 Back
BackKetones and Aldehydes: Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Ketones and Aldehydes
Classes of Carbonyl Compounds
Carbonyl compounds are a fundamental class of organic molecules characterized by the presence of a carbonyl group (C=O). The main classes include ketones, aldehydes, carboxylic acids, esters, acid chlorides, and amides. Ketones and aldehydes are central to organic chemistry due to their reactivity and prevalence in both biological and industrial contexts.
Ketones: Carbonyl group bonded to two carbon atoms.
Aldehydes: Carbonyl group bonded to at least one hydrogen atom.
Carboxylic acids, esters, acid chlorides, amides: Carbonyl group bonded to other functional groups.

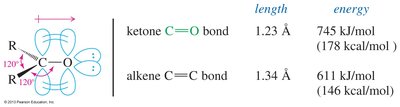
Structure of the Carbonyl Group
The carbonyl group consists of a carbon atom double-bonded to an oxygen atom. Both atoms are sp2 hybridized, resulting in a planar structure. The C=O bond is shorter, stronger, and more polar than the C=C bond found in alkenes.
Bond length: C=O is 1.23 Å; C=C is 1.34 Å.
Bond energy: C=O is 745 kJ/mol; C=C is 611 kJ/mol.
Polarity: The carbonyl group is highly polar, making the carbon electrophilic.
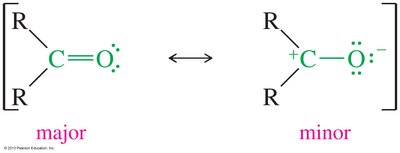
Resonance in Carbonyl Compounds
Carbonyl groups exhibit resonance, with the major resonance form having all atoms completing their octet and no formal charges. The carbonyl carbon is partially positive, making it susceptible to nucleophilic attack.
Major resonance form: Neutral, octet-complete.
Minor resonance form: Formal charges on carbon and oxygen.
Electrophilicity: Carbonyl carbon is an electrophile.
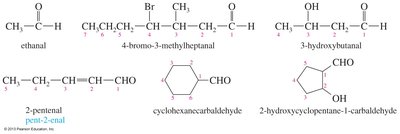
Nomenclature of Ketones and Aldehydes
Ketone Nomenclature
Ketones are named by replacing the alkane suffix '-e' with '-one' and numbering the chain to give the carbonyl carbon the lowest possible number. Cyclic ketones assign the carbonyl carbon as position 1, and the carbonyl takes precedence over double bonds.
Example: Acetone (propanone).
Aldehyde Nomenclature
Aldehydes are named by replacing the '-e' ending of the parent alkane with '-al'. The aldehyde carbon is always number 1. If attached to a ring, the suffix 'carbaldehyde' is used.
Example: Ethanal, pent-2-enal, cyclohexanecarbaldehyde.
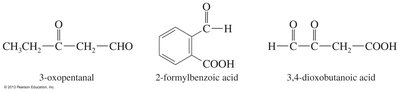
Carbonyl as Substituent
When a carbonyl group is not the highest priority functional group, it is named as a substituent: 'oxo' for ketones and 'formyl' for aldehydes. Aldehydes have higher priority than ketones.
Example: 3-oxopentanal, 2-formylbenzoic acid.
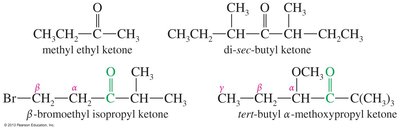
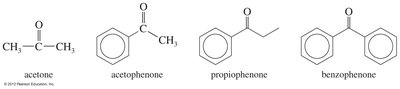
Common Names for Ketones and Aldehydes
Many ketones and aldehydes have historical common names, often based on their alkyl attachments or Greek letters for position. These names are important for recognizing compounds in both academic and industrial settings.
Example: Acetone, acetophenone, benzophenone.
Physical Properties
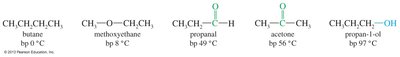
Boiling Points
Ketones and aldehydes are more polar than alkanes or ethers, resulting in higher boiling points. However, they cannot hydrogen-bond to each other, so their boiling points are lower than comparable alcohols.
Example: Acetone (bp 56°C), propan-1-ol (bp 97°C).
Solubility
Ketones and aldehydes are good solvents for alcohols. The lone pair of electrons on the oxygen atom can accept hydrogen bonds from O—H or N—H groups. Acetone and acetaldehyde are miscible with water.
