 Back
BackNitriles and Carboxylic Acids: Structure, Synthesis, and Spectroscopy
Study Guide - Smart Notes

Nitriles: Structure, Nomenclature, and Synthesis
Introduction to Nitriles
Nitriles are organic compounds containing the cyano group (-C≡N). They are valuable synthetic intermediates in organic chemistry, though relatively few occur naturally. Nitriles are found in some natural products, such as calyculin A (anticancer activity), lotuastralin (toxin), and bursatellin.
Structure: The nitrile group consists of a carbon triple-bonded to a nitrogen atom.
Applications: Used in the synthesis of amines, carboxylic acids, and other functional groups.
Nomenclature of Nitriles
Nitriles are named based on the parent alkane, with the suffix -nitrile added. The nitrile carbon is included in the main chain. For cyclic compounds, the suffix -carbonitrile is used, replacing -carboxylic acid. When a higher-priority group is present, the nitrile is named as a cyano substituent. Some nitriles have common names, such as acetonitrile and benzonitrile.
Example: CH3CN is called acetonitrile.
Example: C6H5CN is called benzonitrile.
Preparation of Nitriles
Nitriles can be synthesized by several methods:
1. SN2 Displacement with Cyanide Ion: Alkyl halides react with NaCN to form nitriles. This method is limited to primary and secondary alkyl halides due to steric hindrance in SN2 reactions. The reaction results in inversion of stereochemistry and a one-carbon extension of the chain.
2. Dehydration of 1° Amides: Primary amides can be dehydrated using reagents like SOCl2 or POCl3 to yield nitriles. This method is more general and not limited by steric constraints.
Reactions of Nitriles
General Reactivity
The chemistry of nitriles is governed by the polarization of the C≡N bond, similar to carbonyl compounds. Nucleophilic addition is a common reaction pathway.
Hydrolysis of Nitriles
Nitriles can be hydrolyzed to carboxylic acids under acidic or basic conditions. Acidic hydrolysis is milder and preferred, while basic hydrolysis is harsher and may yield amide intermediates.
Acidic Hydrolysis:
Basic Hydrolysis:
Mechanism (Acidic Conditions): Protonation of the nitrile, nucleophilic attack by water, and subsequent steps yield the carboxylic acid.
Mechanism (Basic Conditions): Hydroxide attacks the nitrile, forming an amide intermediate, which is further hydrolyzed to the acid.
Reduction of Nitriles
Nitriles can be reduced to primary amines or aldehydes, depending on the reducing agent:
With LiAlH4: (primary amine)
With DIBAH: (aldehyde)
Reaction with Grignard Reagents
Nitriles react with Grignard reagents to form ketones after hydrolysis:
Spectroscopic Identification of Nitriles and Carboxylic Acids
IR Spectroscopy
Infrared (IR) spectroscopy is a powerful tool for identifying functional groups:
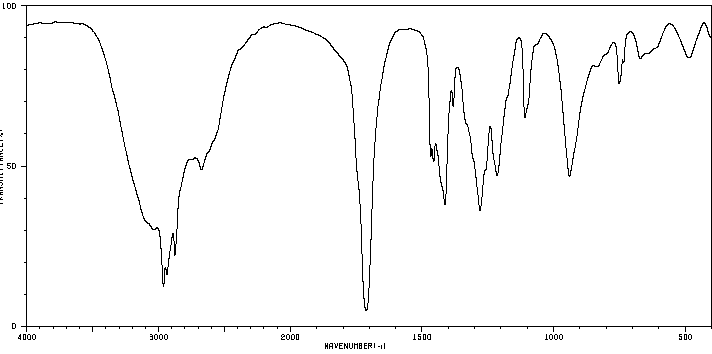
Carboxylic Acids: Broad O-H stretch (2500–3300 cm-1), strong C=O stretch (1710–1760 cm-1).
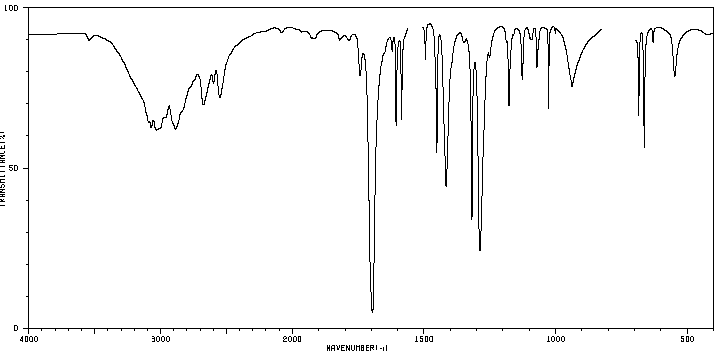
Nitriles: Sharp C≡N stretch (2230–2250 cm-1), highly diagnostic.
Example IR Spectra:
Benzoic acid: Shows broad O-H and strong C=O stretches.
