 Back
BackEthers, Epoxides, and Thioethers: Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Ethers, Epoxides, and Thioethers
Introduction
Ethers, epoxides, and thioethers are important classes of organic compounds characterized by the presence of oxygen or sulfur atoms bonded to carbon. Their unique structures and reactivity make them valuable in both biological and industrial contexts. - Ethers have the general formula R—O—R′, where R and R′ are alkyl or aryl groups. - Epoxides are cyclic ethers with a three-membered ring, resulting in significant ring strain and high reactivity. - Thioethers (sulfides) are analogs of ethers, with sulfur replacing the oxygen atom (R—S—R′).
Biological and Industrial Relevance
- Ethers and epoxides are found in natural products and pharmaceuticals. - Thioethers are present in vitamins and cofactors, such as biotin (vitamin B7). 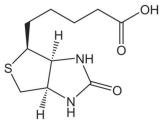
Structure and Bonding
Hybridization and Bond Angles
Both water (H₂O) and ethers (R–O–R’) have sp³ hybridized oxygen atoms. The presence of bulky alkyl groups in ethers increases the C–O–C bond angle due to steric effects. - Bond angle: Larger alkyl groups lead to wider bond angles in ethers compared to water.
Physical Properties of Ethers
Polarity and Hydrogen Bonding
Ethers are polar compounds due to the electronegative oxygen atom, but they cannot form hydrogen bonds with each other. - Hydrogen bond acceptors: Ethers can accept hydrogen bonds from water and alcohols. - Boiling point: Ethers have lower boiling points than alcohols because they lack intermolecular hydrogen bonding. 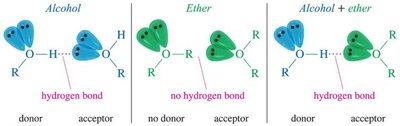
Boiling Points and Solvent Properties
- Ethers with larger alkyl groups have higher boiling points due to increased London dispersion forces. - Ethers are excellent solvents for organic reactions, dissolving both polar and nonpolar substances. - Their low boiling points facilitate easy removal after reactions.
Complex Formation with Metals
Ethers can stabilize metal cations, forming complexes such as Grignard reagents and borane-THF complexes. 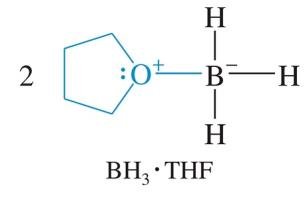
Crown Ethers
Crown ethers are polyethers capable of complexing metal cations within their ring structure, enhancing solubility and reactivity of ionic compounds in nonpolar media. 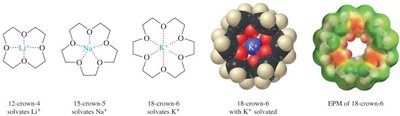
Nomenclature of Ethers
Common Names
- Name the two alkyl groups attached to oxygen in alphabetical order, followed by "ether" (e.g., diethyl ether, methyl tert-butyl ether).
IUPAC Names
- The more complex alkyl group is the root name; the other is named as an "alkoxy" substituent (e.g., methoxyethane).
Nomenclature of Cyclic Ethers
- Cyclic ethers are named based on ring size and heteroatoms. - Three-membered rings: oxirane; four-membered: oxetane; five-membered: oxolane; six-membered: oxane. - Unsaturated cyclic ethers are named as derivatives of pyran or furan.
Nomenclature of Epoxides
- Name the parent alkene and add "oxide" or treat oxygen as a substituent (epoxy) with locants. 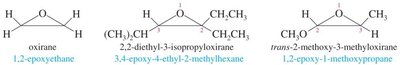
Spectroscopy of Ethers
Infrared (IR) Spectroscopy
- The C—O stretch appears in the fingerprint region (1000–1200 cm–1). - Absence of C=O or OH stretches suggests the compound is an ether. 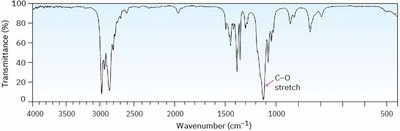
Mass Spectrometry (MS)
- Ethers undergo α-cleavage to form resonance-stabilized oxonium ions.
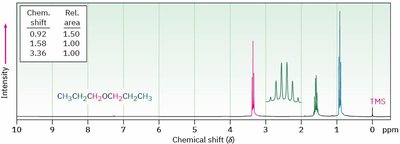
Nuclear Magnetic Resonance (NMR) Spectroscopy