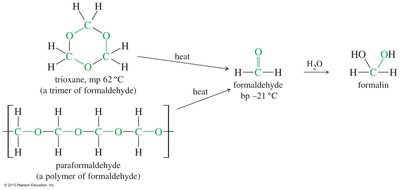
Formaldehyde
Formaldehyde is a gas at room temperature. Formalin is a 40% aqueous solution of formaldehyde, used in preservation and disinfection. Trioxane is a cyclic trimer of formaldehyde, generated under acid-catalyzed conditions, and can revert to formaldehyde upon heating.
Spectroscopic Properties
Infrared (IR) Spectroscopy
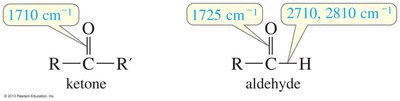
Ketones and aldehydes show a strong C=O stretch in IR spectroscopy. Ketones absorb around 1710 cm-1, while simple aldehydes absorb around 1725 cm-1. Aldehydes also show two C—H stretches at 2710 cm-1 and 2810 cm-1.
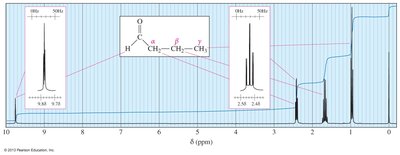
1H NMR Spectroscopy
Protons near the carbonyl group are more deshielded and appear at higher chemical shift values. The α, β, and γ protons show decreasing δ values with increasing distance from the carbonyl group.
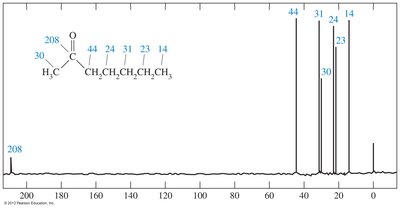
13C NMR Spectroscopy
The carbonyl carbon in ketones appears at high δ values (e.g., 208 ppm), while α carbons appear at lower values (e.g., 30–44 ppm).
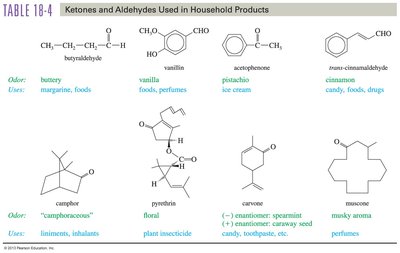
Industrial and Household Importance
Industrial Uses
Acetone and methyl ethyl ketone are common industrial solvents. Formaldehyde is used in polymer production (e.g., Bakelite) and as a food additive and flavoring agent.
Household Products
Ketones and aldehydes are found in many household products, contributing to odors and flavors. Examples include vanillin (vanilla), acetophenone (pistachio), and trans-cinnamaldehyde (cinnamon).
Compound | Odor | Uses |
|---|---|---|
Vanillin | Vanilla | Foods, perfumes |
Acetophenone | Pistachio | Ice cream |
trans-Cinnamaldehyde | Cinnamon | Candy, foods, drugs |
Camphor | Camphoraceous | Liniments, inhalants |
Carvone | Spearmint/caraway | Toothpaste, oils |
Synthesis of Ketones and Aldehydes
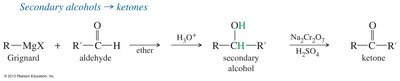
Grignard Reagents
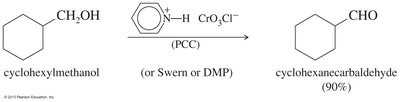
Grignard reagents can be used to synthesize alcohols, which are then oxidized to ketones or aldehydes. Secondary alcohols are oxidized to ketones, while primary alcohols are oxidized to aldehydes using selective oxidants like PCC or Swern oxidation.
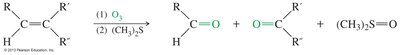
Ozonolysis of Alkenes
Ozonolysis cleaves double bonds in alkenes, producing ketones and aldehydes as products.
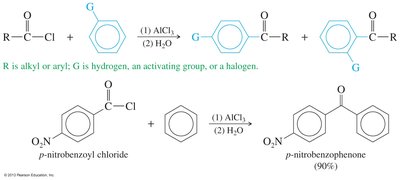
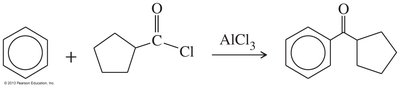
Friedel–Crafts Acylation
Reaction of an acyl halide with an aromatic ring produces a ketone, a key method for synthesizing aromatic ketones.
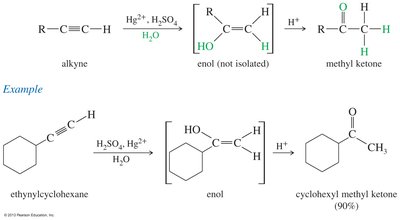
Hydration of Alkynes
Markovnikov hydration of alkynes produces enols, which rapidly tautomerize to ketones. Internal alkynes can yield mixtures of ketones.
