 Back
BackIonic and Molecular Compounds: Structure, Properties, and Bonding
Study Guide - Smart Notes

Ions and Ionic Compounds
Formation of Ions
Atoms form ions by gaining or losing electrons to achieve a stable electron configuration similar to noble gases. Metals typically lose electrons to form cations (positively charged ions), while non-metals gain electrons to form anions (negatively charged ions).
Cation: Formed when a metal loses electrons.
Anion: Formed when a non-metal gains electrons.
Isoelectronic: Species with the same number of electrons.
Example: Sodium (Na) loses one electron to become Na+, while chlorine (Cl) gains one electron to become Cl-.
The Octet Rule
Main group elements tend to achieve eight valence electrons (an octet) in their outer shell through chemical reactions. Metals lose electrons to resemble the noble gas before them; non-metals gain electrons to resemble the noble gas after them.
Octet Rule: Atoms react to achieve eight valence electrons.
Duet Rule: Hydrogen achieves stability with two valence electrons.
Example: Calcium (Ca) loses two electrons to form Ca2+ with a noble gas configuration.
Ionic Bonding and Properties
Ionic Bond Formation
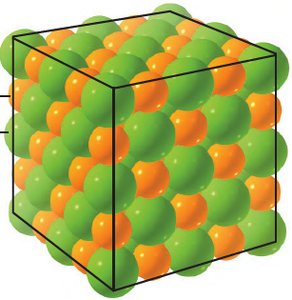
Ionic bonds are formed by the electrostatic attraction between cations and anions. The resulting compound is electrically neutral and typically forms a crystalline lattice.
Ionic Compound: Composed of a metal (cation) and a non-metal (anion).
Crystal Lattice: Regular arrangement of ions in a solid.
Example: Sodium chloride (NaCl) is formed from Na+ and Cl-.
Properties of Ionic Compounds
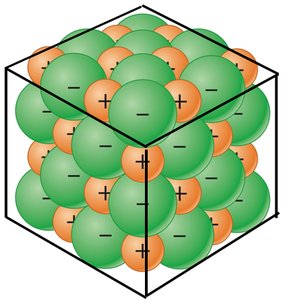
Ionic compounds exhibit characteristic physical properties due to strong electrostatic forces between ions.
Physical State: Solid at room temperature.
Conductivity: Conduct electricity when dissolved in water (aqueous solution).
Melting and Boiling Points: High due to strong ionic bonds.
Durability: Hard and brittle.

Naming Ions and Ionic Compounds
Naming Monatomic Cations
Cations are named by adding the word "ion" to the element name. If the metal can have multiple charges (Type II metals), a Roman numeral is used to indicate the charge.
Type I Metals: Have a single possible charge (e.g., Na+ is "sodium ion").
Type II Metals: Have multiple possible charges (e.g., Fe3+ is "iron(III) ion").
Naming Monatomic Anions
Anions are named by changing the ending of the element's name to "-ide" (e.g., Cl- is "chloride").
Base Name: The root of the nonmetal's name.
Naming Polyatomic Ions
Polyatomic ions are groups of atoms with an overall charge. Oxyanions (containing oxygen) are named based on the number of oxygens and their charge.
-ate: More oxygens (e.g., sulfate SO42-).
-ite: Fewer oxygens (e.g., sulfite SO32-).
Naming Ionic Compounds
Ionic compounds are named by writing the cation first, followed by the anion. If the cation is a Type II metal, its charge is indicated with a Roman numeral.
Example: FeCl3 is "iron(III) chloride".
Writing Formula Units of Ionic Compounds
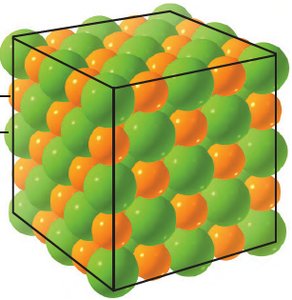
Formula Unit and Crystal Lattice
The formula unit represents the simplest ratio of ions in an ionic solid. Ionic solids exist as a crystal lattice, not as discrete molecules.
Formula Unit: Simplest ratio of ions (e.g., NaCl).
Crystal Lattice: Stable arrangement of cations and anions.
