 Back
BackOrganic Chemistry Exam 1 Practice – Study Notes and Mechanisms
Study Guide - Smart Notes

Organic Chemistry Exam 1 Practice – Study Notes
General Chemistry Review
This section covers foundational concepts necessary for understanding organic chemistry, including atomic structure, periodic trends, and basic chemical principles.
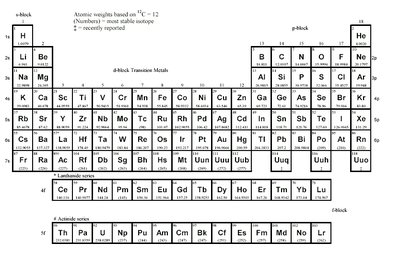
Periodic Table: The periodic table organizes elements by increasing atomic number and groups elements with similar chemical properties.
Atomic Number (Z): Number of protons in the nucleus of an atom.
Atomic Mass: Weighted average mass of an element's isotopes, based on 12C = 12.
Groups and Periods: Vertical columns are groups (similar valence electron configurations); horizontal rows are periods.
Blocks: s-block, p-block, d-block, and f-block refer to the type of atomic orbital being filled.
Organic Molecules and Intermolecular Forces
Organic molecules are primarily composed of carbon and hydrogen, often with oxygen, nitrogen, halogens, and other elements. Their physical and chemical properties are influenced by intermolecular forces.
Functional Groups: Specific groups of atoms within molecules that determine chemical reactivity (e.g., alcohols, alkenes, alkynes, carboxylic acids).
Intermolecular Forces: Include hydrogen bonding, dipole-dipole interactions, and London dispersion forces.
Acidity and Basicity: Alkynes are weakly acidic; carboxylic acids are more acidic and can be deprotonated by strong bases.
Stereochemistry
Stereochemistry focuses on the spatial arrangement of atoms in molecules and how this affects their properties and reactions.
Isomers: Molecules with the same molecular formula but different structures.
Types of Isomers:
Enantiomers: Non-superimposable mirror images.
Diastereomers: Stereoisomers that are not mirror images.
Constitutional Isomers: Differ in connectivity of atoms.
Identical: Same structure and connectivity.
Chirality: A molecule is chiral if it cannot be superimposed on its mirror image.
Alkanes, Alkenes, and Alkynes
Hydrocarbons are classified based on the types of bonds between carbon atoms.
Alkanes: Saturated hydrocarbons with single bonds.
Alkenes: Unsaturated hydrocarbons with at least one double bond. Stability increases with more substituents on the double bond.
Alkynes: Unsaturated hydrocarbons with at least one triple bond. Terminal alkynes are weakly acidic.
Acids and Bases
Acid-base reactions are fundamental in organic chemistry, affecting reactivity and mechanisms.
Acid Strength: Measured by pKa; lower pKa means stronger acid.
Deprotonation: Strong bases (e.g., NaNH2) can deprotonate weak acids like alkynes.
Solvents: Water is not ideal for deprotonating alkynes due to its own acidity.
Radical Reactions
Radical reactions involve species with unpaired electrons and are important in halogenation and other organic transformations.
Radical Stability: Allylic and benzylic radicals are more stable due to resonance.
Halide Radicals: Fluoride radicals are highest in energy; iodide radicals are lowest.
Mechanism Example: Radical monochlorination of ethane involves initiation, propagation, and termination steps.
Mechanisms and Synthesis
Understanding reaction mechanisms is crucial for predicting products and designing synthetic routes.
Mechanistic Arrows: Show movement of electrons during reactions.
Resonance Forms: Multiple valid Lewis structures for a molecule; the most stable form is usually the one with the most delocalized charge.
Synthetic Strategies: Involve choosing appropriate reagents and intermediates to construct target molecules.
Nomenclature
Systematic naming of organic compounds follows IUPAC conventions to ensure clarity and consistency.
Alkenes and Alkynes: Number the longest chain containing the double or triple bond; indicate position and configuration (E/Z, R/S).
Halides: Prefixes for halogen substituents (bromo-, chloro-, etc.) and their positions.
Cycloalkenes: Indicate ring size and substituent positions.
Functional Groups
Recognizing and naming functional groups is essential for understanding reactivity and mechanisms.
Common Functional Groups: Alcohols, alkenes, alkynes, carboxylic acids, halides, amines, ethers.
Identification: Circle and name functional groups in complex molecules for practice.
Tables
The periodic table is a key reference for atomic numbers, weights, and element classification.
Block | Elements | Properties |
|---|---|---|
s-block | Groups 1-2, H, He | Alkali and alkaline earth metals; highly reactive |
p-block | Groups 13-18 | Nonmetals, metalloids, halogens, noble gases |
d-block | Transition metals | Variable oxidation states, colored compounds |
f-block | Lanthanides, actinides | Rare earth and radioactive elements |
Example Equations
Deprotonation of Terminal Alkyne:
Radical Halogenation (Initiation):
Radical Halogenation (Propagation):