Resonance Structures and Tautomers
Resonance structures involve electron movement, while tautomers are constitutional isomers differing by the position of protons and double bonds. Keto-enol tautomerism is a rapid equilibrium process.
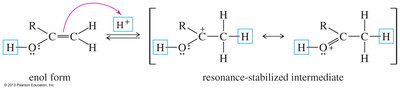
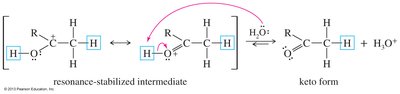
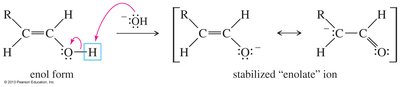
Mechanisms of Tautomerism
Acid-catalyzed tautomerism involves proton addition and loss, while base-catalyzed tautomerism involves deprotonation and reprotonation.
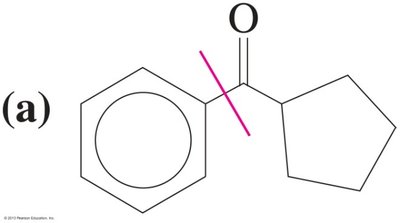
Solved Synthesis Problems
Ketones and aldehydes can be synthesized from smaller fragments using Grignard reactions, Friedel–Crafts acylation, or hydroboration-oxidation of alkynes. Epoxides can be used to add carbon atoms in Grignard reactions.
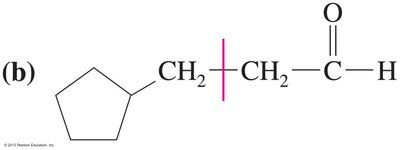
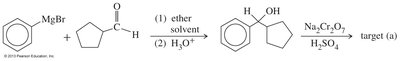
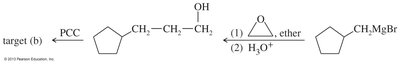
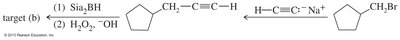
Ketones from Carboxylic Acids
Organolithium reagents attack carboxylate anions to form dianions, which upon protonation and dehydration yield ketones.
Ketones and Aldehydes from Nitriles
Grignard or organolithium reagents attack nitriles, forming imines that are hydrolyzed to ketones. Aluminum hydrides (DIBAL-H) can reduce nitriles to aldehydes.
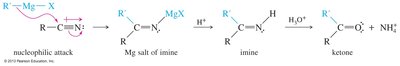
Aldehydes from Acid Chlorides
Lithium aluminum tri(t-butoxy)hydride is a mild reducing agent that selectively reduces acid chlorides to aldehydes.

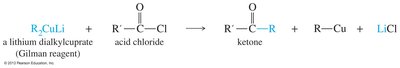
Lithium Dialkyl Cuprate Reagents
Lithium dialkylcuprates (Gilman reagents) transfer alkyl groups to acid chlorides, forming ketones. Grignards and organolithiums cannot perform this reaction.
Reactions of Ketones and Aldehydes
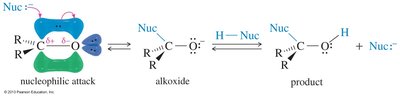
Nucleophilic Addition
Strong nucleophiles attack the carbonyl carbon, forming an alkoxide ion that is then protonated. Aldehydes are more reactive than ketones due to less steric hindrance and greater electrophilicity.
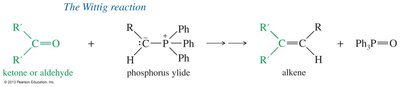
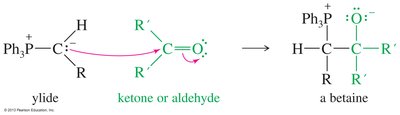
The Wittig Reaction
The Wittig reaction converts carbonyl groups into new C=C double bonds using phosphorus ylides. This reaction is important for constructing alkenes in organic synthesis.
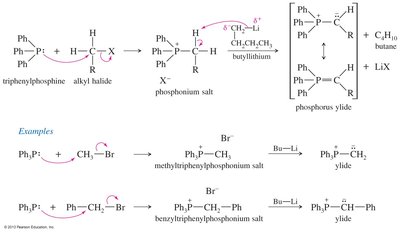
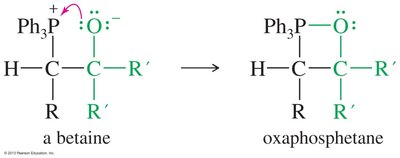
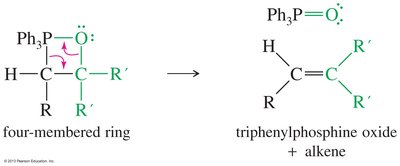
Preparation and Mechanism of Phosphorus Ylides
Phosphorus ylides are prepared from triphenylphosphine and alkyl halides via SN2, followed by deprotonation with butyllithium. The Wittig mechanism involves betaine and oxaphosphetane intermediates, ultimately forming an alkene and triphenylphosphine oxide.
