 Back
BackEthers, Epoxides, Thiols, Sulfides, and Disulfides: Structure, Properties, and Reactions
Study Guide - Smart Notes

Ethers, Epoxides, Thiols, Sulfides, and Disulfides
Introduction
This chapter explores the structure, nomenclature, physical properties, synthesis, and reactivity of ethers, epoxides, thiols, sulfides, and disulfides. These functional groups are essential in organic synthesis and biological chemistry, with unique reactivity and applications.
Ethers
Structure and Nomenclature
General Structure: Ethers have the formula R-O-R', where R and R' can be alkyl or aryl groups. Common examples include diethyl ether and anisole.
Common Names: Simple ethers are named by listing the groups attached to oxygen in alphabetical order, followed by "ether" (e.g., methyl vinyl ether, diphenyl ether).
IUPAC Nomenclature: The longest carbon chain is chosen as the parent, and the alkoxy group is named as a substituent (e.g., 2-chloro-5-ethoxyheptane).
Cyclic Ethers: Important solvents include tetrahydrofuran (THF) and dioxane.
Physical Properties
Boiling Points: Ethers have boiling points similar to alkanes of comparable size because they cannot hydrogen bond with themselves. The C–O bond dipole does not significantly affect boiling point.
Solubility: Ethers can hydrogen bond with water, making them moderately soluble. Their solubility is generally less than alcohols but much greater than alkanes.
Comparison Table:
Compound | Boiling Point (°C) | Solubility (g/100 mL water) |
|---|---|---|
Pentane | 36 | 0.002 |
Diethyl ether | 35 | 6.9 |
Methyl n-propyl ether | 37 | — |
n-Butanol | — | 7.3 |
Hexane | — | 0.002 |
Additional info: Alcohols are generally more soluble than ethers due to their ability to hydrogen bond with water, but the difference is often small for small molecules.
Spectroscopic Characterization
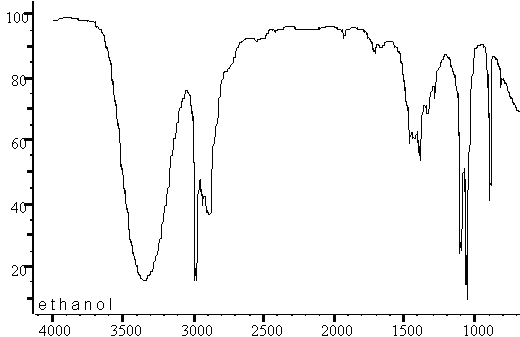
IR Spectroscopy: The C–O stretch appears at 1070–1150 cm–1 (not diagnostic).
NMR Spectroscopy: OCH protons appear at 3.3–4.0 ppm. If oxygen is present without a carbonyl or OH, it is likely an ether.

Synthesis of Ethers
Acid-Catalyzed Dehydration of Alcohols: Symmetrical ethers can be made by reacting alcohols with strong acid.
Alkoxymercuration/Demercuration: Addition of alcohols to alkenes using Hg(OAc)2 and NaBH4.
Williamson Ether Synthesis: SN2 reaction of alkyl halides with alkoxides. Works best with primary alkyl halides and alkoxides.
Solvolysis of Alkyl Halides: Useful for tertiary and activated secondary/primary halides.
Intramolecular Ether Synthesis: Formation of cyclic ethers (e.g., THF, dioxane) from dihalides and diols.
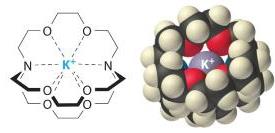
Crown Ethers and Cryptands
Crown Ethers: Cyclic polyethers that selectively bind metal cations, enhancing solubility of ionic compounds in nonpolar solvents.
Cryptands: More complex analogs of crown ethers, encapsulating ions more completely.

Reactions of Ethers
Stability: Ethers are generally inert and used as solvents, but can be cleaved by strong acids (HI > HBr > HCl).
Cleavage Mechanism: Proceeds via SN1 or SN2 depending on the structure; cannot stop at the half-way point.
Special Reagents: BBr3 and TMS-I are effective for ether cleavage.
Epoxides (Oxiranes)
Structure and Nomenclature
Structure: Epoxides are three-membered cyclic ethers, highly strained and reactive.
Nomenclature: Named as derivatives of oxirane, with the "epoxy" prefix, or as oxides of the corresponding alkene.
Physical Properties
Boiling Points: Higher than open-chain ethers due to larger dipoles and ring strain.
Bond Angles: The C–O–C bond angle is about 60°, causing significant angle strain.
Synthesis of Epoxides
From Alkenes: By halohydrin formation followed by base treatment, or by direct reaction with peracids (e.g., m-CPBA).
Asymmetric Epoxidation: Sharpless epoxidation uses chiral catalysts to produce enantioenriched epoxides from allylic alcohols.

Reactions of Epoxides
Ring Opening: Epoxides undergo ring-opening reactions with nucleophiles. The regio- and stereochemistry depend on the reaction conditions.
With Strong Nucleophiles: Attack occurs at the less substituted carbon (SN2-like).
With Weak Nucleophiles (acid-catalyzed): Attack occurs at the more substituted carbon due to carbocation-like transition state.
Stereochemistry: Opening of trans-epoxides gives meso compounds; cis-epoxides give racemic mixtures.
Thiols, Sulfides, and Disulfides
Thiols
Structure: Thiols (R–SH) are sulfur analogs of alcohols.
Acidity: Thiols are more acidic than alcohols (lower pKa), as sulfur stabilizes negative charge better than oxygen.
Compound | pKa |
|---|---|
Ethanol | 15.9 |
Ethyl mercaptan | 10.6 |
Phenol | 10.0 |
Thiophenol | 6.6 |
Acetic acid | 4.7 |
Thioacetic acid | 3.3 |
Additional info: The increased acidity is due to the larger size and polarizability of sulfur, which stabilizes the negative charge on the conjugate base.
Synthesis and Reactions of Thiols and Disulfides
Synthesis: Thiols can be synthesized by nucleophilic substitution of alkyl halides with HS–.
Oxidation: Thiols are easily oxidized to disulfides (R–S–S–R) by mild oxidants (e.g., O2, halogens).
Reduction: Disulfides can be reduced back to thiols by reducing agents (e.g., Zn/HCl, dithiothreitol).
Strong Oxidation: Leads to sulfonic acids (R–SO3H), which are strong acids and good leaving groups in SN2 reactions.
Sulfides and Their Oxidation
Sulfides (Thioethers): Analogous to ethers but with sulfur replacing oxygen. More nucleophilic and less basic than ethers.
Oxidation: Sulfides can be oxidized to sulfoxides (R–S(=O)–R') and sulfones (R–S(=O)2–R'). Dimethyl sulfoxide (DMSO) is a common solvent and oxidant.
Summary Table: Key Properties and Reactions
Functional Group | General Formula | Key Properties | Typical Reactions |
|---|---|---|---|
Ether | R–O–R' | Low reactivity, moderate solubility, inert solvent | Cleavage by strong acids |
Epoxide | Three-membered ring | Strained, highly reactive | Ring opening by nucleophiles |
Thiol | R–SH | More acidic than alcohols | Oxidation to disulfides |
Sulfide | R–S–R' | Good nucleophile | Oxidation to sulfoxide/sulfone |
Disulfide | R–S–S–R | Formed by oxidation of thiols | Reduction to thiols |
