 Back
BackNuclear Magnetic Resonance (NMR) Spectroscopy: Principles and Interpretation
Study Guide - Smart Notes

Nuclear Magnetic Resonance Spectroscopy
Introduction to NMR
Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful analytical technique used to determine the structure of organic compounds. It provides detailed information about the chemical environment of nuclei, especially hydrogen (1H) and carbon (13C), in a molecule. NMR is non-destructive and requires only a small sample. - Key Nuclei Studied: 1H, 13C, 15N, 19F, 31P - Sample Preparation: Samples are placed in specialized tubes and inserted into the NMR spectrometer. 

Basic Principles of NMR
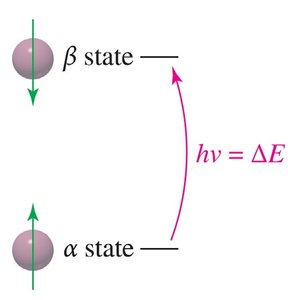
NMR relies on the interaction between nuclear spins and an external magnetic field. Nuclei with an odd atomic or mass number possess a nuclear spin, generating a magnetic moment. When placed in a magnetic field, these nuclei align either with (alpha-spin, lower energy) or against (beta-spin, higher energy) the field. - Resonance: When irradiated with radio-frequency photons matching the energy difference (ΔE) between spin states, nuclei absorb energy and transition between states. - Free Induction Decay (FID): The absorbed energy is released as a decaying signal, which is converted to a spectrum via Fourier Transform. 
Magnetic Field and Energy States
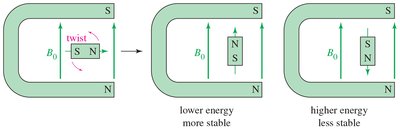
- Magnetic Moment: The spinning nucleus acts like a tiny bar magnet. - External Magnetic Field (B0): Aligns the nuclear magnets, creating distinct energy states. - Alpha-spin: Aligned with the field, lower energy. - Beta-spin: Aligned against the field, higher energy. 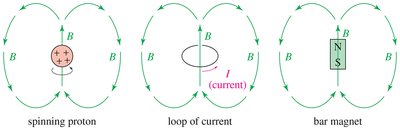
Magnetic Shielding and Chemical Environment
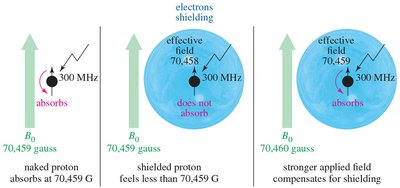
The local chemical environment affects the magnetic field experienced by nuclei. Electrons surrounding the nucleus create an induced magnetic field that opposes the external field, shielding the nucleus. - Shielded Nucleus: Requires a stronger external field to achieve resonance. - Deshielded Nucleus: Absorbs at a lower field due to less electron shielding. 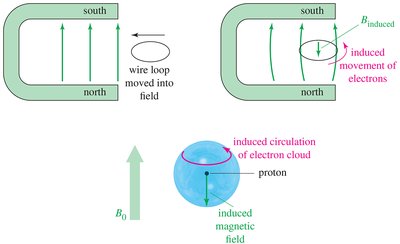
Proton NMR (1H NMR)
Interpreting Proton NMR Spectra