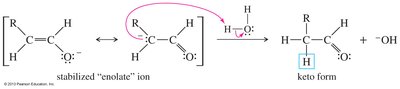
Hydration of Ketones and Aldehydes
In aqueous solution, ketones and aldehydes are in equilibrium with their hydrates (geminal diols). The equilibrium favors the keto form for ketones, but formaldehyde favors the hydrated form.

Acid- and Base-Catalyzed Hydration
Hydration occurs via nucleophilic addition, with water or hydroxide as the nucleophile. Acid catalysis activates the carbonyl, while base catalysis involves direct attack by hydroxide.
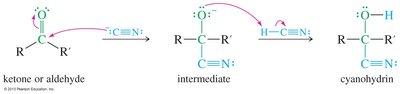
Cyanohydrin Formation
Cyanohydrins are formed by base-catalyzed nucleophilic addition of cyanide ion to the carbonyl group, followed by protonation. HCN is highly toxic.
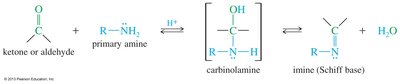
Formation of Imines
Ammonia or primary amines react with ketones or aldehydes to form imines (Schiff bases), which are nitrogen analogues of carbonyl compounds. Optimum pH for this reaction is around 4.5.
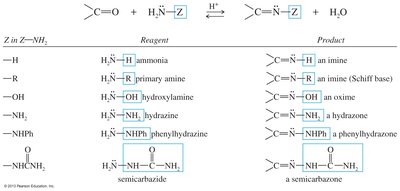
Other Condensations with Amines
Ketones and aldehydes can react with various amines to form oximes, hydrazones, phenylhydrazones, and semicarbazones, each with distinct reagents and products.
Reagent | Product |
|---|---|
Ammonia | Imine |
Primary amine | Imine (Schiff base) |
Hydroxylamine | Oxime |
Hydrazine | Hydrazone |
Phenylhydrazine | Phenylhydrazone |
Semicarbazide | Semicarbazone |
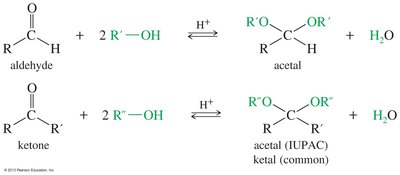
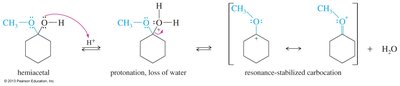
Formation and Hydrolysis of Acetals
Acetals are formed by acid-catalyzed reaction of aldehydes or ketones with alcohols. Cyclic acetals are formed by addition of diols and are used as protecting groups in synthesis. Acetals are stable in base but hydrolyze easily in acid.
Oxidation and Reduction of Ketones and Aldehydes
Oxidation of Aldehydes
Aldehydes are easily oxidized to carboxylic acids. The Tollens test is a classic method for detecting aldehydes, where silver ion is reduced to metallic silver.
Reduction Reagents
Sodium borohydride (NaBH4) reduces ketones to secondary alcohols and aldehydes to primary alcohols. Lithium aluminum hydride (LiAlH4) is a stronger reducing agent, capable of reducing carboxylic acids and derivatives. Catalytic hydrogenation can reduce carbonyls and unsaturated bonds.
Deoxygenation: Clemmensen and Wolff–Kishner Reductions
Clemmensen and Wolff–Kishner reductions are used to remove oxygen from ketones and aldehydes, converting them to hydrocarbons.
Summary Table: Key Reactions and Properties
Reaction | Reagent | Product |
|---|---|---|
Oxidation of primary alcohol | PCC, Swern | Aldehyde |
Oxidation of secondary alcohol | Na2Cr2O7, H2SO4 | Ketone |
Ozonolysis of alkene | O3, (CH3)2S | Ketone/Aldehyde |
Friedel–Crafts acylation | Acyl chloride, AlCl3 | Aromatic ketone |
Hydration of alkyne | HgSO4, H2SO4 | Ketone |
Reduction of nitrile | DIBAL-H | Aldehyde |
Reduction of acid chloride | LiAlH(O-t-Bu)3 | Aldehyde |
Reduction of ketone/aldehyde | NaBH4, LiAlH4 | Alcohol |
