Back
BackIntroduction to Organic Compounds: Structure, Classification, and Properties
Study Guide - Smart Notes

Chapter 3: Introduction to Organic Compounds
Overview of Alkanes and Cycloalkanes
Alkanes and cycloalkanes are the simplest organic compounds, consisting only of carbon and hydrogen atoms. Their chemical inertness is due to the strength and non-polarity of C–C and C–H bonds, making them primarily useful as molecular frameworks for attaching functional groups and as the basis for IUPAC nomenclature.
Alkanes: Acyclic hydrocarbons with the general formula .
Cycloalkanes: Cyclic hydrocarbons with ring structures.
Importance: Serve as molecular scaffolds and illustrate key concepts in organic structure.
Constitutional Isomerism
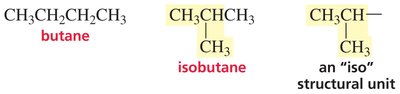
Constitutional isomers are compounds with the same molecular formula but different atom connectivity. This leads to distinct chemical and physical properties, such as boiling points, and contributes to the structural diversity of organic molecules.
Definition: Isomers with different bonding sequences.
Example: Butane and isobutane are constitutional isomers of C4H10.
Structural Diversity: The number of possible isomers increases rapidly with molecular size.
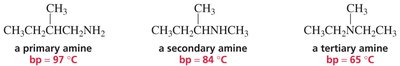
Classification of Alkyl Halides, Alcohols, and Amines
Organic compounds such as alkyl halides, alcohols, and amines are classified based on the number of carbon atoms attached to the functional group-bearing atom. This classification affects their chemical reactivity and physical properties.
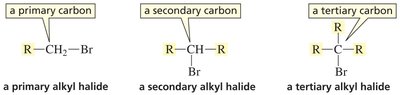
Alkyl Halides
Primary (1°): Halogen attached to a carbon bonded to one other carbon.
Secondary (2°): Halogen attached to a carbon bonded to two other carbons.
Tertiary (3°): Halogen attached to a carbon bonded to three other carbons.
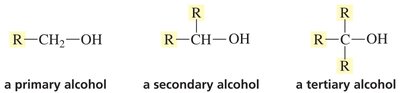
Alcohols
Primary (1°): OH group on a carbon bonded to one other carbon.
Secondary (2°): OH group on a carbon bonded to two other carbons.
Tertiary (3°): OH group on a carbon bonded to three other carbons.
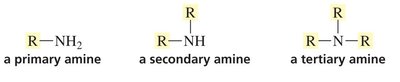
Amines
Primary (1°): Nitrogen bonded to one carbon.
Secondary (2°): Nitrogen bonded to two carbons.
Tertiary (3°): Nitrogen bonded to three carbons.
Physical Properties of Organic Compounds
The physical properties of organic compounds, such as boiling point, melting point, and solubility, are determined by the types and strengths of intermolecular forces present.
Boiling Point
Intermolecular Forces: van der Waals (dispersion), dipole-dipole, and hydrogen bonding.
van der Waals Forces: Present in all molecules; strength increases with surface area.
Dipole-Dipole Interactions: Occur in polar molecules; stronger than van der Waals.
Hydrogen Bonding: Strongest; occurs with N–H and O–H bonds.
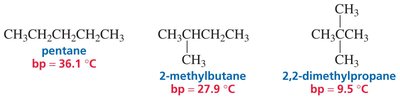
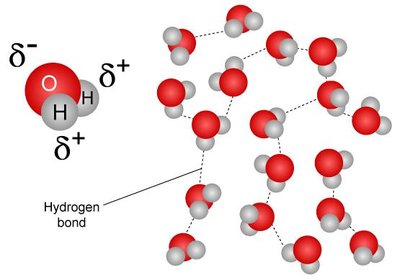
Boiling Point Trends: Molecules with more hydrogen bonding have higher boiling points.
Example: Ethanol (78°C) > Ethylamine (17°C) > Dimethyl ether (−24°C) > Propane (−42°C).

Melting Point
Melting point is influenced by how well molecules pack in the solid phase. Crystalline structures with efficient packing require more energy to melt.
Solubility
Polar Compounds: More soluble in water due to hydrogen bonding and dipole interactions.
Nonpolar/Large Compounds: Generally insoluble in water unless ionized.
Application: Basis for extraction techniques in organic chemistry.
Conformations of Organic Molecules
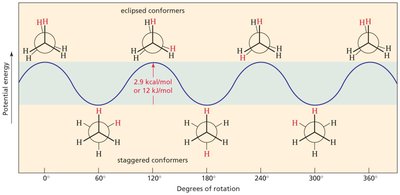
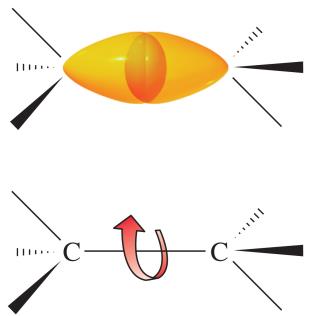
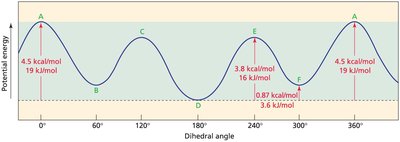
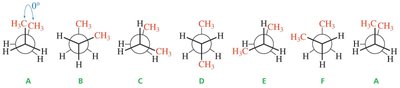
Conformations are different spatial arrangements of atoms resulting from rotation around single (σ) bonds. These are not isolable, as they rapidly interconvert at room temperature.