Covalent Bonding and Molecular Compounds
Covalent Bond Formation
Covalent bonds involve the sharing of valence electrons between nonmetal atoms to achieve a stable electron configuration.
Octet Rule: Atoms share electrons to achieve eight valence electrons.
Duet Rule: Hydrogen shares electrons to achieve two valence electrons.
Properties of Covalent Compounds
Covalent compounds differ from ionic compounds in their physical properties.
Physical State: Can be solid, liquid, or gas at room temperature.
Conductivity: Poor electrical conductors.
Melting and Boiling Points: Generally lower than ionic compounds.
Lewis Dot Structures
Drawing Lewis Dot Structures
Lewis dot structures represent the arrangement of valence electrons in molecules and ions. The goal is to satisfy the octet rule for each atom.
Count total valence electrons.
Place the least electronegative atom in the center.
Connect atoms with single bonds.
Add electrons to satisfy the octet rule.
Hydrogen only needs two electrons (duet rule).
Multiple Bonds
Double and triple bonds are formed when single bonds are insufficient to satisfy the octet rule.
Single Bond: One pair of shared electrons.
Double Bond: Two pairs of shared electrons.
Triple Bond: Three pairs of shared electrons.
Lewis Dot Structures for Ions
For ions, adjust the number of valence electrons according to the charge and enclose the structure in brackets with the charge indicated.
Cations: Fewer valence electrons.
Anions: More valence electrons.
Exceptions to the Octet Rule
Some elements can have fewer or more than eight electrons (incomplete or expanded octets), or possess an odd number of electrons (free radicals).
Incomplete Octet: Groups 2A and 3A.
Expanded Octet: Groups 5A, 6A, 7A, 8A.
Free Radicals: Odd number of valence electrons.
VSEPR Theory and Molecular Geometry
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the geometry of molecules by minimizing repulsion between electron groups around the central atom.
Electron Groups: Lone pairs and bonding pairs.
Molecular Shapes: Determined by the number of electron groups.
Electron Geometry
Electron geometry is based on the number of electron groups around the central atom.
2 groups: Linear
3 groups: Trigonal planar
4 groups: Tetrahedral
Molecular Geometry
Molecular geometry considers both bonding pairs and lone pairs to determine the true shape of the molecule.
2 groups: Linear
3 groups: Trigonal planar or bent
4 groups: Tetrahedral, trigonal pyramidal, or bent
Bond Angles
Bond angles are determined by the number of electron groups and the presence of lone pairs.
2 groups: 180°
3 groups: 120°
4 groups: 109.5° (decreases with lone pairs)
Polarity and Intermolecular Forces
Electronegativity and Dipole Moment
Electronegativity measures an atom's ability to attract electrons. A significant difference in electronegativity between atoms leads to polar bonds and dipole moments.
Polarity: Unequal sharing of electrons.
Dipole Moment: Arrow points toward the more electronegative atom.
Bond Classification: Pure covalent, polar covalent, or ionic based on ΔEN.
Molecular Polarity
Molecular polarity depends on both the polarity of individual bonds and the overall shape of the molecule.
Nonpolar Molecule: Perfect shape, no lone pairs on central atom.
Polar Molecule: Imperfect shape or polar bonds.
Intermolecular Forces
Intermolecular forces are attractions between molecules and determine many physical properties.
London Dispersion Forces: Present in all molecules; weakest.
Dipole-Dipole Forces: Between polar molecules.
Hydrogen Bonding: Strongest; occurs when H is bonded to F, O, or N.
Ion-Dipole Forces: Between ions and polar molecules.
Example: Water (H2O) exhibits hydrogen bonding, dipole-dipole, and London dispersion forces.
Type of Force | Exists Between | Strength | Example |
|---|---|---|---|
London Dispersion | All molecules | Weakest | CH4 |
Dipole-Dipole | Polar molecules | Moderate | HCl |
Hydrogen Bonding | H bonded to F, O, N | Strongest | H2O |
Ion-Dipole | Ions and polar molecules | Variable | NaCl & H2O |
Additional info: The notes cover foundational concepts in general chemistry relevant to organic chemistry, including bonding, molecular structure, and intermolecular forces, which are essential for understanding organic molecules and their behavior.