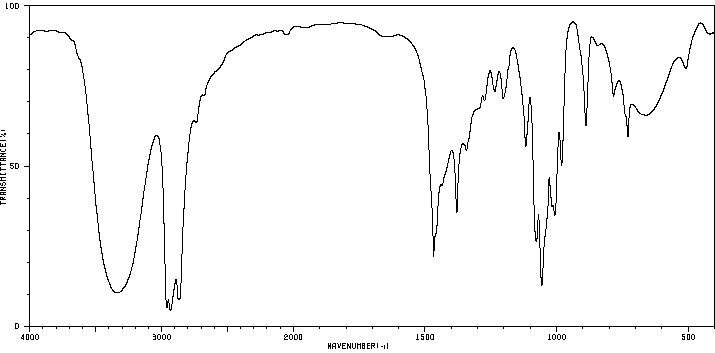
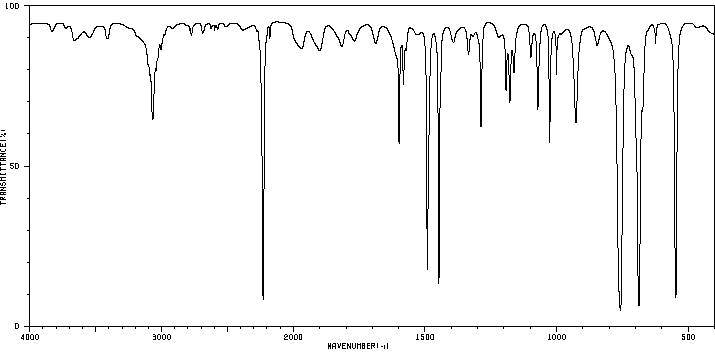
Pentanoic acid and 1-pentanol: Comparison highlights the presence of carboxylic acid vs. alcohol functional groups.
Benzonitrile: Shows a sharp C≡N stretch.
1H NMR Spectroscopy
Protons adjacent to carbonyl or nitrile groups are slightly deshielded, appearing at δ 2.0–2.6 (carboxylic acids) or δ 2.0–2.7 (nitriles). Carboxylic acid O-H protons appear downfield (δ 10.5–12.0) and are exchangeable with D2O. Acidic impurities can broaden or eliminate the O-H signal.
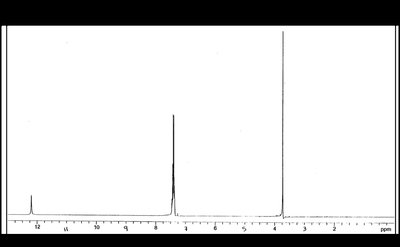
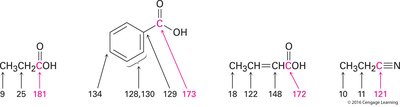
13C NMR Spectroscopy
Carboxylic acid carbons resonate at δ 165–185, while nitrile carbons appear at δ 115–130, distinct from C=O signals.
Mass Spectrometry
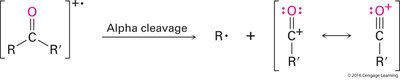
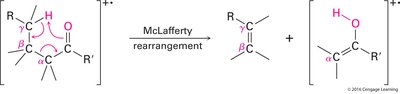
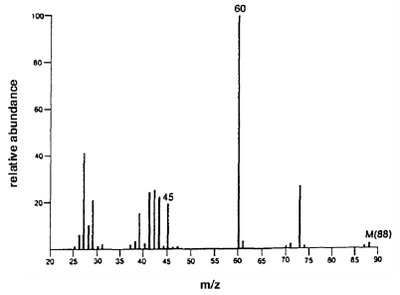
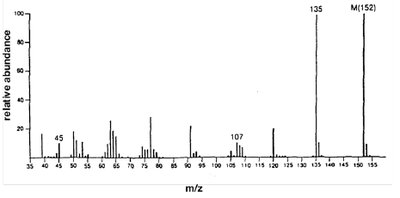
Carboxylic acids show two characteristic fragmentation modes:
Alpha Cleavage: Produces an acylium ion.
McLafferty Rearrangement: Occurs in acids with gamma hydrogens, yielding a neutral molecule and a rearranged ion.
Example Mass Spectra:
Butanoic acid (MW = 88): Shows characteristic peaks for fragmentation.
p-Methoxybenzoic acid (MW = 152): Shows molecular ion and key fragment peaks.
Summary Table: Key Spectroscopic Features
Functional Group | IR (cm-1) | 1H NMR (δ, ppm) | 13C NMR (δ, ppm) | MS Fragmentation |
|---|---|---|---|---|
Carboxylic Acid | O-H: 2500–3300 (broad) C=O: 1710–1760 (strong) | α-CH: 2.0–2.6 O-H: 10.5–12.0 (variable) | 165–185 | Alpha cleavage, McLafferty rearrangement |
Nitrile | C≡N: 2230–2250 (sharp) | α-CH: 2.0–2.7 | 115–130 | Alpha cleavage |
Conclusion
Nitriles and carboxylic acids are important functional groups in organic chemistry, with distinct methods of synthesis and characteristic spectroscopic signatures. Mastery of their reactivity and identification is essential for advanced study and practical applications in organic synthesis.
