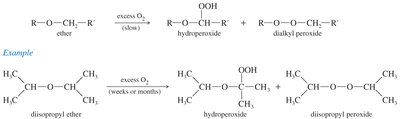
Autoxidation of Ethers
Ethers slowly oxidize in the presence of atmospheric oxygen to form hydroperoxides and dialkyl peroxides, which are highly explosive. Precautions include not distilling to dryness and storing in tightly capped bottles.
Thioethers (Sulfides)
Thioethers are sulfur analogs of ethers with the general formula R—S—R′. They are named similarly to ethers, using "sulfide" in the common name or "alkylthio" in the IUPAC system. Thioethers are easily synthesized by the Williamson ether synthesis using a thiolate ion as the nucleophile.
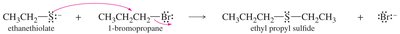
Reactions of Sulfides
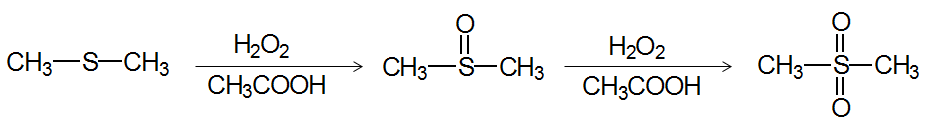
Sulfides are easily oxidized to sulfoxides and sulfones. They also react with unhindered alkyl halides to give sulfonium salts and are used as mild reducing agents.
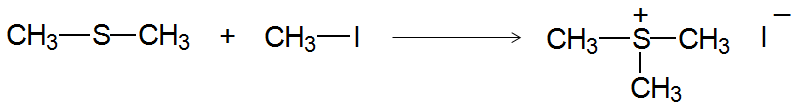

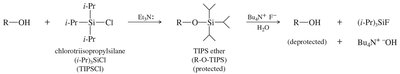
Silyl Ethers as Protecting Groups
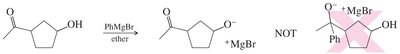
Silyl ethers are used as protecting groups for alcohols in organic synthesis. They are resistant to some acids, bases, and oxidizing agents, and can be easily formed and hydrolyzed. Protecting the alcohol as a silyl ether ensures selective reactions, such as Grignard additions to carbonyls.
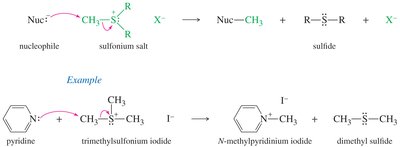
Sulfonium Salts as Alkylating Agents
Sulfonium salts are used as alkylating agents because the leaving group formed is neutral, facilitating nucleophilic substitution reactions.
Epoxides: Synthesis and Reactions
Synthesis of Epoxides
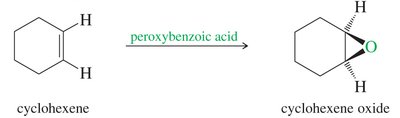
Peroxyacid Oxidation: Alkenes are converted to epoxides using peroxyacids such as meta-chloroperoxybenzoic acid (MCPBA). The most electron-rich double bond reacts faster, allowing selective epoxidation.
Halohydrin Cyclization: Treatment of a halohydrin with base leads to epoxide formation via internal SN2 attack.

Reactions of Epoxides
Acid-Catalyzed Ring Opening: Protonation of the oxygen followed by nucleophilic attack opens the ring, producing trans-1,2-diols.
Base-Catalyzed Ring Opening: Hydroxide ion attacks and opens the ring, yielding diols after protonation.
Ring Opening with Hydrohalic Acids: Halide ion attacks the protonated epoxide, forming halohydrins and then 1,2-dihalides.
Reaction with Grignard and Organolithium Reagents: Strong bases open the epoxide ring by attacking the less hindered carbon.
Epoxides are more reactive than acyclic ethers due to ring strain (~25 kcal/mol), making ring opening thermodynamically favored.
Summary Table: Ether, Epoxide, and Thioether Properties
Compound | Structure | Key Properties |
|---|---|---|
Ether | R—O—R′ | Polar, hydrogen bond acceptor, solvent, low reactivity |
Epoxide | Three-membered ring | High ring strain, reactive, forms diols |
Thioether | R—S—R′ | Sulfur analog, easily oxidized, reducing agent |
Silyl Ether | R—O—Si—R′ | Protecting group for alcohols |
Additional info: These notes expand on the original slides by providing definitions, examples, and academic context for each topic, ensuring completeness and clarity for exam preparation.