Proton NMR provides information about the number and types of protons in a molecule, their chemical environment, and their interactions with neighboring protons. - Number of Signals: Indicates the number of distinct proton environments. - Chemical Shift: Position of signals (in ppm) reveals the electronic environment. - Integration: Area under each signal is proportional to the number of protons. - Splitting (Multiplicity): Shape of signals shows the number of adjacent protons. 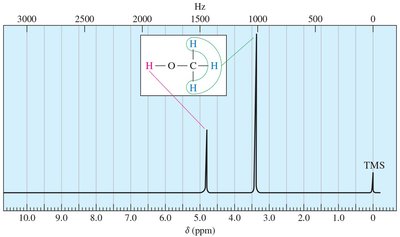
Chemical Shift (δ)
Chemical shift is measured in parts per million (ppm) and is independent of the spectrometer used. Tetramethylsilane (TMS) is used as an internal standard (0.00 ppm). - Formula: 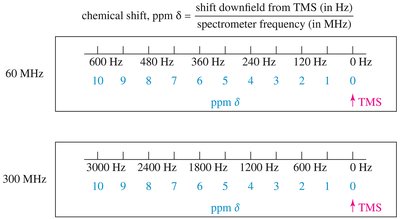
Factors Affecting Chemical Shift
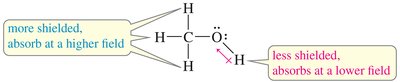
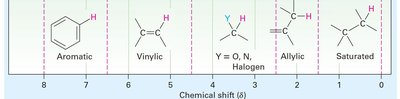
- Electronegativity: Electronegative atoms (O, N, halogens) deshield protons, shifting signals downfield. - Spatial Orientation: Proximity to electron-withdrawing groups increases deshielding. 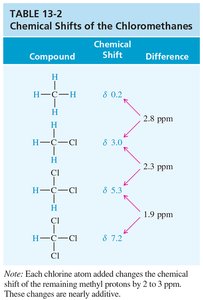
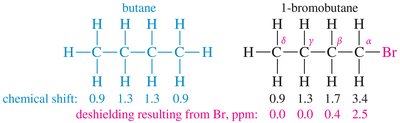
Special Cases: Aromatic, Alkene, Alkyne, and Aldehyde Protons
- Aromatic Protons: Deshielded by ring current, appear at δ 7–8. 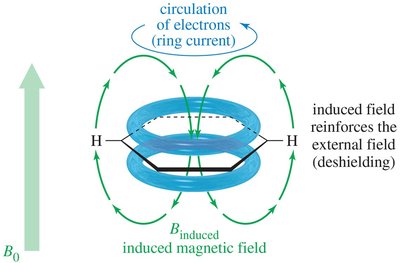 - Alkene Protons: Deshielded by pi electrons, appear at δ 5–6.
- Alkene Protons: Deshielded by pi electrons, appear at δ 5–6. 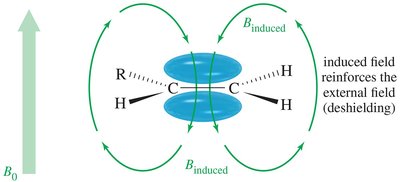 - Alkyne Protons: Shielded, appear at δ 2.5.
- Alkyne Protons: Shielded, appear at δ 2.5. 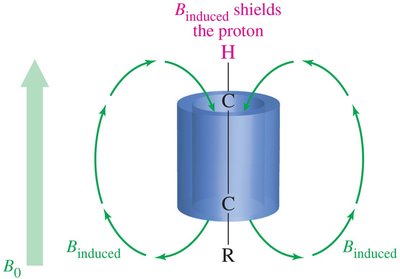 - Aldehyde Protons: Strongly deshielded, appear at δ 9–10.
- Aldehyde Protons: Strongly deshielded, appear at δ 9–10. 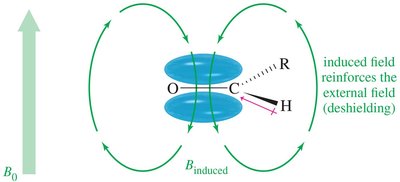
O-H and N-H Protons
- Hydrogen Bonding: Causes broadening and shifting of peaks. - Carboxylic Acid O-H: Appears at δ > 10 due to strong deshielding.
Integration: Quantifying Protons
The area under each NMR signal is proportional to the number of protons contributing to that signal. - Example: In tert-butyl methyl ether, the tert-butyl hydrogens produce a signal three times larger than the methyl hydrogens. 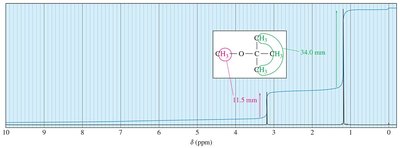
Spin-Spin Splitting (Multiplicity)
Signals are split into multiplets due to interactions with adjacent, nonequivalent protons. - N + 1 Rule: A signal split by N equivalent protons appears as N + 1 peaks. - Common Multiplets: Singlet, doublet, triplet, quartet, etc. 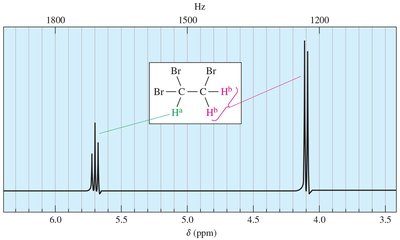
Examples of Multiplet Patterns
- Ethyl Group: CH3 signal is a triplet; CH2 signal is a quartet. 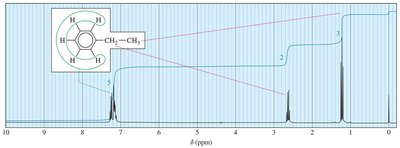 - Isopropyl Group: CH signal is a septet; CH3 signals are doublets.