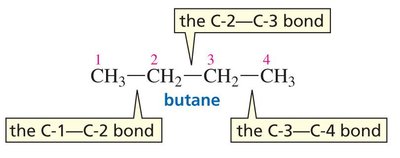
Newman Projections: Used to visualize conformations, such as in ethane and butane.
Eclipsed vs. Staggered: Eclipsed conformations are higher in energy due to torsional strain; staggered are lower in energy.
Butane: Multiple C–C bonds allow for more conformational diversity. Not all eclipsed or staggered conformations have the same energy due to steric strain.
Cycloalkanes: Structure and Strain
Cycloalkanes are ring-shaped alkanes that can experience angle strain when bond angles deviate from the ideal tetrahedral value (109.5°). This affects their stability and reactivity.
Angle Strain
Small Rings: Cyclopropane and cyclobutane have significant angle strain and eclipsed hydrogens.
Ring Puckering: Cyclobutane and cyclopentane adopt non-planar conformations to reduce strain.
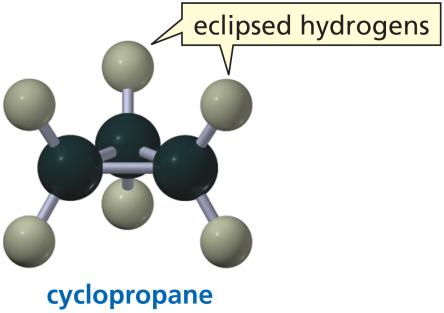
Non-Planarity: Cycloalkanes with four or more carbons are not planar.
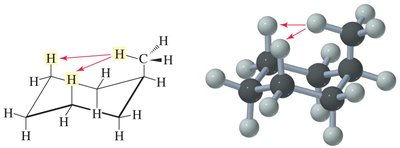
Cyclohexane Conformations
Chair and Boat: Cyclohexane adopts chair and boat conformations; chair is most stable due to minimal angle and torsional strain.
Axial and Equatorial Positions: Substituents can be axial (up/down) or equatorial (outward); large groups prefer equatorial positions to minimize steric hindrance.
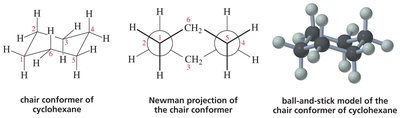
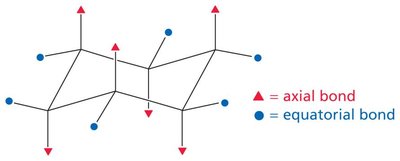
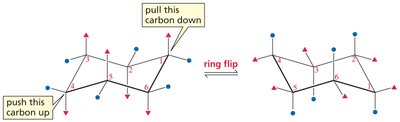
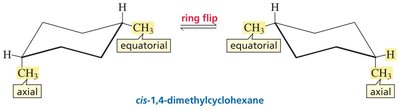
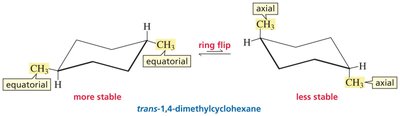
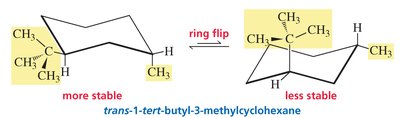
Ring Flip: Chair conformers interconvert via a ring flip, exchanging axial and equatorial positions.
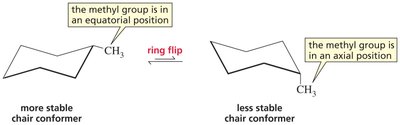
Stability: The most stable chair conformer places bulky substituents in equatorial positions.
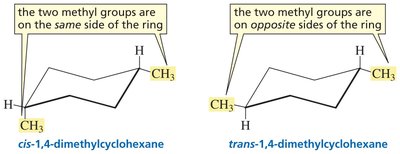
Cis-Trans Isomerism in Cycloalkanes
Cycloalkanes with two or more substituents can exhibit cis-trans isomerism, where substituents are on the same (cis) or opposite (trans) sides of the ring. These are stereoisomers, sharing the same connectivity but differing in spatial arrangement.
cis Isomer: Substituents on the same side of the ring.
trans Isomer: Substituents on opposite sides of the ring.
Stability: For 1,4-dimethylcyclohexane, cis isomers have equally stable conformations; trans isomers have one more stable conformation.
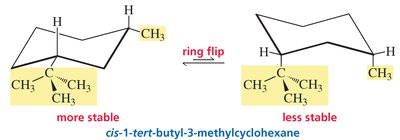
Bulky Groups: In 1-tert-butyl-3-methylcyclohexane, the tert-butyl group prefers the equatorial position in both cis and trans isomers.
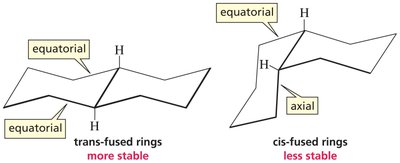
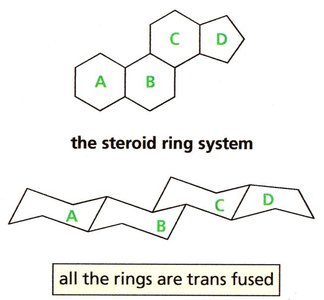
Fused Rings and Steroid Structures
Fused rings are compounds where two rings share two carbon atoms. The stereochemistry of fusion (cis or trans) affects the overall shape and stability of the molecule. In steroids, the four rings are fused in a trans manner, resulting in a flat structure suitable for embedding in cell membranes.
Trans-Fused: More stable, equatorial positions favored.
Cis-Fused: Less stable, axial positions present.