- 13C NMR: C—O signal between δ 65–90. - 1H NMR: H—C—O signal between δ 3.5–4. 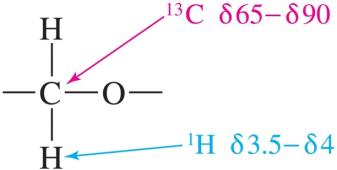
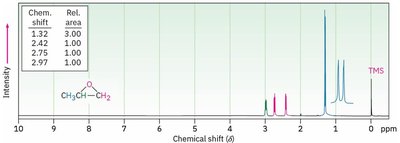
Synthesis of Ethers
Williamson Ether Synthesis
- Involves SN2 attack of an alkoxide ion on a primary alkyl halide or tosylate. - Alkoxide is generated by reacting alcohol with Na, K, or NaH.
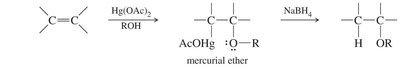
Alkoxymercuration–Demercuration
- Alcohol reacts with a mercurinium ion intermediate, following Markovnikov orientation. 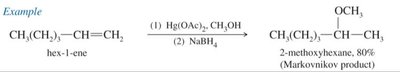
Bimolecular Condensation (Industrial Synthesis)
- Two alcohol molecules condense under acidic conditions to form an ether and water. - At higher temperatures, elimination to form alkene is favored. 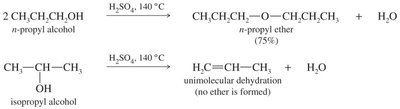
Reactions of Ethers
Cleavage by HX (HBr or HI)
- Ethers are cleaved by heating with concentrated HBr or HI. - Reactivity: HI > HBr. - Mechanism: Protonation, cleavage, and conversion of alcohol fragment to alkyl halide (not for phenols).
Autoxidation
- Ethers oxidize slowly in air to form hydroperoxides and dialkyl peroxides, which are explosive. - Precautions: Avoid distilling to dryness; store in tightly capped bottles.
Thioethers (Sulfides)
Structure and Nomenclature
- Thioethers are ethers with sulfur replacing oxygen. - Common names use "sulfide" instead of "ether"; IUPAC names use "alkylthio" instead of "alkoxy."
Synthesis of Thioethers
- Prepared by Williamson ether synthesis using a thiolate ion as nucleophile.
Reactions of Thioethers
- Oxidation: Sulfides are easily oxidized to sulfoxides and sulfones. - Reaction with Alkyl Halides: Sulfides react with alkyl halides to form sulfonium salts, useful as alkylating agents.
Synthesis and Reactions of Epoxides
Synthesis of Epoxides
- Peroxyacid Method: Alkenes react with peroxyacids (e.g., MCPBA) to form epoxides. 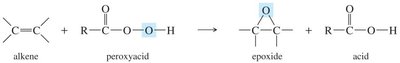
 - Halohydrin Cyclization: Treatment of halohydrin with base leads to epoxide via internal SN2 attack.
- Halohydrin Cyclization: Treatment of halohydrin with base leads to epoxide via internal SN2 attack.
Reactions of Epoxides
- Acid-Catalyzed Opening: Protonation followed by nucleophilic attack at the more substituted carbon. - Base-Catalyzed Opening: Strong nucleophiles attack the less substituted carbon. - Reaction with Grignard and Organolithium Reagents: Strong bases open the epoxide ring, forming new C–C bonds. 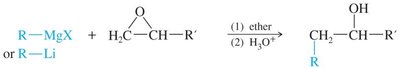
Uses of Epoxides: Epoxy Resins
- Epoxy resins are formed from bisphenol A and epichlorohydrin, used in adhesives and coatings. 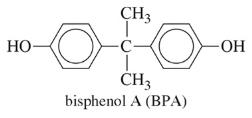
Summary Table
Compound | Synthesis | Reactions |
|---|---|---|
Ether | Williamson, Bimolecular Condensation, Alkoxymercuration–demercuration | Cleavage by HX, Autoxidation |
Thioether | Williamson Method | Oxidation, Reaction with Alkyl Halides |
Epoxide | Alkene with peroxyacid, Halohydrin with base | Acid/Base Catalyzed Ring Opening, Grignard/Organolithium |
Practice Problems and Mechanisms
Epoxide Nomenclature Practice

IR and NMR Spectra Interpretation



Alkoxymercuration–Demercuration Mechanism


Bimolecular Condensation Mechanism

Epoxide Synthesis and Ring Opening


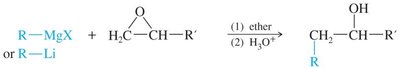
Epoxy Resin Formation

Additional info:
- The notes include expanded academic context for clarity and completeness. - Practice problems and mechanisms are referenced for exam preparation.