- Isopropyl Group: CH signal is a septet; CH3 signals are doublets. 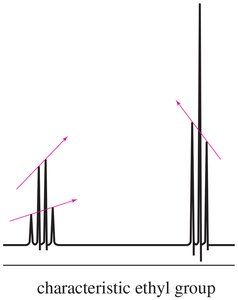
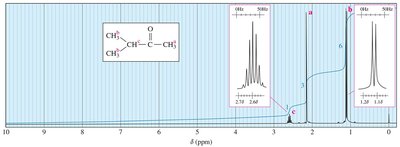
Coupling Constants (J)
The coupling constant (J) is the distance between peaks in a multiplet, measured in Hz. It is independent of the external field strength and provides information about the spatial relationship between protons. 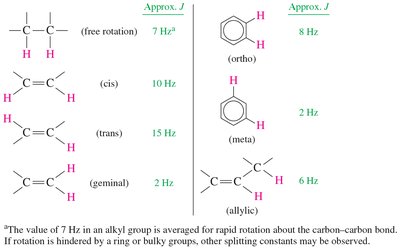
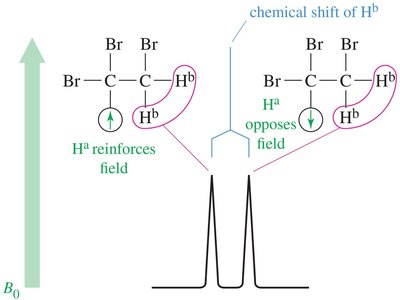
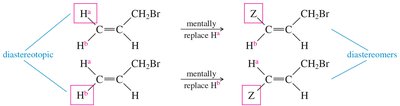
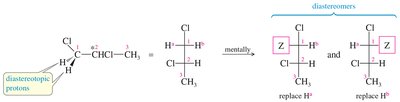
Stereochemical Nonequivalence and Diastereotopic Protons
Protons in a CH2 group may be nonequivalent if replacing each with a different group produces stereoisomers. Diastereotopic protons can split each other, leading to complex splitting patterns. 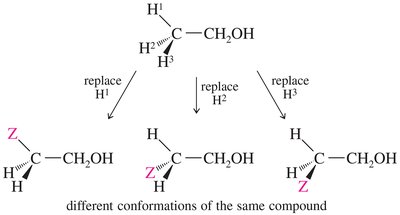
Carbon-13 NMR (13C NMR)
Principles of 13C NMR
Carbon-13 is only 1% of natural carbon and has a magnetic spin. 13C NMR provides information about the number and types of carbon atoms in a molecule. - Resonance Frequency: About one-fourth that of hydrogen. - Peak Areas: Not proportional to the number of carbons. - Spin-Spin Splitting: 13C rarely splits due to adjacent 13C, but can couple with attached protons.
Proton Spin Decoupling
To simplify spectra, protons are irradiated so that each carbon gives a single, unsplit peak.
Off-Resonance Decoupling
13C nuclei are split only by directly attached protons, following the N + 1 rule.
Interpreting 13C NMR Spectra
- Number of Signals: Indicates the number of distinct carbon environments. - Chemical Shift: Reveals the type of functional group. - Splitting Pattern: Indicates the number of attached protons.
Advanced NMR Techniques
2D and 3D NMR
- 2D NMR: Correlates proton-proton (H-H COSY) and carbon-proton (C-H COSY) interactions. - 3D NMR (MRI): Used in medical imaging to visualize internal structures noninvasively.
Summary Table: NMR Signal Interpretation
Feature | Interpretation |
|---|---|
Number of signals | Number of distinct environments |
Chemical shift (δ, ppm) | Electronic environment, functional group |
Integration | Relative number of protons |
Multiplicity (splitting) | Number of adjacent protons (N + 1 rule) |
Coupling constant (J, Hz) | Spatial relationship between protons |
Practice and Application
By understanding the principles and interpretation of NMR spectra, students can deduce the structure of unknown organic compounds using chemical shifts, integration, and splitting patterns. Example: Given a molecular formula and NMR data, propose a structure and verify it against the spectral evidence. Additional info: This guide expands on brief slide points to provide a comprehensive, self-contained overview of NMR spectroscopy for organic chemistry students.
